Advanced Z-M-OR Catalytic Systems for Sustainable Carboxylation of Pharmaceutical Intermediates
Introduction to Next-Generation Carboxylation Technology
The chemical industry is currently undergoing a paradigm shift towards sustainable synthesis, driven by the urgent need to utilize renewable carbon feedstocks and minimize toxic waste. Patent CN102947000A introduces a groundbreaking methodology utilizing Z-M-OR form complexes, where M represents a transition metal such as gold, silver, or copper, and Z acts as a robust two-electron donor ligand. This technology specifically targets the direct carboxylation of substrates at C-H or N-H bonds using carbon dioxide, a plentiful and non-toxic C1 source. Unlike traditional methods that rely on hazardous carbon monoxide or stoichiometric organometallic reagents, this catalytic system operates under remarkably mild conditions, typically between 0°C and 100°C with CO2 pressures ranging from 1 to 10 bar. For R&D directors and process chemists, this represents a significant leap forward in atom economy and operational safety, providing a reliable pathway to synthesize high-purity pharmaceutical intermediates without the logistical burdens associated with handling toxic gases.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the introduction of carboxyl groups into organic frameworks has been plagued by severe limitations regarding safety, selectivity, and substrate scope. Traditional palladium-catalyzed carbonylation processes, while effective, frequently necessitate the use of gaseous carbon monoxide, a highly toxic and flammable reagent that requires specialized infrastructure and rigorous safety protocols to manage. Furthermore, these legacy methods often demand forcing temperatures and high catalyst loadings, which can lead to thermal degradation of sensitive functional groups commonly found in advanced drug candidates. Another critical drawback is the lack of regioselectivity; conventional electrophilic aromatic substitution or metal-mediated activations often yield complex mixtures of regioisomers, necessitating costly and time-consuming purification steps that erode overall process efficiency. Additionally, many existing protocols are restricted to electron-rich arenes, failing to activate the electron-deficient heterocycles that are ubiquitous in modern medicinal chemistry.
The Novel Approach
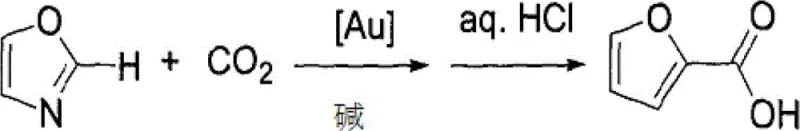
The innovative approach detailed in the patent data circumvents these challenges by employing well-defined Z-M-OR complexes, such as N-heterocyclic carbene (NHC) gold hydroxides, to activate the most acidic C-H or N-H bonds directly. This method leverages the inherent acidity of the substrate rather than relying on pre-functionalized halides or directing groups, thereby streamlining the synthetic route. By utilizing carbon dioxide as the sole carbon source, the process eliminates the need for toxic CO gas entirely, aligning with green chemistry principles. The system demonstrates exceptional tolerance for various functional groups and achieves high regioselectivity, preferentially carboxylating the position with the lowest pKa value. As illustrated in the reaction scheme below, oxazoles can be converted to their corresponding 2-carboxylic acids in quantitative yields under mild conditions, showcasing the practical utility of this technology for generating valuable building blocks.
