Advanced Graphene Oxide Catalysis for High-Purity 3,5-Diiodo Salicyloyl Chloride Manufacturing
The landscape of pharmaceutical intermediate manufacturing is constantly evolving, driven by the relentless pursuit of higher purity, improved atom economy, and sustainable processing conditions. A significant breakthrough in this domain is documented in patent CN115073285A, which discloses a novel synthetic method for 3,5-diiodo salicyloyl chloride, a critical building block in the synthesis of various bioactive molecules and biochemical reagents. This technology represents a paradigm shift from traditional high-temperature acyl chlorination processes to a sophisticated, catalyst-driven approach utilizing graphene oxide. For R&D directors and process chemists, the introduction of this nanomaterial catalyst offers a compelling solution to long-standing challenges regarding selectivity and energy consumption. The patent details a robust protocol that not only enhances reaction kinetics but also ensures the structural integrity of sensitive functional groups, thereby delivering a product of exceptional quality suitable for demanding downstream applications in drug discovery and development.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of acyl chlorides from salicylic acid derivatives has been plagued by significant operational inefficiencies and selectivity issues. As illustrated in prior art, such as the work by Wuyuelin et al., the conventional route typically relies on refluxing 3,5-diiodosalicylic acid with thionyl chloride in high-boiling solvents like chlorobenzene at temperatures reaching 132°C. 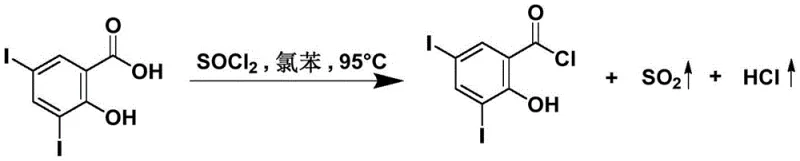 This aggressive thermal environment poses severe risks, primarily the unwanted chlorination of the phenolic hydroxyl group, which leads to complex impurity profiles that are difficult to separate. Furthermore, the use of chlorobenzene creates substantial downstream processing burdens; its high boiling point necessitates energy-intensive distillation steps for solvent removal, drastically increasing production costs and cycle times. The reported yields for these legacy methods often struggle to exceed 70%, with purities capping around 98%, which is frequently insufficient for high-value pharmaceutical applications requiring stringent impurity control. Additionally, the corrosive nature of the reaction mixture at elevated temperatures accelerates equipment degradation, leading to increased maintenance costs and potential safety hazards in a commercial plant setting.
This aggressive thermal environment poses severe risks, primarily the unwanted chlorination of the phenolic hydroxyl group, which leads to complex impurity profiles that are difficult to separate. Furthermore, the use of chlorobenzene creates substantial downstream processing burdens; its high boiling point necessitates energy-intensive distillation steps for solvent removal, drastically increasing production costs and cycle times. The reported yields for these legacy methods often struggle to exceed 70%, with purities capping around 98%, which is frequently insufficient for high-value pharmaceutical applications requiring stringent impurity control. Additionally, the corrosive nature of the reaction mixture at elevated temperatures accelerates equipment degradation, leading to increased maintenance costs and potential safety hazards in a commercial plant setting.
The Novel Approach
In stark contrast, the methodology outlined in CN115073285A leverages the unique properties of graphene oxide to facilitate a much milder and more efficient transformation. By introducing graphene oxide as a heterogeneous catalyst, the reaction can proceed effectively at temperatures between 40°C and 65°C, a dramatic reduction from the >130°C required previously. This温和 condition is pivotal for preserving the phenolic hydroxyl group, ensuring that the thionyl chloride reacts exclusively with the carboxylic acid functionality. The new process utilizes lower-boiling solvents such as dichloromethane or mixed systems with hexane, which are far easier to recover and recycle, thereby simplifying the workup procedure to a simple rotary evaporation. The result is a streamlined workflow that delivers 3,5-diiodo salicyloyl chloride with yields approaching 99.6% and purities up to 99.9%. This leap in performance metrics underscores the transformative potential of nanocatalysis in fine chemical synthesis, offering a cleaner, faster, and more reliable alternative to classical thermal activation.
