Scalable Synthesis of Beta-p-Hydroxyphenethyl Alcohol via Phenol Protection Strategy
Scalable Synthesis of Beta-p-Hydroxyphenethyl Alcohol via Phenol Protection Strategy
The global demand for high-purity pharmaceutical intermediates continues to surge, driven by the expanding applications of antioxidant compounds in both therapeutic and nutraceutical sectors. Patent CN100432035C introduces a robust and economically viable technical method for synthesizing beta-p-hydroxyphenethyl alcohol, also known as Tyrosol, a molecule of significant interest due to its potent inhibitory effects on active oxides and its role as a precursor for cardiovascular drugs like Metoprolol. Unlike traditional extraction methods from natural sources which suffer from low yields and complex fermentation timelines, this patented chemical synthesis offers a streamlined three-step pathway starting from ubiquitous raw materials. The innovation lies in a strategic protection-deprotection sequence that circumvents the inherent reactivity issues of phenol, ensuring high regioselectivity and operational safety. For R&D directors and procurement specialists seeking a reliable pharmaceutical intermediate supplier, understanding the mechanistic elegance and commercial scalability of this route is paramount for securing long-term supply chain resilience.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of beta-p-hydroxyphenethyl alcohol has been plagued by significant technical and economic hurdles that render many established routes unsuitable for modern industrial application. Early approaches, such as those described in European patent EP0449603, relied on 4-acetoxystyrene as a starting material; however, this precursor is not a common commodity chemical, creating severe bottlenecks in raw material sourcing and driving up costs prohibitively for mass production. Furthermore, alternative pathways utilizing phenol and ethylene oxide, as seen in Japanese patent JP2000327610, often necessitate the use of hazardous organometallic catalysts like butyllithium or triisobutylaluminum. 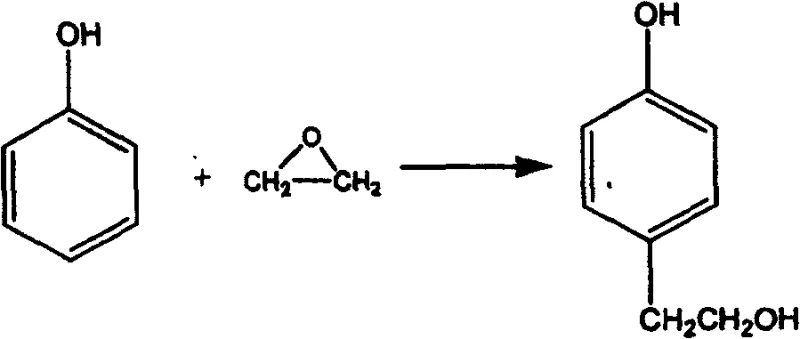 These pyrophoric reagents pose extreme safety risks, requiring stringent inert atmosphere conditions and specialized handling equipment that drastically increase capital expenditure and operational complexity. Additionally, routes employing Grignard reagents or strong reducing agents like lithium aluminum hydride introduce moisture sensitivity and expensive reagent costs, while multi-step sequences involving nitration and diazotization result in poor overall atom economy and excessive waste generation.
These pyrophoric reagents pose extreme safety risks, requiring stringent inert atmosphere conditions and specialized handling equipment that drastically increase capital expenditure and operational complexity. Additionally, routes employing Grignard reagents or strong reducing agents like lithium aluminum hydride introduce moisture sensitivity and expensive reagent costs, while multi-step sequences involving nitration and diazotization result in poor overall atom economy and excessive waste generation.
