Advanced Chiral Resolution Technology for Scalable Production of HIV Drug Intermediates
Advanced Chiral Resolution Technology for Scalable Production of HIV Drug Intermediates
The global demand for next-generation antiretroviral therapies continues to drive innovation in the synthesis of critical pharmaceutical intermediates. Patent CN111056939A introduces a significant breakthrough in the preparation of (1R,3S)-3-aminocyclopentanol, a pivotal chiral building block for the synthesis of Bictegravir. This novel methodology addresses long-standing challenges in optical purity and yield that have plagued previous synthetic routes. By leveraging a sophisticated mixed-solvent recrystallization technique, the process ensures the production of high-purity intermediates essential for modern HIV treatment regimens. As the market for integrase inhibitors expands, securing a reliable supply chain for such complex chiral amines becomes a strategic priority for pharmaceutical manufacturers worldwide.
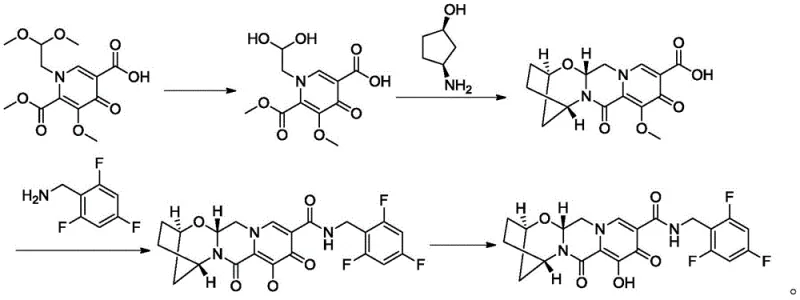
Bictegravir represents a cornerstone in contemporary HIV-1 infection management, often administered in fixed-dose combinations. The structural complexity of this molecule necessitates precise stereochemical control during its assembly. The intermediate (1R,3S)-3-aminocyclopentanol serves as a crucial scaffold, and its efficient production directly impacts the overall cost and availability of the final drug substance. Traditional methods often struggled with separating the desired enantiomer from its mirror image, leading to material loss and increased waste. The technology disclosed in this patent offers a streamlined pathway that enhances both economic efficiency and environmental sustainability, aligning with the rigorous standards expected by top-tier regulatory bodies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art techniques, such as those described in WO2015195656A2, typically relied on resolution strategies using chiral acids like (S)-naproxen in单一 solvent systems. While conceptually sound, these historical approaches frequently suffered from suboptimal outcomes, including low resolution yields and insufficient optical enrichment. For instance, earlier protocols utilizing 2-butanone and water often required multiple recrystallization cycles to achieve acceptable purity, which drastically reduced the overall mass balance of the process. Furthermore, the inability to consistently precipitate the desired diastereomeric salt under varying conditions introduced significant batch-to-batch variability. These inefficiencies not only inflated manufacturing costs but also created bottlenecks in the supply chain, making it difficult to meet the growing global demand for HIV medications without compromising on quality or lead times.
The Novel Approach
The innovative strategy presented in CN111056939A fundamentally reengineers the purification stage by introducing a ternary solvent system comprising a ketone, methanol, and water. This specific combination creates a unique solubility profile that selectively favors the crystallization of the target (1R,3S)-isomer salt while keeping impurities and the undesired enantiomer in solution. By fine-tuning the volume ratios of acetone or 2-butanone with methanol and water, the process achieves a delicate thermodynamic balance that maximizes recovery. This approach eliminates the need for excessive recycling or complex chromatographic separations, thereby simplifying the operational workflow. The result is a robust protocol that delivers superior optical purity in fewer steps, representing a paradigm shift in how chiral cyclopentyl amines are manufactured for high-value pharmaceutical applications.
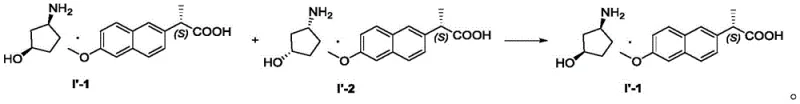
Mechanistic Insights into Diastereomeric Salt Resolution
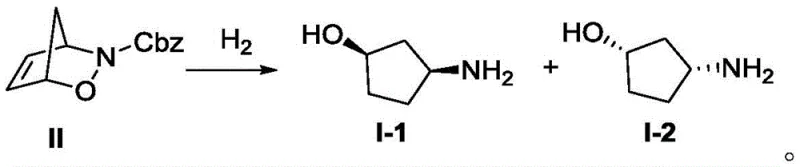
The chemical foundation of this process rests on the formation of diastereomeric salts between the racemic amine mixture and the chiral resolving agent, (S)-naproxen. Initially, the crude cis-3-aminocyclopentanol is generated via the catalytic hydrogenation of a protected precursor, specifically Compound II, using palladium on carbon. This reduction step is critical as it establishes the cis-stereochemistry required for the subsequent resolution. Following the removal of the protecting group and catalyst, the free amine mixture reacts with (S)-naproxen to form two distinct diastereomeric salts. These salts possess different physical properties, particularly solubility, which is the driving force behind their separation. The precise control of reaction temperature and solvent composition during salt formation ensures that the equilibrium favors the precipitation of the less soluble diastereomer, which corresponds to the desired (1R,3S) configuration.
