Scalable Metal-Free Synthesis of 5H-1,4-Benzodiazepines for Commercial API Manufacturing
Introduction to Advanced Benzodiazepine Synthesis Technology
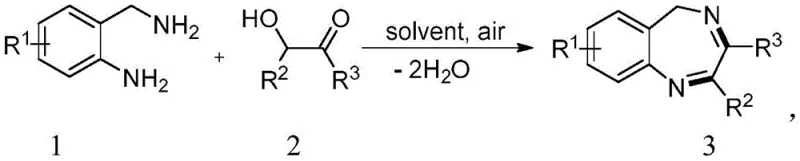
The pharmaceutical industry is constantly seeking more efficient and environmentally benign pathways for constructing complex heterocyclic scaffolds, particularly those serving as critical cores for bioactive molecules. Patent CN111925334B introduces a groundbreaking methodology for the synthesis of 5H-1,4-benzodiazepine compounds, a privileged structure found in numerous therapeutic agents ranging from anxiolytics to anticonvulsants. This innovation specifically addresses the long-standing challenges associated with traditional cyclization methods by leveraging a tandem oxidation-cyclization sequence driven solely by molecular oxygen from the air. By eliminating the dependency on stoichiometric oxidants or precious metal catalysts, this technology represents a paradigm shift towards sustainable chemical manufacturing, offering a robust solution for the production of high-purity pharmaceutical intermediates.
The significance of this patent lies in its ability to streamline the synthetic route while simultaneously enhancing the safety profile of the manufacturing process. Conventional approaches often rely on harsh conditions or toxic reagents that complicate downstream purification and waste management. In contrast, the disclosed method utilizes readily available o-aminobenzylamine derivatives and 2-hydroxy-2-phenylacetophenones as starting materials, reacting them in a solvent acid medium under an air atmosphere. This approach not only simplifies the operational procedure but also ensures that the only byproduct generated is water, thereby achieving excellent atom economy. For R&D directors and process chemists, this translates to a cleaner reaction profile that minimizes the formation of difficult-to-remove impurities, facilitating faster scale-up and regulatory approval.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the 1,4-benzodiazepine ring system has been achieved through various strategies, each carrying inherent drawbacks that hinder industrial application. Traditional methods often involve the condensation of o-aminobenzylamines with benzils or the cyclization with 1,2-disubstituted alkynes, processes that frequently necessitate elevated temperatures and the use of excess oxidizing agents. These harsh conditions can lead to the degradation of sensitive functional groups and the generation of complex impurity profiles that are costly to separate. Furthermore, many established protocols rely on transition metal catalysts, which introduce the risk of heavy metal contamination in the final active pharmaceutical ingredient (API). Removing trace metals to meet stringent ICH Q3D guidelines requires additional purification steps, such as scavenging or recrystallization, which inevitably reduce overall yield and increase production costs.
Another significant limitation of prior art is the reliance on specialized or hazardous oxidants that pose safety risks during large-scale operations. The handling of strong oxidizing agents requires specialized equipment and strict safety protocols, adding to the capital expenditure and operational complexity of the manufacturing facility. Additionally, multi-step sequences are often required to prepare the necessary precursors, extending the lead time for material availability and increasing the cumulative environmental footprint of the synthesis. For supply chain managers, these inefficiencies translate into higher volatility in raw material costs and potential bottlenecks in production scheduling. The need for a more direct, safer, and economically viable synthetic route has therefore become a critical priority for modern pharmaceutical manufacturing.
The Novel Approach
The novel approach detailed in patent CN111925334B overcomes these obstacles by employing a direct oxidative cyclization strategy that utilizes atmospheric oxygen as the terminal oxidant. This method enables the one-pot conversion of o-aminobenzylamines and 2-hydroxy-2-phenylacetophenones into the target 5H-1,4-benzodiazepine scaffold with remarkable efficiency. The reaction is promoted by simple carboxylic acids, such as acetic acid, which serve a dual role as both the solvent and the catalyst, thereby eliminating the need for exotic ligands or additives. By operating under an air balloon or open air conditions, the process harnesses an inexhaustible and free source of oxidation power, drastically reducing the cost of goods sold (COGS) associated with reagent procurement. This green chemistry principle not only aligns with global sustainability goals but also simplifies the engineering controls required for the reaction vessel.