The Novel Approach
In stark contrast to these hazardous and inefficient legacy methods, the process disclosed in CN100432035C employs a clever three-stage sequence comprising esterification, haloalkylation, and hydrolysis, utilizing phenol and acetic anhydride as the foundational feedstocks. This novel approach effectively mitigates the high electronegativity of the phenolic oxygen atom, which typically leads to undesirable O-alkylation side reactions, by first converting the phenol into phenyl acetate. This protection step directs subsequent alkylation exclusively to the para-position of the aromatic ring when reacted with 1,2-dihaloethane under acidic catalysis. 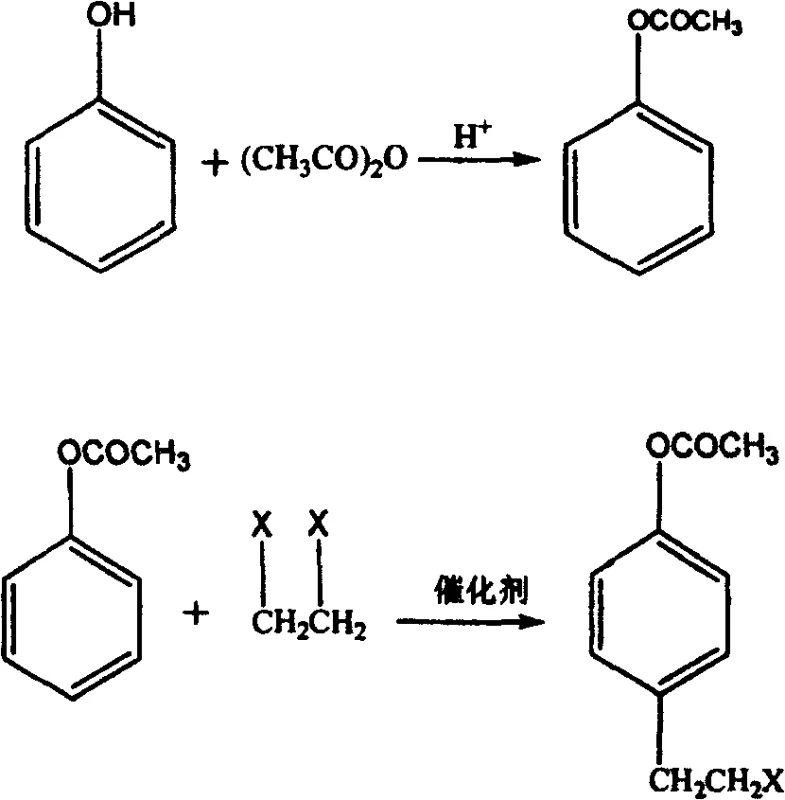 The final hydrolysis step cleanly removes the acetate protecting group to yield the target beta-p-hydroxyphenethyl alcohol with high purity. By replacing exotic catalysts with common mineral acids and utilizing standard polar solvents, this methodology transforms a complex synthetic challenge into a manageable, safe, and cost-effective industrial process that aligns perfectly with the requirements for cost reduction in fine chemical manufacturing.
The final hydrolysis step cleanly removes the acetate protecting group to yield the target beta-p-hydroxyphenethyl alcohol with high purity. By replacing exotic catalysts with common mineral acids and utilizing standard polar solvents, this methodology transforms a complex synthetic challenge into a manageable, safe, and cost-effective industrial process that aligns perfectly with the requirements for cost reduction in fine chemical manufacturing.
Mechanistic Insights into Esterification-Directed Haloalkylation
The core chemical ingenuity of this patent resides in the manipulation of electronic effects on the aromatic ring to control regioselectivity during the carbon-carbon bond formation step. In the initial esterification phase, the reaction between phenol and acetic anhydride converts the strongly activating hydroxyl group into an acetoxy group; while still activating, the acetoxy group moderates the electron density distribution, reducing the nucleophilicity of the oxygen atom itself. This is critical because direct alkylation of free phenol with dihaloethanes often results in ether formation (O-alkylation) rather than the desired ring substitution. By masking the hydroxyl functionality, the synthesis forces the electrophilic attack of the haloethyl carbocation species, generated in situ by the acidic catalyst, to occur at the para-position of the benzene ring, thereby forming the acetic acid-(4-beta-haloethyl) phenyl ester intermediate with high fidelity.
Following the successful construction of the carbon skeleton, the final hydrolysis mechanism serves as a precise deprotection strategy to reveal the bioactive phenolic moiety. The intermediate ester is subjected to nucleophilic attack by hydroxide ions or other basic species in a polar solvent medium, cleaving the ester bond through a standard acyl substitution mechanism. This step is remarkably efficient, operating under mild temperatures ranging from ambient to 120°C, which preserves the integrity of the sensitive beta-hydroxyethyl side chain that might otherwise degrade under harsher acidic or high-thermal conditions found in other synthetic routes. The ability to perform this deprotection using inexpensive inorganic bases like sodium carbonate or potassium carbonate further underscores the practical utility of this mechanism, ensuring that impurity profiles remain clean and that downstream purification processes, such as recrystallization from chloroform, yield needle-like crystals of exceptional quality suitable for pharmaceutical applications.
How to Synthesize Beta-p-Hydroxyphenethyl Alcohol Efficiently
The execution of this synthesis requires careful attention to reaction parameters to maximize yield and minimize byproduct formation, particularly during the haloalkylation stage where temperature control is vital. The process begins with the reflux of phenol and acetic anhydride, followed by a rigorous workup to isolate pure phenyl acetate, which then serves as the substrate for the key carbon-chain extension reaction. Operators must ensure the use of dry polar solvents such as N,N-dimethylformamide or dimethyl sulfoxide to facilitate the solubility of reactants and the stability of the cationic intermediates. Detailed standardized synthesis steps are provided in the guide below to ensure reproducibility and safety compliance.
