Advanced Chiral Quinoline Imidazoline Ligands and Iron Complexes for Scalable Asymmetric Hydrogenation
The pharmaceutical and fine chemical industries are constantly seeking more sustainable and cost-effective methods for synthesizing chiral building blocks, which are critical for drug development. Patent CN114989137A introduces a groundbreaking class of chiral imine-containing quinoline imidazoline compounds, represented by general formula (1), and their corresponding transition metal complexes. These novel ligands address a significant gap in asymmetric catalysis by enabling the use of earth-abundant iron instead of precious metals. The structural versatility of these compounds allows for fine-tuning of steric and electronic properties, making them highly effective homogeneous catalysts. 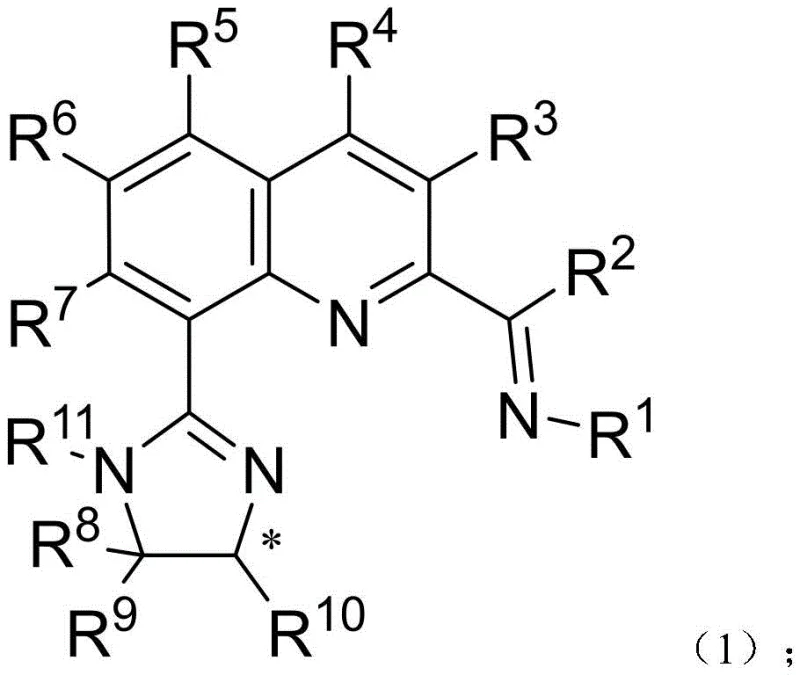 . This innovation represents a pivotal shift towards greener chemistry, offering a reliable pathway for producing high-purity pharmaceutical intermediates with exceptional optical purity, often exceeding 90% enantiomeric excess.
. This innovation represents a pivotal shift towards greener chemistry, offering a reliable pathway for producing high-purity pharmaceutical intermediates with exceptional optical purity, often exceeding 90% enantiomeric excess.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, asymmetric hydrogenation of olefins has been dominated by catalytic systems based on noble metals such as rhodium, ruthenium, and iridium. While effective, these metals suffer from severe drawbacks including high cost, geological scarcity, and potential toxicity issues related to residual metal contamination in final drug products. Furthermore, existing literature, such as Patent CN112851479A, indicates that while some iron-based systems exist, they are often limited to styrene-type substrates and perform poorly with aliphatic 1,1-dialkyl olefins. This limitation restricts their utility in synthesizing complex aliphatic chains found in many bioactive molecules. The reliance on expensive ligands and harsh reaction conditions in older methodologies further exacerbates the economic and environmental burden on manufacturers seeking cost reduction in chiral ligand manufacturing.
The Novel Approach
The technology disclosed in CN114989137A overcomes these barriers through a unique molecular architecture that combines a quinoline backbone with a chiral imidazoline ring. This specific arrangement creates a robust coordination environment that stabilizes low-valent iron species, enabling them to catalyze the hydrogenation of challenging 1,1-dialkyl olefins with high efficiency. 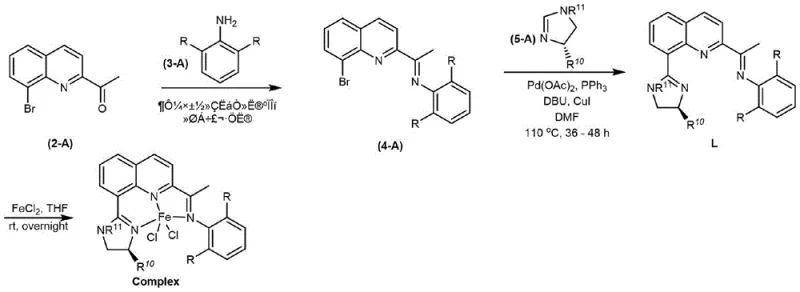 . By shifting the paradigm from noble metals to iron, this approach not only drastically lowers the cost of goods but also simplifies the supply chain by utilizing widely available base metal salts. The method demonstrates that high enantioselectivity is achievable without the premium price tag of traditional catalysts, positioning it as a superior choice for the commercial scale-up of complex pharmaceutical intermediates.