Moreover, the substrate scope of this new methodology is exceptionally broad, accommodating a wide variety of electronic and steric environments on both the amine and ketone components. As demonstrated in the patent examples, substituents such as methyl, methoxy, halogens (chloro, bromo, iodo, fluoro), and even electron-withdrawing groups like trifluoromethyl and cyano are well-tolerated, yielding the corresponding products in high isolated yields ranging from 70% to over 95%. This versatility allows medicinal chemists to rapidly access diverse libraries of analogs for structure-activity relationship (SAR) studies without needing to re-optimize reaction conditions for each new derivative. The robustness of the reaction under relatively mild thermal conditions (80°C to 120°C) further enhances its appeal for commercial scale-up, ensuring consistent quality and reproducibility across different batch sizes.
Mechanistic Insights into Air-Oxidative Tandem Cyclization
The mechanistic pathway of this transformation involves a sophisticated cascade of condensation and oxidation events orchestrated by the acidic medium and molecular oxygen. Initially, the nucleophilic attack of the primary amine group of the o-aminobenzylamine on the carbonyl carbon of the 2-hydroxy-2-phenylacetophenone leads to the formation of an imine intermediate. This step is facilitated by the protonation of the carbonyl oxygen by the acetic acid solvent, which increases the electrophilicity of the ketone. Subsequently, the secondary amine moiety within the same molecule attacks the adjacent hydroxyl-bearing carbon, promoting a cyclization event that closes the seven-membered diazepine ring. This intramolecular condensation results in a dihydro-intermediate that retains the saturation at the C2-C3 position, requiring a final oxidative aromatization step to achieve the fully conjugated 5H-1,4-benzodiazepine system.
The critical role of air in this mechanism cannot be overstated, as it serves as the hydrogen acceptor to drive the final dehydrogenation. Molecular oxygen abstracts hydrogen atoms from the dihydro-intermediate, regenerating the aromaticity of the newly formed heterocycle and releasing water as the sole byproduct. This oxidative step is thermodynamically favorable and proceeds smoothly under the heated reaction conditions without the need for external radical initiators or metal co-catalysts. The absence of metal species ensures that the reaction mixture remains free from transition metal residues, a key advantage for pharmaceutical applications where metal limits are strictly regulated. Understanding this mechanism allows process engineers to optimize oxygen mass transfer, for instance by ensuring adequate headspace or gentle stirring, to maintain consistent reaction rates and prevent the accumulation of partially oxidized intermediates.
How to Synthesize 5H-1,4-Benzodiazepine Efficiently
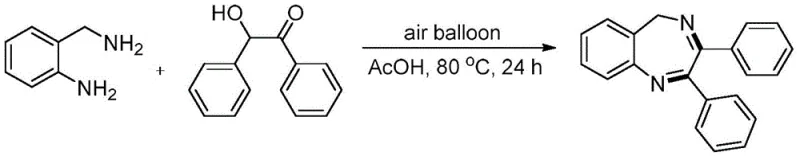
Implementing this synthesis in a laboratory or pilot plant setting requires careful attention to the stoichiometry and reaction environment to maximize yield and purity. The standard protocol involves charging a pressure tube or reactor with the o-aminobenzylamine and the 2-hydroxy-2-phenylacetophenone derivative in a molar ratio that slightly favors the amine, typically around 1.5 equivalents relative to the ketone. Acetic acid is then added in a substantial excess, acting as the reaction medium, with volumes typically ranging from 10 to 15 equivalents relative to the limiting reagent. An air balloon is attached to the vessel to maintain a constant supply of oxygen while preventing the ingress of moisture or contaminants, and the mixture is heated to between 80°C and 120°C. Reaction progress is monitored via TLC or GC-MS, with completion generally achieved within 24 to 48 hours depending on the steric bulk of the substrates.
- Charge a reaction vessel with o-aminobenzylamine and 2-hydroxy-2-phenylacetophenone in a molar ratio of approximately 1.5: 1.
- Add glacial acetic acid as both solvent and promoter, typically using 10-15 equivalents relative to the ketone substrate.
