Advanced Enzymatic Synthesis of (R)-Praziquantel for Commercial Scale API Production
The pharmaceutical industry has long recognized the critical importance of chirality in drug efficacy, particularly in the realm of antiparasitic agents where the (R)-enantiomer of praziquantel demonstrates superior therapeutic activity compared to its racemic counterpart. Patent CN103333942A introduces a groundbreaking synthetic methodology that leverages enzymatic dynamic kinetic resolution to produce optically pure (R)-praziquantel intermediates with exceptional efficiency. This technology represents a paradigm shift from traditional chemical synthesis, utilizing the high stereospecificity of lipases in conjunction with green ionic liquid solvents to overcome decades-old industrial challenges. By integrating biocatalysis with conventional organic transformations, this route not only achieves product purity exceeding 98% but also establishes a robust foundation for the scalable manufacturing of high-quality active pharmaceutical ingredients. For global procurement teams and R&D directors, this innovation offers a tangible solution to the historical difficulties associated with purifying praziquantel, ensuring a more reliable supply of this essential medicine.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of praziquantel has relied on synthetic routes that involve harsh reaction conditions and the utilization of hazardous chemical reagents, posing significant risks to both operational safety and environmental compliance. Traditional pathways often necessitate the use of toxic substances such as potassium cyanide and hexamethylene tetramine, which require stringent handling protocols and complex waste treatment systems to mitigate their harmful impact. Furthermore, the conventional chemical resolution methods frequently suffer from suboptimal yields and limited stereoselectivity, resulting in a significant portion of the inactive or potentially objectionable (S)-enantiomer that must be separated and discarded. This inefficiency not only drives up the cost of goods sold but also complicates the supply chain by introducing additional purification steps that extend lead times. The reliance on volatile organic solvents and heavy metal catalysts in these legacy processes further exacerbates the environmental footprint, making it increasingly difficult for manufacturers to meet modern regulatory standards for green chemistry.
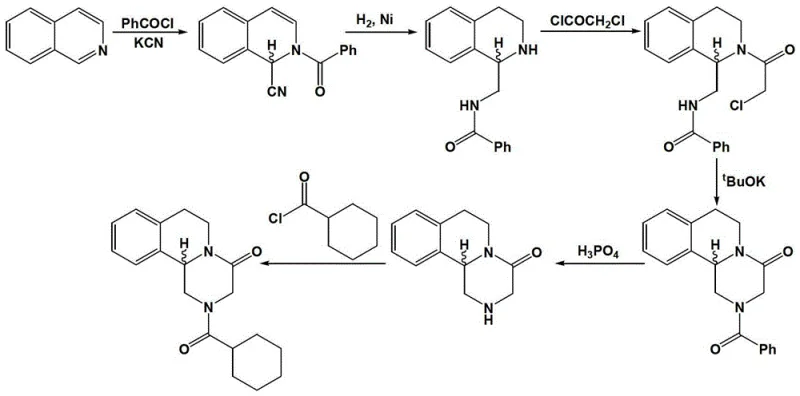
The Novel Approach
In stark contrast to these legacy methods, the novel enzymatic approach described in the patent data utilizes a sophisticated dynamic kinetic resolution strategy that transforms the racemic mixture directly into the desired optically active intermediate with high precision. By employing specific lipases, such as Candida antarctica lipase B or Candida rugosa, within a water-saturated ionic liquid medium, the process achieves a level of stereocontrol that is unattainable through standard chemical catalysis. This biocatalytic system operates under mild temperature conditions, typically ranging from 20°C to 50°C, which significantly reduces energy consumption and minimizes the degradation of sensitive intermediates. The use of ionic liquids not only enhances the stability and activity of the enzyme but also facilitates the easy separation of the product from the reaction mixture, allowing for the potential recycling of the solvent system. This methodological advancement effectively resolves the thirty-year industrial bottleneck of purifying left-handed praziquantel, offering a cleaner, safer, and more economically viable pathway for large-scale production.
Mechanistic Insights into Lipase-Catalyzed Dynamic Kinetic Resolution
The core of this technological breakthrough lies in the precise mechanistic interaction between the lipase enzyme and the racemic tetrahydroisoquinoline ester substrate within the unique microenvironment provided by the ionic liquid. The lipase exhibits high regioselectivity and stereoselectivity, preferentially hydrolyzing one enantiomer of the ester to yield the corresponding chiral carboxylic acid while leaving the other enantiomer intact or allowing it to racemize in situ under the basic conditions provided by additives like TBAH. This dynamic kinetic resolution ensures that the theoretical yield can approach 100%, as the unwanted enantiomer is continuously converted into the desired form rather than being wasted. The ionic liquid serves as a crucial component, stabilizing the transition state of the enzymatic reaction and preventing enzyme denaturation, which is a common issue in traditional organic solvents. The reaction proceeds through a well-defined catalytic cycle where the enzyme-substrate complex forms, undergoes hydrolysis, and releases the chiral acid product, which is then immediately available for downstream processing without extensive purification.
