Advanced Urea-Based Synthesis of Carisoprodol: Technical Insights for Commercial Scale-Up
The pharmaceutical industry continuously seeks robust synthetic pathways that balance economic efficiency with rigorous safety standards, particularly for widely prescribed skeletal muscle relaxants like Carisoprodol. Patent CN101289441A introduces a transformative methodology that redefines the production landscape for this critical active pharmaceutical ingredient and its key intermediate, 5-methyl-5-propyl-1,3-dioxane-2-one. By shifting away from traditional, hazardous reagents such as phosgene or expensive carbonates, this invention leverages urea as a primary building block in a condensation reaction catalyzed by activated metal oxides. This strategic pivot not only mitigates the severe environmental and safety liabilities associated with phosgene handling but also streamlines the purification process by generating ammonia gas as a benign, easily separable byproduct. For global supply chain leaders, this represents a pivotal opportunity to secure a more sustainable and cost-effective source of high-purity pharmaceutical intermediates without compromising on the structural integrity or therapeutic efficacy of the final drug product.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Carisoprodol has been plagued by significant operational hurdles and safety concerns inherent to legacy manufacturing routes. The traditional phosgene pathway, while chemically direct, imposes extreme labor protection requirements due to the acute toxicity of phosgene gas, creating substantial regulatory burdens and potential liability risks for manufacturing facilities. Alternatively, the carbonate route utilizing diethyl carbonate presents distinct thermodynamic challenges; the condensation reaction generates ethanol as a byproduct, which forms a difficult-to-separate azeotrope with common solvents like toluene. This necessitates complex, energy-intensive distillation processes to drive the equilibrium forward, resulting in higher utility costs and increased consumption of raw materials due to co-distillation losses. Furthermore, the reliance on metal alkoxides in these older methods often requires careful neutralization steps to prevent product degradation during aqueous workups, adding layers of complexity that hinder scalability and reduce overall process reliability for large-scale commercial operations.
The Novel Approach
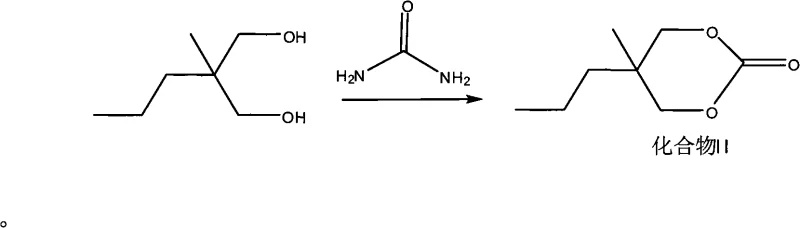
The innovative strategy outlined in the patent data fundamentally resolves these bottlenecks by employing urea, a commodity chemical with superior availability and cost profiles, as the carbonyl source. In this streamlined process, 2-methyl-2-propyl-1,3-propanediol undergoes condensation with urea under the influence of activated metal oxides, yielding the cyclic carbonate intermediate with remarkable efficiency. A critical advantage of this chemistry is the generation of ammonia gas as the sole byproduct, which naturally evolves from the reaction mixture, thereby eliminating the need for complex azeotropic distillation equipment and significantly reducing energy consumption. This simplification allows for a more direct transition to downstream processing, where the intermediate is subsequently ammonolyzed with isopropylamine to form the carbamate precursor.
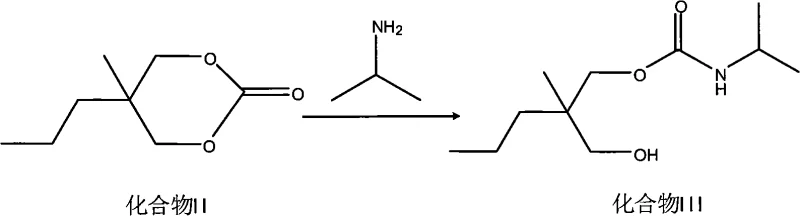
Following this, the precursor reacts with sodium cyanate to finalize the Carisoprodol structure, ensuring a continuous and logically sequenced production flow that minimizes unit operations and maximizes throughput potential for industrial applications.
Mechanistic Insights into Metal Oxide-Catalyzed Urea Condensation
The core of this technological breakthrough lies in the heterogeneous catalysis provided by activated metal oxides, which facilitate the nucleophilic attack of the diol hydroxyl groups onto the urea carbonyl carbon. The patent specifies that metal oxides such as Zinc Oxide, Magnesium Oxide, or Aluminum Oxide, when calcined at high temperatures around 450°C, exhibit enhanced surface activity that promotes the cyclization reaction effectively. This catalytic system operates optimally at temperatures exceeding 90°C, approaching the decomposition threshold of urea, yet the specific activation of the catalyst ensures that the reaction proceeds selectively towards the desired cyclic carbonate rather than degrading into biuret or other urea decomposition products. The mechanism likely involves the coordination of the diol oxygen atoms to the metal centers, increasing their nucleophilicity and stabilizing the transition state during the elimination of ammonia. This precise control over reaction kinetics is essential for maintaining high selectivity and preventing the formation of polymeric side products that could complicate downstream purification efforts.
