Scalable Synthesis of Pyrimidine Carboxylic Acids via Boc-Protection Strategy
The pharmaceutical industry continuously demands robust synthetic routes for heterocyclic building blocks, particularly pyrimidine derivatives which serve as critical scaffolds in antiviral and anticancer drug development. Patent CN109467536B introduces a transformative methodology for synthesizing 2-chloro/hydroxypyrimidine-5-carboxylic acid, addressing long-standing inefficiencies in traditional carboxylation techniques. By leveraging a strategic halogen-metal exchange followed by trapping with di-tert-butyl dicarbonate (Boc2O), this innovation bypasses the thermodynamic and kinetic limitations associated with direct carbon dioxide insertion. For R&D directors and process chemists, this represents a significant leap forward in achieving high-purity intermediates with exceptional batch consistency. The protocol not only enhances yield profiles but also simplifies the operational complexity typically required for cryogenic carboxylations, thereby offering a more reliable pharmaceutical intermediate supplier pathway for large-scale manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of pyrimidine-5-carboxylic acids has been plagued by suboptimal yields and rigorous operational constraints that hinder commercial viability. Literature precedents, such as those cited in J. Med. Chem., describe direct carboxylation using butyllithium and carbon dioxide at temperatures as low as -90°C, yet these methods frequently result in yields as poor as 26%. Such low efficiency is unacceptable for industrial applications where material throughput and cost-effectiveness are paramount. Furthermore, the alternative route involving the hydrolysis of ester precursors often requires multi-step sequences starting from aminopyrimidines, involving hazardous diazotization steps with sodium nitrite and zinc chloride. These traditional pathways introduce significant safety risks, generate substantial heavy metal waste, and suffer from poor atom economy. The reliance on extreme cryogenic conditions also imposes a heavy burden on energy consumption and equipment maintenance, creating bottlenecks in the supply chain for high-purity pharmaceutical intermediates.
The Novel Approach
The innovative strategy disclosed in the patent fundamentally reimagines the construction of the carboxylic acid moiety by utilizing Boc2O as a stable electrophilic source of the carboxyl group. Instead of struggling with the gaseous and difficult-to-handle CO2, the process employs a controlled metalation of 2-chloro-5-bromopyrimidine followed by immediate trapping with the anhydride. This approach allows the reaction to proceed with much greater tolerance to temperature variations, typically ranging from -20°C to room temperature depending on the specific metal reagent chosen. The resulting tert-butyl ester intermediate is stable and easily isolable, providing a crucial checkpoint for quality control before the final deprotection step. This modular design enables the production of both 2-chloro and 2-hydroxy derivatives from a common intermediate simply by altering the hydrolysis conditions, thereby streamlining the manufacturing of complex pharmaceutical intermediates and reducing the need for separate synthetic lines.
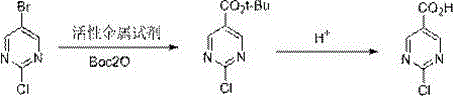
Mechanistic Insights into Halogen-Metal Exchange and Divergent Hydrolysis
The core of this synthetic breakthrough lies in the precise execution of the halogen-metal exchange at the C5 position of the pyrimidine ring, which is electronically activated by the adjacent nitrogen atoms. When an active metal reagent, such as isopropyl magnesium chloride-lithium chloride complex or n-butyllithium, interacts with the bromine substituent, it generates a highly reactive organometallic species. This nucleophile immediately attacks the carbonyl carbon of Boc2O, forming a tetrahedral intermediate that collapses to release tert-butoxide and establish the new carbon-carbon bond. The presence of lithium chloride additives is particularly beneficial as it enhances the solubility and reactivity of the Grignard species, ensuring complete conversion even at slightly elevated temperatures compared to pure lithiation. This mechanistic pathway effectively suppresses side reactions such as nucleophilic attack on the ring nitrogens or self-condensation, which are common pitfalls in heterocyclic functionalization.
Following the formation of the tert-butyl ester, the process offers a unique divergent capability driven by the choice of hydrolysis conditions. For the synthesis of 2-chloropyrimidine-5-carboxylic acid, mild acidic conditions are sufficient to cleave the tert-butyl group without disturbing the chlorine atom at the C2 position, preserving the handle for subsequent cross-coupling reactions. Conversely, subjecting the intermediate to alkaline reflux conditions triggers a nucleophilic aromatic substitution where the hydroxide ion displaces the chlorine atom. This transformation is facilitated by the electron-withdrawing nature of the newly formed carboxylate group, which activates the ring towards substitution. This dual-pathway mechanism provides unparalleled flexibility for medicinal chemists, allowing access to distinct chemical spaces from a single starting material while maintaining strict control over impurity profiles and regioselectivity throughout the synthesis.
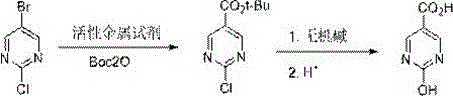
How to Synthesize 2-Chloropyrimidine-5-Carboxylic Acid Efficiently
Implementing this synthesis requires careful attention to the stoichiometry of the active metal reagent and the temperature profile during the metalation step to ensure maximum conversion. The detailed standardized synthesis steps involve dissolving 2-chloro-5-bromopyrimidine in an etheral solvent like THF or 2-MeTHF, cooling the mixture to the specified range, and slowly adding the metal reagent to manage exotherms. Once the organometallic species is generated, Boc2O is introduced to trap the intermediate, followed by a warming period to complete the reaction. The workup involves quenching with acid, extracting the product, and finally performing the specific hydrolysis step tailored to the desired final compound. For a comprehensive guide on exact molar ratios, solvent choices, and purification techniques, please refer to the structured protocol below.