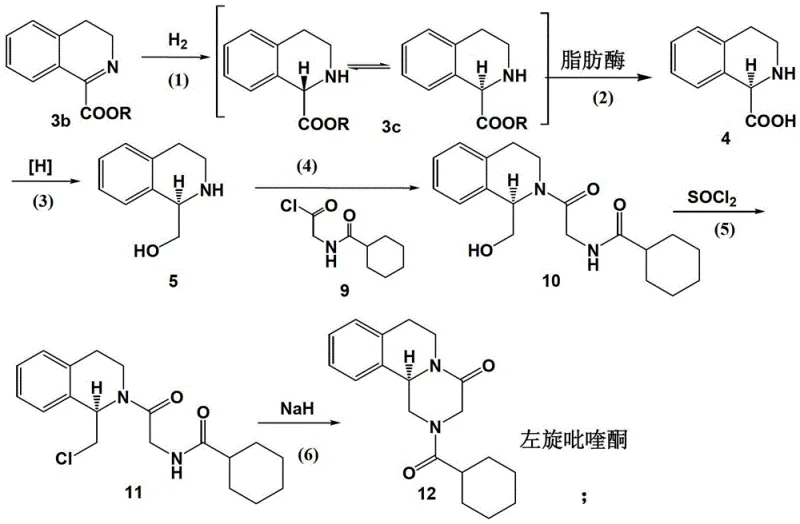
Controlling the impurity profile in the synthesis of chiral pharmaceutical intermediates is paramount for ensuring patient safety and meeting regulatory specifications, and this enzymatic route offers distinct advantages in this regard. The high specificity of the biocatalyst minimizes the formation of side products that are commonly associated with non-selective chemical reagents, thereby simplifying the downstream purification process. The use of a membrane reactor system further enhances purity by retaining the enzyme within the reaction zone while allowing the product and unreacted substrate to pass through, preventing protein contamination in the final API. Additionally, the mild reaction conditions prevent the thermal degradation of intermediates, which can often lead to the formation of difficult-to-remove impurities in high-temperature chemical processes. The resulting intermediate, characterized by its specific structural motif as shown in the patent data, possesses an optical purity greater than 98% e.e., setting a new standard for quality in praziquantel manufacturing.
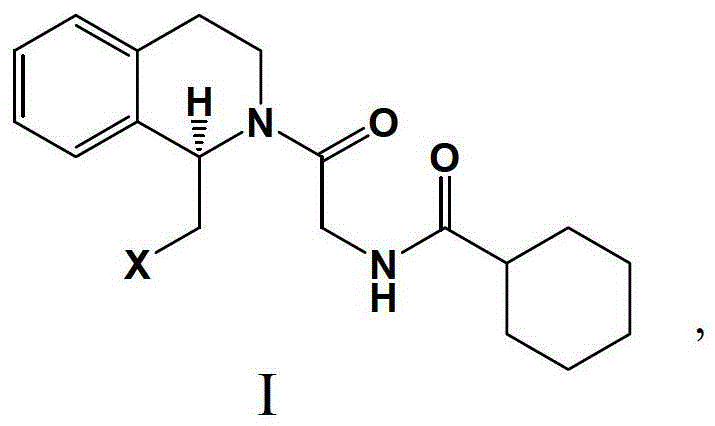
How to Synthesize (R)-Praziquantel Efficiently
The implementation of this synthesis route involves a sequence of well-coordinated steps beginning with the catalytic hydrogenation of dihydroisoquinoline esters to form the racemic tetrahydroisoquinoline intermediate, which serves as the substrate for the biocatalytic resolution. Following the enzymatic step, the chiral acid is reduced to the corresponding alcohol using borane or sodium borohydride, followed by acylation and a final base-mediated cyclization to close the piperazine ring and form the target molecule. Each step is optimized for maximum yield and minimal waste, utilizing recoverable catalysts and solvents to enhance the overall sustainability of the process. The detailed standardized synthesis steps, including specific molar ratios, temperature profiles, and workup procedures, are outlined in the technical guide below to ensure reproducibility and compliance with GMP standards.
- Catalytic hydrogenation of dihydroisoquinoline ester derivatives using Pd/C or Raney Nickel to form racemic tetrahydroisoquinoline intermediates.
- Enzymatic dynamic kinetic resolution using lipase (e.g., CAL-B) in water-saturated ionic liquids to isolate the optically pure (R)-acid intermediate.