- Attach an air balloon to the vessel and heat the mixture to 80-120°C for 24-48 hours until conversion is complete.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this metal-free air oxidation technology offers profound advantages for procurement and supply chain management within the fine chemical and pharmaceutical sectors. The most immediate benefit is the drastic reduction in raw material costs associated with the elimination of expensive transition metal catalysts and specialized ligands. Traditional cross-coupling or oxidation reactions often rely on palladium, copper, or rhodium complexes, which are subject to significant price volatility and supply constraints. By replacing these with inexpensive acetic acid and ambient air, manufacturers can stabilize their input costs and insulate themselves from market fluctuations in precious metal prices. Furthermore, the simplicity of the reagent profile simplifies inventory management, reducing the need for storing hazardous oxidants or sensitive catalysts under inert atmospheres.
Supply chain reliability is further enhanced by the robustness and scalability of the process. The reaction conditions are mild enough to be performed in standard glass-lined or stainless steel reactors without requiring specialized high-pressure equipment capable of handling dangerous gases like pure oxygen. This lowers the barrier to entry for contract manufacturing organizations (CMOs) and allows for flexible production scheduling across multiple facilities. The high atom economy and the generation of water as the only byproduct significantly reduce the volume of chemical waste requiring treatment, leading to lower disposal costs and a smaller environmental footprint. For supply chain heads, this translates into a more resilient and sustainable sourcing strategy that aligns with corporate social responsibility (CSR) objectives while ensuring continuous availability of critical intermediates for downstream API synthesis.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts removes the need for costly metal scavenging steps and extensive purification protocols, directly lowering the cost of goods sold. Additionally, the use of air as a free oxidant replaces expensive stoichiometric oxidants, resulting in substantial savings on reagent procurement. The simplified workup procedure reduces solvent consumption and labor hours, contributing to overall operational efficiency and margin improvement.
- Enhanced Supply Chain Reliability: Utilizing commodity chemicals like acetic acid and o-aminobenzylamines ensures a stable supply of raw materials, as these are produced globally in large volumes. The process does not depend on single-source specialty reagents, mitigating the risk of supply disruptions. The mild reaction conditions also reduce equipment wear and tear, minimizing unplanned downtime and maintenance costs, thereby ensuring consistent delivery schedules to customers.
- Scalability and Environmental Compliance: The green nature of this synthesis, characterized by water as the sole byproduct, facilitates easier compliance with increasingly stringent environmental regulations. The absence of heavy metals simplifies the regulatory filing process for new drug applications, accelerating time-to-market. The process is inherently safe and scalable, allowing for seamless transition from gram-scale R&D to ton-scale commercial production without significant re-engineering of the process parameters.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthetic methodology. These answers are derived directly from the experimental data and claims presented in patent CN111925334B, providing clarity on the practical aspects of the technology. Understanding these details is crucial for project managers and technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios. The responses highlight the versatility, safety, and efficiency of the air-oxidative approach compared to legacy methods.
Q: Does this synthesis method require transition metal catalysts?
A: No, the method described in patent CN111925334B operates without any transition metal catalysts or ligands, utilizing acetic acid as a promoter and air as the oxidant.
Q: What are the typical reaction conditions for this benzodiazepine synthesis?
A: The reaction is typically conducted in acetic acid at temperatures ranging from 80°C to 120°C for a duration of 24 to 48 hours under an air atmosphere.
Q: Is this process suitable for large-scale pharmaceutical manufacturing?
A: Yes, the absence of expensive metals and the use of air as a safe oxidant make this process highly scalable and compliant with strict pharmaceutical impurity guidelines.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5H-1,4-Benzodiazepine Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this metal-free synthesis technology for the production of high-value pharmaceutical intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from the laboratory bench to full-scale manufacturing. Our state-of-the-art facilities are equipped to handle air-sensitive and oxidative reactions with the highest standards of safety and quality control. We are committed to delivering products that meet stringent purity specifications, leveraging our rigorous QC labs to verify that every batch is free from residual metals and impurities, thus safeguarding the integrity of your final drug product.
We invite you to collaborate with us to leverage this innovative synthetic route for your next development program. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and timeline. By partnering with NINGBO INNO PHARMCHEM, you gain access to not just a supplier, but a strategic ally dedicated to optimizing your supply chain. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise can drive efficiency and reliability in your 5H-1,4-benzodiazepine sourcing strategy.
