Advanced Rhodium-Catalyzed Hydrogenation for Scalable Tetrahydroquinoxaline Pharmaceutical Intermediates Production
Advanced Rhodium-Catalyzed Hydrogenation for Scalable Tetrahydroquinoxaline Pharmaceutical Intermediates Production
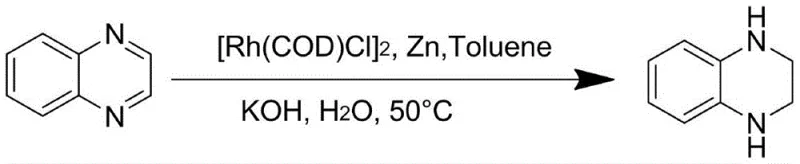
The pharmaceutical and agrochemical industries are constantly seeking robust, scalable, and cost-effective methodologies for constructing privileged scaffolds such as tetrahydroquinoxalines. A significant breakthrough in this domain is detailed in patent CN110483420B, which discloses a novel preparation method for tetrahydroquinoxaline compounds utilizing a rhodium-catalyzed transfer hydrogenation system. This technology represents a paradigm shift from traditional high-pressure hydrogenation or harsh chemical reduction methods, offering a pathway that utilizes water as a benign hydrogen source in conjunction with zinc powder and a rhodium catalyst. For R&D directors and process chemists, this approach offers a compelling alternative that operates under mild temperatures ranging from 40°C to 80°C, thereby minimizing thermal degradation risks and energy consumption. The strategic implementation of this patent-protected methodology allows for the efficient synthesis of complex heterocyclic intermediates essential for drug discovery and development pipelines.
From a supply chain and procurement perspective, the adoption of this rhodium-catalyzed protocol addresses critical pain points associated with the sourcing and handling of hazardous reagents. By replacing high-pressure hydrogen gas cylinders or explosive hydride reagents with stable zinc powder and water, the process inherently lowers the barrier to entry for contract manufacturing organizations (CMOs) and fine chemical suppliers. This transition not only enhances workplace safety but also streamlines regulatory compliance regarding the storage and transport of dangerous goods. As a reliable pharmaceutical intermediate supplier, understanding and leveraging such innovative synthetic routes is paramount for maintaining competitive advantage in the global market, ensuring consistent delivery of high-purity materials while mitigating the risks associated with volatile supply chains for specialized reducing agents.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of tetrahydroquinoxaline derivatives has relied heavily on catalytic hydrogenation using molecular hydrogen gas under high pressure or chemical reductions employing stoichiometric amounts of metal hydrides. These conventional approaches present substantial logistical and safety challenges, particularly when scaling up to industrial volumes. High-pressure hydrogenation requires specialized autoclaves and rigorous safety protocols to prevent leaks and explosions, leading to significant capital expenditure and increased operational complexity. Furthermore, the use of formic acid or other organic hydrogen donors often generates stoichiometric amounts of waste byproducts, complicating downstream purification and increasing the environmental footprint of the manufacturing process. The harsh conditions associated with these legacy methods can also lead to poor chemoselectivity, resulting in the formation of unwanted side products that compromise the purity profile of the final active pharmaceutical ingredient (API) intermediate.
The Novel Approach
In stark contrast, the methodology described in patent CN110483420B introduces a transformative strategy that utilizes water as the primary hydrogen source, mediated by a rhodium catalyst and activated by zinc powder. This system operates under atmospheric pressure or slight positive pressure of inert gas, completely eliminating the need for high-pressure hydrogen infrastructure. The reaction proceeds smoothly in common organic solvents such as toluene or dichloromethane at moderate temperatures, typically between 40°C and 80°C. This mild condition profile ensures excellent functional group tolerance, allowing for the preservation of sensitive substituents on the quinoxaline ring that might otherwise be compromised under vigorous reducing conditions. The integration of water as a reagent not only drastically reduces raw material costs but also aligns with green chemistry principles by improving atom economy and minimizing hazardous waste generation.