. By shifting the paradigm from noble metals to iron, this approach not only drastically lowers the cost of goods but also simplifies the supply chain by utilizing widely available base metal salts. The method demonstrates that high enantioselectivity is achievable without the premium price tag of traditional catalysts, positioning it as a superior choice for the commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into FeCl2-Catalyzed Asymmetric Hydrogenation
The core of this technology lies in the formation of a stable metal complex, as shown in formula (6), where the chiral ligand coordinates with a transition metal, preferably iron (Fe). 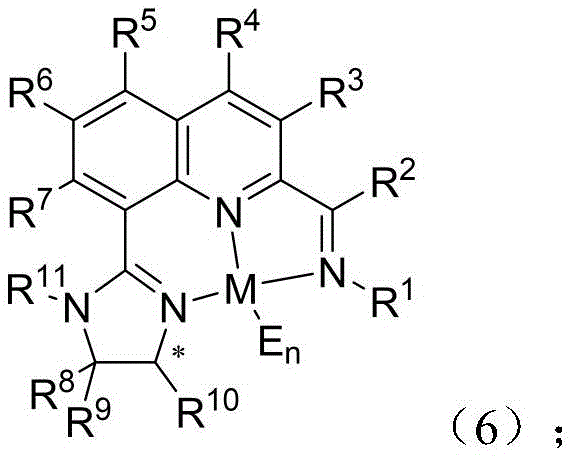 . In this complex, the nitrogen atoms from both the quinoline imine and the imidazoline ring act as donor sites, creating a rigid chiral pocket around the metal center. This geometry is crucial for differentiating between the enantiotopic faces of the olefin substrate during the hydrogenation process. The presence of bulky substituents on the imidazoline ring (R8, R9, R10) and the aniline moiety (R1) provides the necessary steric hindrance to enforce high stereocontrol, ensuring that the hydride transfer occurs selectively to produce the desired chiral alkane.
. In this complex, the nitrogen atoms from both the quinoline imine and the imidazoline ring act as donor sites, creating a rigid chiral pocket around the metal center. This geometry is crucial for differentiating between the enantiotopic faces of the olefin substrate during the hydrogenation process. The presence of bulky substituents on the imidazoline ring (R8, R9, R10) and the aniline moiety (R1) provides the necessary steric hindrance to enforce high stereocontrol, ensuring that the hydride transfer occurs selectively to produce the desired chiral alkane.
The catalytic cycle involves the activation of molecular hydrogen or a silane reducing agent by the iron center, followed by the insertion of the olefin substrate into the metal-hydride bond.  . The rigidity of the quinoline-imidazoline scaffold minimizes non-productive conformational changes, thereby reducing the formation of racemic byproducts. This precise control over the reaction trajectory is what allows the system to achieve enantioselectivity levels greater than 85%, and in optimized cases, exceeding 99%. For R&D teams, understanding this mechanism is vital for impurity control, as it highlights how subtle changes in the R-groups can be leveraged to suppress specific side reactions and maximize the yield of the target enantiomer.
. The rigidity of the quinoline-imidazoline scaffold minimizes non-productive conformational changes, thereby reducing the formation of racemic byproducts. This precise control over the reaction trajectory is what allows the system to achieve enantioselectivity levels greater than 85%, and in optimized cases, exceeding 99%. For R&D teams, understanding this mechanism is vital for impurity control, as it highlights how subtle changes in the R-groups can be leveraged to suppress specific side reactions and maximize the yield of the target enantiomer.