Impurity control is meticulously managed through the optimization of the recrystallization parameters. The patent specifies that the volume-to-mass ratio of the solvent to the solid mixture is a critical variable, with preferred ranges ensuring complete dissolution at reflux followed by controlled crystallization upon cooling. The addition of water as a good solvent to the poor solvent mixture (ketone and methanol) acts as an anti-solvent trigger, inducing supersaturation specifically for the target salt. This mechanism effectively excludes structurally similar impurities and the opposite enantiomer from the crystal lattice. Moreover, the process allows for iterative purification; if the initial crystallization yields an ee value around 80%, a second recrystallization can elevate this to over 99%. This stepwise refinement capability provides a safety net for quality assurance, ensuring that the final product consistently meets the stringent specifications required for API synthesis.
How to Synthesize (1R,3S)-3-Aminocyclopentanol Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for laboratory and pilot-scale production, emphasizing reproducibility and safety. The procedure begins with the preparation of the crude amine, followed by salt formation and the critical recrystallization steps. Operators must pay close attention to solvent ratios and temperature gradients to maximize yield. The detailed standardized synthesis steps below outline the specific conditions required to replicate the high success rates reported in the patent examples, ensuring that technical teams can implement this methodology with confidence.
- Perform catalytic hydrogenation of Compound II using Pd/C in methanol to obtain crude cis-3-aminocyclopentanol mixture.
- React the crude amine mixture with (S)-Naproxen in isopropanol/water to form diastereomeric salts.
- Purify the target salt (Formula I'-1) via recrystallization using a specific ketone/methanol/water solvent system to achieve >90% ee.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement specialists and supply chain directors, the adoption of this advanced resolution technology translates into tangible strategic benefits beyond mere technical superiority. The elimination of complex chromatographic purifications and the reliance on commodity solvents significantly streamline the manufacturing process. This simplification reduces the dependency on specialized equipment and lowers the barrier for scaling up production capacity. Furthermore, the high yield and optical purity reduce the need for reprocessing batches, which minimizes waste generation and associated disposal costs. By integrating this efficient route into the supply chain, organizations can mitigate risks associated with raw material shortages and ensure a steady flow of high-quality intermediates to downstream API manufacturers.
- Cost Reduction in Manufacturing: The process utilizes widely available and inexpensive solvents such as acetone, methanol, and isopropanol, avoiding the need for costly proprietary reagents or transition metal catalysts in the resolution step. The high recovery rate of the desired isomer means that less starting material is wasted, directly lowering the cost of goods sold. Additionally, the simplified workup procedures reduce energy consumption and labor hours required for purification, contributing to substantial overall cost savings in the production of this critical pharmaceutical intermediate.
- Enhanced Supply Chain Reliability: By establishing a robust synthetic route that is less sensitive to minor variations in reaction conditions, suppliers can guarantee more consistent delivery schedules. The ability to achieve high optical purity through recrystallization rather than relying on scarce chiral catalysts removes a potential bottleneck in the supply chain. This reliability is crucial for maintaining continuous production lines for life-saving medications, ensuring that patients have uninterrupted access to their treatments without the risk of stockouts caused by manufacturing delays or quality failures.
- Scalability and Environmental Compliance: The use of green solvents and the reduction of waste streams align with increasingly strict environmental regulations globally. The process is designed to be easily scalable from kilogram to multi-ton quantities without significant re-engineering, facilitating rapid response to market demand surges. The efficient use of materials and the potential for solvent recovery further enhance the sustainability profile of the manufacturing operation, making it an attractive option for companies committed to reducing their carbon footprint and adhering to green chemistry principles.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented resolution technology. These insights are derived directly from the experimental data and comparative examples provided in the patent documentation, offering clarity on performance metrics and operational feasibility. Understanding these details is essential for technical teams evaluating the integration of this process into their existing manufacturing portfolios.
Q: What is the primary advantage of the new recrystallization solvent system?
A: The patented mixed solvent system (ketone/methanol/water) significantly improves the optical purity (ee value) to over 90% in a single step, overcoming the low yield and purity issues of previous methods using single solvents like MEK.
Q: Can this process be scaled for commercial API production?
A: Yes, the process utilizes standard unit operations such as hydrogenation and recrystallization with commercially available solvents, making it highly suitable for large-scale manufacturing without requiring exotic catalysts or extreme conditions.
Q: What is the final optical purity achievable with this method?
A: Through secondary recrystallization, the method can achieve an ee value of up to 99.3%, meeting the stringent quality requirements for HIV integrase inhibitor synthesis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (1R,3S)-3-Aminocyclopentanol Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-purity intermediates play in the development and commercialization of advanced therapeutics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the rigorous demands of the global pharmaceutical market. We are committed to delivering products that adhere to stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical capabilities. Our expertise in chiral resolution and process optimization allows us to offer solutions that are not only chemically superior but also economically viable for long-term partnerships.
We invite you to collaborate with us to explore how this innovative synthesis route can benefit your specific project requirements. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume needs. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our commitment to excellence can support your supply chain goals and accelerate your time to market.