- Perform halogen-metal exchange on 2-chloro-5-bromopyrimidine using Grignard or Lithium reagents at low temperature (-20°C to -75°C).
- React the resulting organometallic intermediate with Di-tert-butyl dicarbonate (Boc2O) to form the tert-butyl ester.
- Hydrolyze the ester using either acidic conditions for the chloro-acid or alkaline reflux for the hydroxy-acid derivative.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, the shift towards this Boc-based methodology offers substantial strategic benefits regarding raw material sourcing and cost stability. Unlike specialized gases or unstable reagents required for older methods, Boc2O is a commodity chemical produced at massive scales for the peptide industry, ensuring a reliable supply chain with competitive pricing. The ability to use various metal reagents, including cost-effective n-butyllithium or robust Grignard complexes, allows manufacturing teams to optimize expenses based on market fluctuations without compromising the integrity of the synthesis. Furthermore, the high yields reported in the patent examples, often exceeding 80% and reaching up to 91%, directly translate to reduced raw material consumption per kilogram of finished product. This efficiency minimizes the environmental footprint and lowers the overall cost of goods sold, making it an attractive option for cost reduction in pharmaceutical intermediate manufacturing.
- Cost Reduction in Manufacturing: The elimination of extreme cryogenic requirements (-90°C) significantly reduces energy consumption and capital expenditure on specialized cooling infrastructure. By operating at more accessible temperatures like -20°C or even 0°C with specific reagents, facilities can utilize standard jacketed reactors, drastically simplifying the production setup. Additionally, the high atom economy of the Boc-trapping reaction ensures that expensive starting materials are converted into valuable product rather than lost to side reactions or low-yield steps. This operational efficiency leads to substantial cost savings over the lifecycle of the product, enhancing the margin potential for downstream drug manufacturers.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route ensures consistent batch-to-batch quality, which is critical for maintaining uninterrupted supply to API manufacturers. The use of stable intermediates like the tert-butyl ester allows for potential storage or transport between steps if necessary, adding flexibility to the production schedule. Moreover, the avoidance of hazardous diazotization reagents and heavy metal catalysts simplifies regulatory compliance and reduces the risk of supply disruptions due to environmental audits or safety incidents. This reliability makes the manufacturer a preferred partner for long-term contracts requiring steady volumes of high-purity intermediates.
- Scalability and Environmental Compliance: Scaling this process from laboratory to commercial production is straightforward due to the homogeneous nature of the reactions and the absence of gas-liquid mass transfer limitations associated with CO2 insertion. The waste streams generated are primarily organic salts and solvents which can be readily treated or recycled using standard industrial protocols. The removal of heavy metals like zinc from the process flow further aligns with modern green chemistry principles and stringent ICH Q3D guidelines for elemental impurities. This environmental compatibility facilitates faster regulatory approval and supports the sustainability goals of global pharmaceutical companies seeking eco-friendly supply chains.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this synthetic route for industrial applications. These answers are derived directly from the experimental data and technical disclosures within the patent documentation to ensure accuracy and relevance for process development teams. Understanding these nuances is essential for successfully transferring this technology from the lab bench to the pilot plant and eventually to full-scale commercial manufacturing.
Q: Why is the Boc-protection method superior to direct CO2 carboxylation?
A: Traditional CO2 carboxylation often suffers from low yields (around 26%) and requires extreme cryogenic conditions (-90°C). The Boc-method offers significantly higher yields (up to 91%) and better batch-to-batch reproducibility.
Q: Can this route produce both chloro and hydroxy derivatives?
A: Yes, the route is divergent. Acidic hydrolysis preserves the chlorine atom to yield 2-chloropyrimidine-5-carboxylic acid, while alkaline reflux facilitates nucleophilic substitution to yield 2-hydroxypyrimidine-5-carboxylic acid.
Q: What active metal reagents are compatible with this synthesis?
A: The process is flexible, supporting Grignard reagents (like iPrMgCl·LiCl), Lithium reagents (n-BuLi), and Alkylmagnesium Lithium reagents (n-Bu3MgLi), allowing optimization based on cost and availability.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Chloropyrimidine-5-Carboxylic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality heterocyclic intermediates play in accelerating drug discovery and development timelines. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from patent literature to industrial reality is seamless and efficient. We adhere to stringent purity specifications and operate rigorous QC labs equipped with advanced analytical instrumentation to guarantee that every batch meets the exacting standards required by global regulatory bodies. Our commitment to excellence extends beyond mere compliance; we actively collaborate with clients to optimize processes for maximum yield and minimal environmental impact.
We invite procurement leaders and R&D directors to engage with us for a Customized Cost-Saving Analysis tailored to your specific project requirements. By leveraging our expertise in pyrimidine chemistry, we can help you identify opportunities to reduce costs and improve supply security for your key building blocks. Please contact our technical procurement team to request specific COA data and route feasibility assessments, and let us demonstrate how our advanced manufacturing capabilities can support your next breakthrough therapy.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
