Advanced Blue LED Catalysis for Scalable 3-Thio-Quinoxalinone Pharmaceutical Intermediates
Advanced Blue LED Catalysis for Scalable 3-Thio-Quinoxalinone Pharmaceutical Intermediates
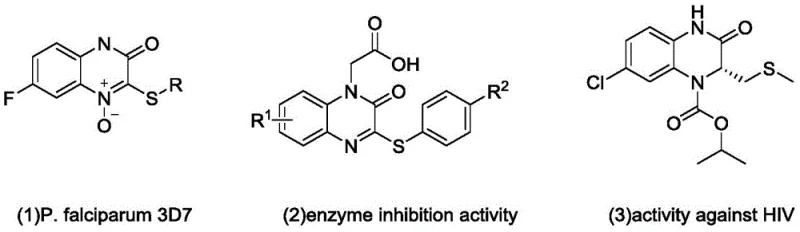
The pharmaceutical industry continuously seeks robust synthetic routes for heterocyclic scaffolds that possess significant biological activity. Patent CN113603651B introduces a groundbreaking catalytic method for preparing 3-sulfur-substituted quinoxalinone derivatives, a class of compounds critical for developing antimalarial, enzyme-inhibiting, and anti-HIV agents. This technology leverages visible light photocatalysis to overcome the severe limitations of traditional thiol-based chemistry, offering a greener and more efficient pathway for producing high-value pharmaceutical intermediates. By utilizing stable disulfides instead of volatile mercaptans, this process addresses major safety and environmental concerns while maintaining high reaction efficiency. For R&D directors and procurement specialists, this represents a significant opportunity to optimize the supply chain for complex nitrogen-containing heterocycles.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of carbon-sulfur bonds at the C3 position of quinoxalinone scaffolds has relied heavily on the use of thiophenols or mercaptans. These reagents are notoriously difficult to manage in an industrial setting due to their extremely foul odor and high toxicity, posing significant health risks to operators and requiring specialized ventilation infrastructure. Furthermore, conventional methods often involve electrocatalytic or harsh photocatalytic conditions that may require specific solvents like N-methylpyrrolidone or dimethylformamide under rigorous control. The reliance on liquid thiols also introduces stability issues during storage and transportation, complicating logistics for global supply chains. These factors collectively increase the cost of goods sold and limit the scalability of processes intended for commercial API manufacturing.
The Novel Approach
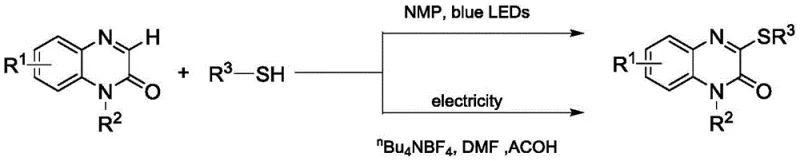
The innovative methodology described in the patent fundamentally shifts the paradigm by employing diaryl disulfides as the sulfur source instead of thiols. This substitution eliminates the offensive odor and toxicity associated with mercaptans, creating a much safer working environment. The reaction is driven by a dual catalytic system comprising cerium chloride and tetrabutylammonium chloride under 465nm blue LED irradiation. This mild condition allows the reaction to proceed efficiently at room temperature without the need for external heating or high-pressure equipment. The use of acetonitrile as a solvent and potassium persulfate as an oxidant further streamlines the workup procedure. This approach not only enhances the sustainability profile of the synthesis but also broadens the substrate scope to include various substituted quinoxalinones, making it a versatile tool for medicinal chemists.
Mechanistic Insights into CeCl3-Catalyzed Photocyclization
The core of this transformation lies in the synergistic interaction between the cerium catalyst and the visible light source. Under 465nm blue LED irradiation, the cerium species likely facilitates the generation of sulfur-centered radicals from the disulfide bond. These radicals then attack the electron-deficient C3 position of the quinoxalinone ring, forming the new carbon-sulfur bond. The presence of tetrabutylammonium chloride acts as a phase-transfer catalyst or stabilizer, enhancing the solubility and reactivity of the ionic species involved. Potassium persulfate serves as a terminal oxidant to regenerate the active catalytic species and drive the reaction to completion. This mechanistic pathway avoids the formation of heavy metal waste often associated with transition metal-catalyzed cross-couplings, aligning with green chemistry principles.