Mechanistic Insights into Graphene Oxide-Catalyzed Acyl Chlorination
The efficacy of this novel synthesis lies in the intricate interaction between the graphene oxide catalyst and the reactants. Graphene oxide is characterized by a two-dimensional carbon lattice decorated with oxygen-containing functional groups, such as epoxides, hydroxyls, and carboxyls. These groups serve as active sites that can interact with thionyl chloride and the carboxylic acid substrate, effectively lowering the activation energy barrier for the nucleophilic acyl substitution. Unlike traditional Lewis acid catalysts that might promote indiscriminate reactivity, the surface chemistry of graphene oxide appears to provide a microenvironment that favors the formation of the acyl chloride intermediate while suppressing side reactions. This selective acceleration allows the reaction to reach completion rapidly once the solution clarifies, typically within 1 to 3 hours, indicating a highly efficient conversion rate. The catalyst essentially acts as a molecular scaffold, organizing the reactants in a manner that facilitates the expulsion of sulfur dioxide and hydrogen chloride gases, driving the equilibrium towards the desired product without the need for excessive thermal input.
From an impurity control perspective, the mechanism offers distinct advantages crucial for regulatory compliance in pharmaceutical manufacturing. The primary impurity concern in salicyloyl chloride synthesis is the formation of the di-chlorinated species where the phenolic oxygen is also substituted. The mild thermal window of 40-65°C enforced by the graphene oxide catalysis is thermodynamically insufficient to activate the phenolic hydroxyl for chlorination under the optimized stoichiometric conditions (molar ratio of acid to thionyl chloride of 1:1.5 to 1:5). 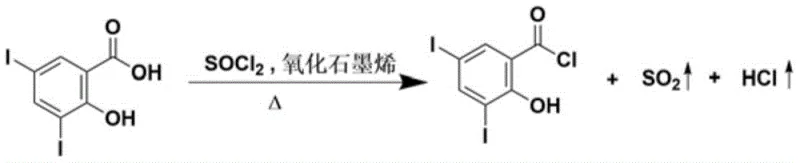 Furthermore, the rapid reaction kinetics minimize the residence time of the sensitive acyl chloride product in the reactive medium, reducing the opportunity for degradation or hydrolysis upon trace moisture exposure. This mechanistic precision ensures that the final crude product is of such high quality that extensive purification steps, such as recrystallization or column chromatography, may be rendered unnecessary, directly impacting the cost of goods sold and manufacturing throughput.
Furthermore, the rapid reaction kinetics minimize the residence time of the sensitive acyl chloride product in the reactive medium, reducing the opportunity for degradation or hydrolysis upon trace moisture exposure. This mechanistic precision ensures that the final crude product is of such high quality that extensive purification steps, such as recrystallization or column chromatography, may be rendered unnecessary, directly impacting the cost of goods sold and manufacturing throughput.
How to Synthesize 3,5-Diiodo Salicyloyl Chloride Efficiently
The implementation of this graphene oxide-catalyzed protocol requires careful attention to reagent ratios and solvent selection to maximize the benefits observed in the patent examples. The process begins with the suspension of the catalyst in a suitable organic solvent, followed by the addition of the 3,5-diiodosalicylic acid and thionyl chloride. The choice of solvent is flexible, ranging from pure dichloromethane to mixtures with n-hexane, allowing process engineers to tune the solubility and boiling point characteristics to their specific reactor configurations. Heating is applied gently to initiate the reflux, and the reaction progress is visually monitored by the clarification of the suspension, which serves as a reliable endpoint indicator before the fixed reaction time is completed. Detailed standard operating procedures regarding exact mass ratios, stirring speeds, and vacuum parameters for solvent removal are critical for reproducibility at scale.
- Charge 3,5-diiodosalicylic acid, graphene oxide catalyst, and organic solvent (e.g., dichloromethane) into a reaction vessel.
- Add thionyl chloride and heat the mixture to 40-65°C under reflux conditions with stirring.