Mechanistic Insights into NHC-Metal Catalyzed C-H Activation
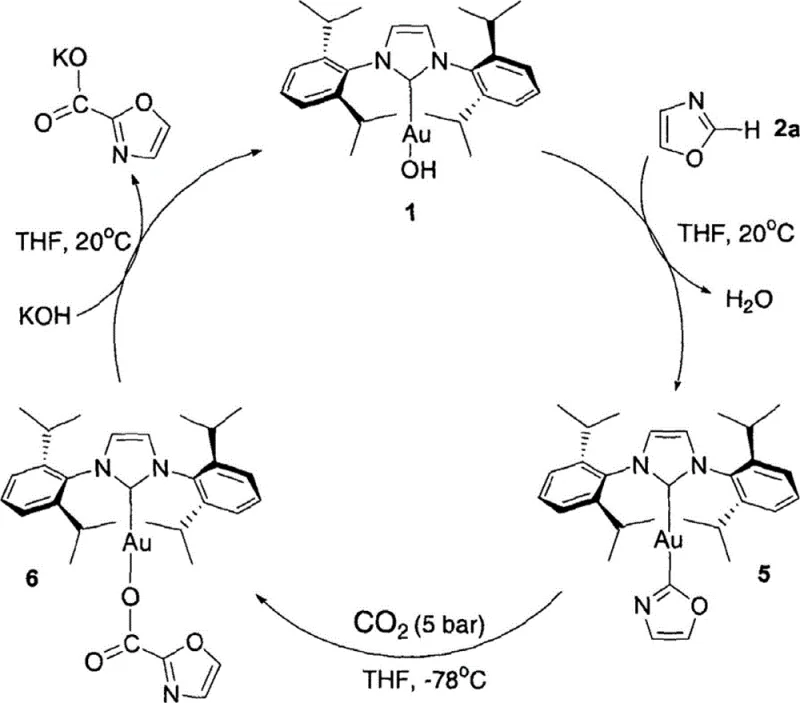
The efficacy of the Z-M-OR catalytic system lies in its unique mechanism of action, which combines Lewis basicity with transition metal coordination to facilitate CO2 insertion. The catalytic cycle initiates with an acid-base exchange where the metal-hydroxide or metal-alkoxide bond (M-OR) deprotonates the substrate at its most acidic site, generating a metal-substrate intermediate and releasing water or alcohol. This step is crucial as it bypasses the high energy barriers associated with oxidative addition typically seen in palladium chemistry. Once the metal-carbon or metal-nitrogen bond is formed, the coordinated metal center activates the incoming carbon dioxide molecule, allowing for facile insertion into the metal-substrate bond to form a metal-carboxylate species. The cycle is completed when an external base, such as potassium hydroxide, displaces the carboxylate product, regenerating the active Z-M-OH catalyst species. This turnover mechanism allows for low catalyst loadings, often as low as 1.5 mol%, while maintaining high turnover frequencies.
Regioselectivity in this system is governed principally by the pKa of the available C-H or N-H bonds relative to the basicity of the catalyst. For instance, gold hydroxide complexes like [(IPr)AuOH] possess a specific pKa window that enables them to selectively deprotonate oxazoles at the C2 position rather than the C3 or C5 positions, a selectivity profile that is difficult to achieve with traditional lithiation or Grignard reagents. The catalytic cycle diagram below elucidates this reversible process, highlighting how the interplay between the NHC ligand sterics and the metal center electronics fine-tunes the reactivity. This precise control over the reaction trajectory ensures that complex heterocyclic scaffolds are functionalized cleanly, minimizing the formation of byproducts and simplifying downstream purification for supply chain teams.
How to Synthesize Carboxylic Acid Derivatives Efficiently
The implementation of this carboxylation technology in a laboratory or pilot plant setting follows a straightforward protocol that emphasizes safety and reproducibility. The process begins with the preparation or procurement of the specific Z-M-OR catalyst, such as [(IPr)AuOH] or [(IPr)CuOH], which can be generated in situ from the corresponding chloride precursors using simple bases like CsOH or KOH. The substrate is then dissolved in a polar aprotic solvent like THF, and the reaction vessel is pressurized with carbon dioxide to the specified range of 1 to 10 bar. Maintaining the temperature within the optimal window of 20°C to 65°C is critical to balancing reaction kinetics with catalyst stability. Detailed standardized synthesis steps for specific substrates, including precise stoichiometric ratios and workup procedures, are provided in the guide below.
- Prepare the Z-M-OR catalyst complex, such as [(IPr)AuOH], by reacting the corresponding metal halide with a strong base like CsOH or KOH in a suitable solvent.