From an impurity control perspective, the high selectivity of this radical mechanism ensures minimal formation of side products. The reaction conditions are mild enough to preserve sensitive functional groups on the aromatic rings, such as methyl or chloro substituents, which might be compromised under harsher thermal conditions. The patent data indicates that the crude products can be purified to greater than 99% purity using standard silica gel column chromatography. This high level of purity is crucial for pharmaceutical intermediates, as it reduces the burden on downstream purification steps and ensures consistent quality for subsequent drug synthesis. The robustness of the catalyst system also suggests low sensitivity to moisture or oxygen, further simplifying process control.
How to Synthesize 3-Thio-Substituted Quinoxalinone Derivatives Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for laboratory and pilot-scale production. It begins with the preparation of the N-alkylated quinoxalinone precursor, followed by the key C-S coupling step. The detailed standardized synthesis steps below describe the precise molar ratios and reaction times required to achieve optimal yields. Adhering to these parameters ensures reproducibility and maximizes the efficiency of the blue LED photocatalytic system.
- Prepare the quinoxalinone precursor (Compound II) via alkylation of quinoxalin-2(1H)-one with appropriate alkyl halides in DMF.
- Combine the precursor, disulfide compound, cerium chloride catalyst, tetrabutylammonium chloride, and potassium persulfate oxidant in acetonitrile.
- Irradiate the reaction mixture with 465nm blue LEDs for 36 hours at room temperature, followed by filtration and column chromatography purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this disulfide-based photocatalytic route offers tangible strategic benefits beyond mere chemical novelty. The shift from liquid thiols to solid disulfides drastically simplifies raw material handling and storage logistics. Solid reagents are generally more stable, have longer shelf lives, and do not require the specialized containment systems needed for volatile, odorous liquids. This translates directly into reduced operational overhead and lower risk of regulatory non-compliance regarding emissions. Additionally, the elimination of high-temperature requirements reduces energy consumption, contributing to lower utility costs and a smaller carbon footprint for the manufacturing facility.
- Cost Reduction in Manufacturing: The removal of expensive and hazardous thiol reagents significantly lowers the raw material cost base. Furthermore, the simplified workup procedure, which involves basic filtration and extraction rather than complex distillation or scrubbing processes, reduces labor and solvent consumption. The use of earth-abundant cerium salts instead of precious metal catalysts like palladium or rhodium further drives down the cost of catalyst loading. These cumulative efficiencies result in substantial cost savings in pharmaceutical intermediate manufacturing without compromising on product quality or yield.
- Enhanced Supply Chain Reliability: Sourcing stable solid disulfides is generally more reliable than managing the supply of volatile thiols, which can degrade or evaporate during transit. The robust nature of the reaction conditions means that production is less susceptible to minor fluctuations in environmental parameters, ensuring consistent batch-to-batch quality. This reliability is critical for maintaining continuous supply to downstream API manufacturers, reducing the risk of stockouts or production delays. The ability to source generic reagents like potassium persulfate and cerium chloride from multiple vendors also mitigates single-source supply risks.
- Scalability and Environmental Compliance: The photochemical nature of the reaction is highly amenable to scale-up using modern flow chemistry reactors or large-scale LED arrays. Operating at room temperature eliminates the safety hazards associated with exothermic runaway reactions common in thermal processes. Moreover, the absence of toxic thiol emissions ensures that the facility remains well within environmental protection agency limits for volatile organic compounds and noxious odors. This compliance ease accelerates the permitting process for new production lines and fosters a sustainable corporate image.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthetic route. These answers are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a factual basis for decision-making.
Q: What are the advantages of using disulfides over thiols in this synthesis?
A: Disulfides are odorless solids that are significantly safer and easier to handle than foul-smelling, toxic liquid thiols, greatly improving workplace safety and environmental compliance.
Q: What is the typical yield and purity of the 3-thio-substituted quinoxalinone products?
A: The patented method achieves yields up to 87% with product purity exceeding 99% after standard column chromatography purification, ensuring high quality for pharmaceutical applications.
Q: Is this photocatalytic process scalable for industrial production?
A: Yes, the reaction operates at room temperature using visible light and stable solid reagents, eliminating the need for high-pressure or high-temperature equipment, which facilitates easy scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Thio-Substituted Quinoxalinone Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this blue LED catalyzed synthesis for the production of high-value quinoxalinone intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped with advanced photoreactors capable of handling visible light chemistry efficiently, ensuring that the benefits of this patent can be realized on an industrial scale. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch meets the exacting standards required by global pharmaceutical clients.
We invite you to collaborate with us to optimize your supply chain for these critical building blocks. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us help you leverage this green chemistry innovation to enhance your product portfolio and reduce time-to-market for your next-generation therapeutics.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