- Reduction of the chiral acid to alcohol, followed by acylation and base-mediated cyclization to yield high-purity (R)-praziquantel.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this enzymatic synthesis route translates into substantial strategic advantages that extend beyond mere technical performance, impacting the overall cost structure and reliability of the supply base. The elimination of toxic reagents and the reduction in purification steps lead to a drastic simplification of the manufacturing process, which inherently lowers the operational expenditure associated with waste disposal and safety compliance. By utilizing a dynamic kinetic resolution, the process maximizes the utilization of raw materials, effectively doubling the yield of the desired chiral intermediate compared to traditional resolution methods where half the material is discarded. This efficiency gain directly contributes to cost reduction in API manufacturing, allowing for more competitive pricing without compromising on quality. Furthermore, the robustness of the enzymatic system ensures consistent batch-to-batch quality, reducing the risk of production delays caused by failed batches or out-of-specification results.
- Cost Reduction in Manufacturing: The transition to an enzymatic process eliminates the need for expensive and hazardous chemical resolving agents and heavy metal catalysts, which significantly reduces the raw material costs and the expenses related to their safe handling and disposal. The ability to recycle the ionic liquid solvent and retain the enzyme in a membrane reactor further decreases the consumption of consumables, leading to substantial cost savings over the lifecycle of the product. Additionally, the higher overall yield means that less starting material is required to produce the same amount of final API, optimizing the capital efficiency of the production facility. These factors combine to create a leaner manufacturing model that is less susceptible to fluctuations in the prices of specialty chemicals.
- Enhanced Supply Chain Reliability: The use of readily available raw materials and stable biocatalysts ensures a resilient supply chain that is less dependent on scarce or geopolitically sensitive reagents. The scalability of the enzymatic process, demonstrated by its successful operation in membrane reactors, allows for flexible production volumes that can be adjusted to meet market demand without significant lead time penalties. The simplified workflow reduces the number of critical process parameters that need to be monitored, lowering the risk of operational failures that could disrupt supply continuity. This reliability is crucial for maintaining the steady flow of essential antiparasitic medications to global markets, particularly in regions where disease burden is high.
- Scalability and Environmental Compliance: The green chemistry principles embedded in this synthesis route, such as the avoidance of toxic cyanides and the use of recyclable solvents, facilitate easier regulatory approval and environmental compliance across different jurisdictions. The process is designed for commercial scale-up, with proven strategies for managing heat transfer and mixing in larger reactors, ensuring that the quality observed at the laboratory scale is maintained at the tonnage level. The reduction in hazardous waste generation aligns with corporate sustainability goals and reduces the liability associated with environmental remediation. This makes the technology not only commercially attractive but also socially responsible, appealing to stakeholders who prioritize ethical manufacturing practices.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this enzymatic synthesis technology, providing clarity on its operational feasibility and benefits. These insights are derived directly from the patent specifications and experimental data, ensuring that the information provided is accurate and relevant for decision-makers evaluating this technology for adoption. Understanding these details is essential for assessing the fit of this process within existing manufacturing frameworks and for planning the necessary adjustments to infrastructure or sourcing strategies.
Q: How does the enzymatic method improve purity compared to traditional chemical resolution?
A: The enzymatic dynamic kinetic resolution utilizes high stereoselectivity of lipases in ionic liquids, achieving optical purity greater than 98% e.e., whereas traditional chemical methods often struggle with lower selectivity and require complex recrystallization steps.
Q: Is the ionic liquid solvent system recyclable for industrial scale-up?
A: Yes, the process employs a membrane reactor system where the lipase is retained, and the ionic liquid can be recovered and reused for subsequent batches, significantly reducing solvent waste and operational costs.
Q: What are the safety advantages of this route over the conventional praziquantel synthesis?
A: This novel route eliminates the need for highly toxic reagents such as potassium cyanide and hexamethylene tetramine used in traditional pathways, thereby enhancing workplace safety and simplifying environmental compliance.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (R)-Praziquantel Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this enzymatic synthesis route and have invested heavily in the infrastructure required to bring such advanced technologies to commercial fruition. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from development to full-scale manufacturing. Our facilities are equipped with state-of-the-art biocatalysis reactors and rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of (R)-praziquantel meets the highest international standards. We are committed to leveraging our technical expertise to optimize this process further, delivering cost-effective and high-quality solutions that support your global supply chain needs.
We invite you to collaborate with us to explore how this innovative synthesis method can enhance your product portfolio and improve your market competitiveness. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating the tangible economic benefits of switching to this greener, more efficient route. Please contact us to request specific COA data and route feasibility assessments, and let us help you secure a sustainable and reliable supply of this critical pharmaceutical intermediate.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