- Mix the substrate containing acidic C-H or N-H bonds with the catalyst and an external base in a solvent like THF under an inert atmosphere.
- Introduce carbon dioxide gas (1-10 bar) to the reaction mixture at moderate temperatures (20-65°C) and stir until conversion is complete, followed by acid workup.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of Z-M-OR catalyzed carboxylation offers transformative benefits that extend beyond mere technical feasibility. The primary advantage lies in the drastic simplification of raw material logistics; by replacing toxic carbon monoxide cylinders with benign carbon dioxide, facilities can significantly reduce regulatory compliance costs and insurance premiums associated with hazardous gas storage. Furthermore, the ability to use CO2, which can be sourced as a byproduct from other industrial processes, opens avenues for substantial cost reduction in pharmaceutical intermediate manufacturing by utilizing a low-cost, abundant feedstock. The mild reaction conditions also translate to lower energy consumption, as there is no need for energy-intensive heating or cryogenic cooling, contributing to a smaller carbon footprint and alignment with corporate sustainability goals.
- Cost Reduction in Manufacturing: The elimination of expensive and toxic reagents like carbon monoxide and organolithiums removes the need for specialized containment equipment and extensive waste treatment protocols. This streamlined approach reduces the overall cost of goods sold (COGS) by minimizing capital expenditure on safety infrastructure and lowering operational expenses related to hazardous waste disposal. Additionally, the high atom economy of using CO2 ensures that nearly all carbon input is incorporated into the final product, maximizing material efficiency.
- Enhanced Supply Chain Reliability: Relying on carbon dioxide as a feedstock mitigates supply risks associated with volatile petrochemical markets. CO2 is widely available and can often be sourced locally, reducing dependency on long-distance transportation of hazardous chemicals. The robustness of the catalyst system, which tolerates a wide range of functional groups without the need for protecting group strategies, further accelerates production timelines and ensures consistent delivery schedules for critical API intermediates.
- Scalability and Environmental Compliance: The process operates at near-ambient pressures and temperatures, making it inherently safer and easier to scale from gram-scale R&D to multi-ton commercial production. The absence of heavy metal waste streams typical of stoichiometric organometallic reactions simplifies effluent treatment, ensuring compliance with increasingly stringent environmental regulations. This scalability ensures that the technology can meet growing market demand without compromising on quality or safety standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this carboxylation technology. These insights are derived directly from the experimental data and mechanistic studies presented in the patent literature, providing clarity on substrate compatibility and process parameters. Understanding these nuances is essential for project managers evaluating the feasibility of integrating this method into existing production lines.
Q: What are the primary advantages of using Z-M-OR complexes over traditional palladium catalysts?
A: Z-M-OR complexes, particularly those based on gold and copper with NHC ligands, operate under significantly milder conditions and utilize non-toxic CO2 instead of hazardous carbon monoxide gas, offering superior safety and regioselectivity.
Q: Can this carboxylation method be applied to electron-deficient aromatic substrates?
A: Yes, the method is highly effective for polyfluorinated arenes and other electron-deficient systems, achieving high yields where traditional Friedel-Crafts type carboxylations often fail due to deactivation.
Q: Is the catalyst system scalable for industrial production of API intermediates?
A: The process utilizes low catalyst loadings (as low as 1.5 mol%) and moderate pressures, making it highly amenable to scale-up for the commercial manufacturing of complex pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pharmaceutical Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of Z-M-OR catalyzed carboxylation in the synthesis of complex pharmaceutical intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory discoveries are seamlessly translated into robust manufacturing processes. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of carboxylated heterocycles or arenes delivered meets the highest industry standards for potency and impurity profiles.
We invite you to collaborate with our technical team to explore how this sustainable carboxylation technology can optimize your specific synthesis routes. By leveraging our expertise in transition metal catalysis and process development, we can provide a Customized Cost-Saving Analysis tailored to your project needs. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, ensuring a secure and efficient supply chain for your next-generation therapeutic candidates.