How to Synthesize Chiral Quinoline Imidazoline Compounds Efficiently
The preparation of these high-value catalysts follows a logical and scalable three-step sequence that begins with readily available starting materials. The process starts with the condensation of a 2-acyl-8-bromoquinoline with a substituted aniline to form the imine intermediate. This is followed by a key palladium-catalyzed coupling reaction with a chiral imidazoline derivative, which installs the critical stereocenter. Finally, the free ligand is complexed with ferrous chloride to generate the active catalyst. The detailed standardized synthesis steps, including specific molar ratios, solvent choices like DMF and THF, and purification protocols via column chromatography, are outlined in the guide below.
- Condense 2-acyl-8-bromoquinoline with substituted aniline to form the imine intermediate.
- Perform a copper-mediated coupling reaction between the bromo-imine and chiral imidazoline under palladium catalysis.
- Complex the resulting chiral ligand with ferrous chloride in tetrahydrofuran to generate the active iron catalyst.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this iron-based catalytic technology offers transformative benefits that extend beyond simple chemistry. The shift from precious metals to iron fundamentally alters the cost structure of chiral synthesis, removing the volatility associated with rhodium and ruthenium markets. Additionally, the robust nature of the ligand synthesis, which tolerates standard organic solvents and moderate temperatures, ensures consistent batch-to-batch quality, a critical factor for regulatory compliance in pharmaceutical manufacturing.
- Cost Reduction in Manufacturing: The most immediate impact is the substantial cost savings derived from replacing expensive noble metal catalysts with inexpensive iron salts. Since iron is one of the most abundant elements on earth, the raw material cost is negligible compared to traditional systems. Furthermore, the elimination of costly heavy metal scavenging steps, often required to meet strict ppm limits for noble metals in APIs, streamlines the downstream processing workflow. This qualitative reduction in processing complexity translates directly to lower operational expenditures and a more competitive pricing model for the final chiral intermediate.
- Enhanced Supply Chain Reliability: Relying on geopolitically scarce noble metals introduces significant supply chain risk. By transitioning to an iron-based system, manufacturers can secure a more stable and resilient supply of catalytic materials. The starting materials for the ligand synthesis, such as bromoquinolines and various anilines, are commodity chemicals with established global supply networks. This diversification of the supply base reduces lead times for high-purity pharmaceutical intermediates and mitigates the risk of production stoppages due to raw material shortages.
- Scalability and Environmental Compliance: The synthetic routes described in the patent utilize common organic solvents like toluene, DMF, and THF, which are well-understood in industrial settings and can be efficiently recovered and recycled. The reaction conditions, often proceeding at room temperature or mild reflux, reduce energy consumption compared to high-pressure hydrogenation processes. From an environmental perspective, the low toxicity of iron aligns perfectly with green chemistry initiatives, simplifying waste disposal and reducing the environmental footprint of the manufacturing facility, which is increasingly important for meeting corporate sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this chiral iron catalyst technology. These answers are derived directly from the experimental data and specifications provided in the patent documentation, ensuring accuracy for your process evaluation.
Q: What is the primary advantage of this iron catalyst over traditional noble metal systems?
A: The primary advantage is the substitution of expensive and scarce noble metals like rhodium and ruthenium with abundant and low-toxicity iron, significantly reducing raw material costs while maintaining high enantioselectivity (>85% ee) for challenging 1,1-dialkyl olefin substrates.
Q: Can this catalytic system be scaled for industrial production?
A: Yes, the synthesis utilizes robust reaction conditions such as refluxing in DMF and room temperature complexation, which are amenable to large-scale manufacturing. The use of common solvents and stable reagents further supports commercial scalability.
Q: What types of substrates are compatible with this hydrogenation method?
A: This system is specifically designed for the asymmetric hydrogenation of 1,1-dialkyl olefin compounds, including those with aryl and alkoxy substituents, producing chiral alkane compounds with high regioselectivity and optical purity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Quinoline Imidazoline Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of this novel catalytic technology for the next generation of chiral drug synthesis. Our team of expert chemists is fully equipped to replicate and optimize the synthesis of these complex quinoline-imidazoline ligands and their iron complexes. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab-scale discovery to full-scale manufacturing is seamless. Our state-of-the-art rigorous QC labs guarantee stringent purity specifications, delivering materials that meet the highest international standards for pharmaceutical applications.
We invite you to collaborate with us to leverage this cost-effective and sustainable catalytic solution for your specific projects. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your current process. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our supply of these advanced chiral ligands can enhance your production efficiency and profitability.