- Perform esterification of phenol with acetic anhydride at 80-180°C to protect the hydroxyl group, yielding phenyl acetate.
- Conduct haloalkylation of phenyl acetate with 1,2-dihaloethane using an acidic catalyst in a polar solvent to form the intermediate ester.
- Execute hydrolysis of the intermediate using an inorganic base or amine to remove the protecting group and obtain the final product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this patented synthesis route represents a strategic opportunity to optimize sourcing costs and mitigate supply risks associated with volatile specialty chemical markets. The reliance on phenol and acetic anhydride, which are produced on a massive global scale for diverse industries, ensures a stable and continuous supply of raw materials that is immune to the shortages often experienced with niche intermediates like 4-acetoxystyrene or specialized organometallic reagents. Furthermore, the elimination of pyrophoric catalysts and moisture-sensitive reagents significantly lowers the barrier to entry for contract manufacturing organizations, allowing for broader supplier qualification and enhanced negotiation leverage. This shift not only secures the supply chain against disruptions but also aligns with increasingly stringent environmental and safety regulations, reducing the liability and insurance costs associated with handling hazardous materials.
- Cost Reduction in Manufacturing: The economic benefits of this process are derived primarily from the substitution of expensive and dangerous reagents with commodity chemicals that offer superior price stability and availability. By avoiding the use of lithium aluminum hydride or Grignard reagents, manufacturers eliminate the need for costly inert gas blanketing systems and specialized waste treatment protocols required for quenching reactive metals. The high yield of the initial esterification step, reported to reach substantial levels, ensures that raw material utilization is maximized, directly translating to a lower cost of goods sold per kilogram of final product. Additionally, the use of simple acid catalysts and recyclable polar solvents reduces the operational expenditure related to catalyst recovery and solvent disposal, creating a leaner and more profitable production model.
- Enhanced Supply Chain Reliability: Supply chain resilience is significantly bolstered by the simplicity and robustness of the reaction conditions, which allow for production in a wider range of facilities without the need for exotic infrastructure. The tolerance of the process to standard industrial equipment means that lead times for high-purity pharmaceutical intermediates can be reduced, as production slots are more readily available across the global CDMO landscape. The stability of the intermediates also allows for potential batch storage or transport if necessary, providing flexibility in logistics planning that is absent in routes requiring immediate consumption of unstable species. This reliability is crucial for maintaining consistent inventory levels for downstream drug manufacturing, preventing costly production stoppages due to intermediate shortages.
- Scalability and Environmental Compliance: From an environmental perspective, this synthesis route offers a greener alternative to traditional methods by minimizing the generation of heavy metal waste and toxic byproducts associated with organometallic chemistry. The aqueous workup procedures and the ability to use common solvents simplify the effluent treatment process, ensuring compliance with rigorous international environmental standards without imposing excessive burdens on waste management budgets. The short synthetic route, consisting of only three distinct chemical transformations, inherently reduces the cumulative energy consumption and solvent usage per unit of product, supporting corporate sustainability goals. This scalability ensures that the process can be seamlessly expanded from pilot plant quantities to multi-ton commercial production to meet growing market demand without compromising on quality or safety.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology, providing clarity for stakeholders evaluating its adoption. These insights are derived directly from the experimental data and process descriptions within the patent documentation, ensuring accuracy and relevance for decision-makers. Specific answers to these queries can be found in the section below to assist in your technical evaluation.
Q: Why is the esterification step critical in this synthesis route?
A: The esterification step protects the phenolic hydroxyl group, preventing unwanted O-alkylation reactions and ensuring selective C-alkylation at the para-position of the benzene ring.
Q: What are the safety advantages of this method compared to Grignard-based routes?
A: This method avoids the use of pyrophoric organometallic reagents like butyllithium or moisture-sensitive Grignard reagents, significantly reducing operational hazards and simplifying safety protocols.
Q: Can this process be scaled for industrial production?
A: Yes, the process utilizes readily available commodity chemicals like phenol and acetic anhydride and operates under mild conditions, making it highly suitable for large-scale commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Beta-p-Hydroxyphenethyl Alcohol Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to maintain competitiveness in the global pharmaceutical market. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We are committed to delivering high-purity beta-p-hydroxyphenethyl alcohol that meets stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical capabilities. By leveraging the efficiencies of the protection-alkylation-hydrolysis route, we can offer our partners a supply solution that balances cost-effectiveness with uncompromising quality standards.
We invite you to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your specific project requirements. Contact us today to request a Customized Cost-Saving Analysis tailored to your volume needs, and let us provide you with specific COA data and route feasibility assessments. Partnering with us ensures not just a supply of chemicals, but a strategic alliance focused on innovation, reliability, and mutual growth in the dynamic landscape of fine chemical intermediates.