- Maintain reaction for 1-3 hours after clarification, then remove solvent under reduced pressure to isolate the product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this synthetic route translates into tangible strategic advantages beyond mere chemical yield. The shift from high-boiling chlorobenzene to volatile solvents like dichloromethane fundamentally alters the energy profile of the manufacturing process. The elimination of high-temperature distillation steps results in significantly reduced utility consumption, directly lowering the variable cost per kilogram of the intermediate. Moreover, the simplified post-treatment, which involves straightforward solvent evaporation rather than complex purification sequences, shortens the overall batch cycle time. This efficiency gain enhances asset utilization, allowing manufacturing facilities to produce more batches within the same timeframe, thereby improving supply responsiveness to market demands. The robustness of the method also implies a more stable supply chain, as the reliance on specialized high-temperature resistant equipment is diminished, reducing the risk of production stoppages due to equipment failure.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the drastic simplification of the downstream processing workflow. By avoiding the use of chlorobenzene, manufacturers eliminate the need for energy-intensive stripping operations required to remove high-boiling solvents, which traditionally account for a significant portion of production costs. Additionally, the near-quantitative yield of 99.6% means that raw material waste is virtually eliminated, maximizing the value extracted from every kilogram of 3,5-diiodosalicylic acid purchased. The reduction in impurity formation also lowers the costs associated with waste disposal and quality control testing, as fewer batches are likely to fail specification limits. Collectively, these factors contribute to a leaner, more cost-effective manufacturing model that improves margin potential for the final API or intermediate.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by the complexity of chemical synthesis, particularly when harsh conditions lead to unpredictable equipment corrosion or maintenance issues. The mild operating conditions of 40-65°C significantly reduce the corrosive stress on reactor vessels and piping, extending the lifespan of capital assets and minimizing unplanned downtime. Furthermore, the use of common, commercially available solvents like dichloromethane and hexane ensures that raw material sourcing remains stable and unaffected by the supply volatility often seen with specialized high-boiling solvents. The high purity of the output (up to 99.9%) reduces the likelihood of downstream rejection by customers, fostering stronger long-term partnerships and reducing the administrative burden of managing returns or complaints. This reliability makes the supplier a more attractive partner for long-term contracts and Just-In-Time delivery models.
- Scalability and Environmental Compliance: Scaling chemical processes from the laboratory to multi-ton production is fraught with challenges, particularly regarding heat transfer and safety. The exothermic nature of acyl chlorination is much easier to manage at lower temperatures, reducing the risk of thermal runaway incidents during scale-up. The graphene oxide catalyst, being a solid, can potentially be recovered and reused, aligning with green chemistry principles and reducing the generation of hazardous waste streams. The lower energy footprint of the process also supports corporate sustainability goals by reducing the carbon emissions associated with heating and solvent recovery. This environmental compatibility simplifies regulatory compliance and permitting, facilitating faster approval for capacity expansions and ensuring the long-term viability of the production site in an increasingly regulated global market.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of 3,5-diiodo salicyloyl chloride using this advanced catalytic method. These insights are derived directly from the experimental data and technical disclosures within the patent literature, providing a transparent view of the process capabilities. Understanding these details is essential for technical teams evaluating the feasibility of integrating this intermediate into their own synthesis pipelines or for procurement specialists assessing supplier qualifications.
Q: Why is graphene oxide used as a catalyst in this synthesis?
A: Graphene oxide introduces abundant oxygen-containing functional groups that increase reaction active sites, allowing for rapid initiation and milder reaction conditions compared to traditional thermal methods.
Q: How does this method prevent side reactions on the phenolic hydroxyl group?
A: By optimizing the reaction temperature to a mild range of 40-65°C and controlling reactant ratios, the process selectively targets the carboxyl group while preserving the sensitive phenolic hydroxyl moiety.
Q: What are the purity specifications achievable with this new route?
A: The patented process demonstrates the capability to achieve product purity levels up to 99.9%, significantly surpassing the typical 98% purity found in commercially available alternatives.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3,5-Diiodo Salicyloyl Chloride Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from patent literature to commercial reality requires deep technical expertise and robust infrastructure. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of the graphene oxide-catalyzed route are fully realized in large-scale manufacturing. We maintain stringent purity specifications through our rigorous QC labs, employing advanced analytical techniques to verify that every batch of 3,5-diiodo salicyloyl chloride meets the exacting standards required for pharmaceutical applications. Our commitment to quality assurance means that we can consistently deliver the high-purity material necessary to support your drug development timelines without compromise.
We invite you to collaborate with us to leverage this cutting-edge synthesis technology for your supply chain. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements and logistical needs. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions based on empirical evidence. Let us demonstrate how our advanced manufacturing capabilities can enhance the efficiency and reliability of your pharmaceutical intermediate supply.