Furthermore, the process demonstrates exceptional robustness regarding stoichiometry; while a 1:1 molar ratio is theoretically sufficient, employing an excess of the diol (greater than 2:1 ratio) drives the equilibrium towards completion and allows for the unreacted diol to be recovered and recycled. This recycling capability is a crucial mechanistic feature that enhances atom economy and reduces waste generation, aligning the process with green chemistry principles. The catalyst itself shows remarkable stability, with data indicating no significant loss in yield after ten consecutive reuse cycles, suggesting that the active sites remain intact and resistant to poisoning or leaching under the reaction conditions. This durability is vital for maintaining consistent batch-to-batch quality in a commercial manufacturing setting.
How to Synthesize 5-methyl-5-propyl-1,3-dioxane-2-one Efficiently
To implement this advanced synthesis route effectively, manufacturers must adhere to specific operational parameters regarding temperature control and catalyst preparation to ensure optimal yields and purity profiles. The process begins with the activation of the metal oxide catalyst, followed by the condensation reaction which can be run either solvent-free or in aromatic solvents depending on the desired thermal profile. Detailed standard operating procedures for catalyst activation, reaction monitoring via TLC, and the subsequent aminolysis and cyanation steps are critical for successful technology transfer.
- Condense 2-methyl-2-propyl-1,3-propanediol with urea using an activated metal oxide catalyst to form the cyclic carbonate intermediate.
- Perform aminolysis on the cyclic carbonate intermediate using isopropylamine in a toluene solvent to generate the carbamate precursor.
- React the carbamate precursor with sodium cyanate and hydrochloric acid to finalize the synthesis of Carisoprodol.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this urea-based synthesis route offers compelling strategic advantages that directly impact the bottom line and operational resilience. By replacing expensive and logistically complex reagents like diethyl carbonate or hazardous phosgene with readily available urea, the raw material cost structure is significantly optimized, providing a buffer against market volatility in specialty chemical pricing. The elimination of energy-intensive azeotropic distillation steps translates to drastic reductions in utility consumption, specifically steam and cooling water, which are major cost drivers in large-scale chemical manufacturing. Additionally, the simplified workflow reduces the total cycle time per batch, allowing facilities to increase throughput capacity without requiring capital expenditure on new reactor trains or separation columns.
- Cost Reduction in Manufacturing: The substitution of high-cost carbonates with low-cost urea fundamentally alters the variable cost equation, delivering substantial savings on raw material procurement. Moreover, the ability to recycle excess diol and reuse the heterogeneous catalyst over multiple batches minimizes waste disposal costs and maximizes the value extracted from every kilogram of input material. The removal of complex distillation sequences further lowers operational expenditures by reducing energy demand and maintenance requirements associated with high-efficiency fractionation columns.
- Enhanced Supply Chain Reliability: Sourcing urea is inherently more stable and less prone to geopolitical disruptions compared to specialized reagents like triphosgene or specific metal alkoxides, ensuring a consistent supply of critical inputs. The robustness of the catalyst system, which tolerates multiple reuse cycles without performance degradation, reduces the frequency of catalyst replenishment orders and mitigates the risk of production stoppages due to catalyst supply shortages. This reliability is essential for maintaining uninterrupted production schedules and meeting delivery commitments to downstream pharmaceutical customers.
- Scalability and Environmental Compliance: The process generates ammonia gas as a primary byproduct, which can be easily captured and neutralized or utilized, avoiding the generation of liquid waste streams laden with organic solvents or salts that require costly treatment. The absence of heavy metals or highly toxic reagents simplifies regulatory compliance and reduces the burden on environmental health and safety teams, facilitating smoother audits and permitting processes. This environmental profile supports sustainable manufacturing goals and enhances the corporate reputation of companies adopting this greener synthetic pathway.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this urea-based synthesis technology for Carisoprodol production. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a factual basis for decision-making.
Q: What are the primary advantages of using urea over diethyl carbonate in Carisoprodol synthesis?
A: Using urea significantly reduces raw material costs compared to diethyl carbonate. Furthermore, the byproduct is ammonia gas, which escapes the reaction system naturally, eliminating the need for energy-intensive azeotropic distillation required to remove ethanol in traditional carbonate routes.
Q: Can the metal oxide catalyst be reused in this process?
A: Yes, the activated metal oxide catalysts, such as Zinc Oxide, can be recycled multiple times. Patent data indicates that the catalyst maintains its activity and yield efficiency even after ten cycles of use, contributing to long-term process sustainability.
Q: How does this method improve safety compared to phosgene-based routes?
A: This method completely avoids the use of phosgene or triphosgene, which are highly toxic and require stringent safety protocols. By utilizing urea and sodium cyanate under controlled conditions, the process drastically lowers occupational health risks and environmental hazards.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Carisoprodol Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting efficient and safe synthetic routes to meet the growing global demand for muscle relaxant therapies. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless and robust. We are committed to delivering high-purity Carisoprodol and its intermediates that meet stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical capabilities. By leveraging advanced catalytic technologies similar to those described in recent patents, we optimize our manufacturing processes to deliver superior value to our partners.
We invite pharmaceutical companies and contract manufacturers to collaborate with us to explore how this optimized synthesis route can enhance your supply chain efficiency. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our manufacturing excellence can support your long-term strategic goals in the competitive pharmaceutical marketplace.