The versatility of this novel approach is further evidenced by its compatibility with a wide array of substrates, including those bearing electron-withdrawing halogens and electron-donating alkyl groups. The use of additives such as potassium hydroxide or ligands like bipyridine (Bpy) fine-tunes the catalytic activity, ensuring high conversion rates and selectivity. For procurement managers, this translates to a more flexible supply chain where raw material availability is less constrained by the geopolitical or logistical issues often associated with specialized reducing gases. The ability to achieve high yields, such as the 91% yield reported for the unsubstituted tetrahydroquinoxaline in Example 1, demonstrates the robustness of this chemistry for commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Rhodium-Catalyzed Transfer Hydrogenation
The core of this technological advancement lies in the intricate interplay between the rhodium catalyst, the zinc reductant, and the water molecule. Mechanistically, the zinc powder serves as a sacrificial electron donor, activating the rhodium center and facilitating the cleavage of the O-H bond in water to generate reactive hydride species in situ. This transfer hydrogenation mechanism bypasses the kinetic barriers associated with direct hydrogen gas activation, allowing the reduction to proceed efficiently at lower temperatures. The rhodium complex, whether it be [Rh(COD)Cl]2 or [Cp*RhCl2]2, cycles through oxidation states to deliver hydride equivalents to the electron-deficient nitrogen-containing heterocycle. This precise control over the reduction potential is crucial for preventing over-reduction of the aromatic benzene ring fused to the pyrazine moiety, ensuring that only the desired di-imine bonds are saturated to form the tetrahydroquinoxaline core.
From an impurity control perspective, the mild nature of this catalytic system significantly reduces the formation of degradation products and polymeric tars that often plague traditional reduction methods. The use of water as a proton source ensures that the reaction medium remains relatively neutral to slightly basic (depending on the additive used), which suppresses acid-catalyzed decomposition pathways. Furthermore, the specific molar ratios optimized in the patent, such as a substrate-to-zinc ratio of 1:3 to 1:4, ensure that there is sufficient reducing power to drive the reaction to completion without excessive excess that would complicate workup. The ability to monitor reaction progress via thin-layer chromatography (TLC) allows for real-time quality control, enabling operators to quench the reaction immediately upon substrate consumption, thereby maximizing yield and minimizing the residence time of the product in the reaction mixture which could lead to secondary reactions.
How to Synthesize Tetrahydroquinoxaline Efficiently
Implementing this synthesis route requires strict adherence to inert atmosphere techniques to protect the sensitive rhodium catalyst and the reactive zinc surface from oxidation. The process begins with the preparation of a dry solvent system, typically toluene, under a nitrogen or argon blanket. The catalyst and additives are introduced first to establish the active catalytic species before the substrate is added. This sequential addition is critical for maximizing catalyst turnover numbers and ensuring consistent batch-to-batch reproducibility. The following guide outlines the standardized operational procedure derived from the patent examples, providing a clear roadmap for process chemists aiming to replicate these results in a pilot or production setting.
- Under an inert nitrogen or argon atmosphere, dissolve the rhodium metal catalyst (such as [Rh(COD)Cl]2), zinc powder, and an additive like potassium hydroxide in a dry solvent such as toluene.
- Add the quinoxaline-based reaction substrate to the mixture, followed by the addition of water, ensuring the molar ratio of substrate to water is maintained between 1: 10 and 1:30.
- Heat the reaction mixture to a temperature between 40°C and 80°C, monitor progress via TLC until substrate consumption is complete, and purify the residue using silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
The transition to this rhodium-catalyzed water-based reduction methodology offers profound commercial benefits that extend far beyond the laboratory bench. For procurement managers tasked with optimizing the cost of goods sold (COGS), the replacement of expensive and hazardous hydrogen gas or metal hydrides with commodity chemicals like zinc powder and water represents a significant opportunity for cost reduction in pharmaceutical intermediate manufacturing. The elimination of high-pressure reactor requirements also lowers the barrier for contract manufacturing partners, potentially expanding the pool of qualified suppliers and driving down processing fees through increased competition. Additionally, the simplified workup procedures, often involving standard silica gel chromatography or crystallization, reduce the consumption of specialized purification resins and solvents, further contributing to overall process economics.
- Cost Reduction in Manufacturing: The utilization of water as a hydrogen source fundamentally alters the cost structure of the reduction step. Unlike molecular hydrogen which requires compression, storage, and safety monitoring infrastructure, water is universally available and virtually free. The zinc powder used as a co-reductant is a bulk commodity chemical with a stable global supply chain, insulating manufacturers from the price volatility often seen with specialty gases. Furthermore, the high atom economy of the reaction means that a larger proportion of the input mass ends up in the final product, reducing waste disposal costs and improving the overall material efficiency of the process. This economic efficiency is compounded by the ability to run the reaction at near-atmospheric pressure, which significantly reduces energy consumption related to pressurization and heating compared to traditional high-pressure hydrogenation units.
- Enhanced Supply Chain Reliability: Dependence on specialized reagents can create single points of failure in a supply chain, but this method relies on widely available materials. Zinc powder, rhodium catalysts, and common organic solvents are stocked by numerous global chemical distributors, ensuring continuity of supply even during regional disruptions. The robustness of the reaction conditions, which tolerate a range of temperatures and solvent choices (toluene, DCM, acetonitrile), provides process engineers with the flexibility to substitute solvents based on local availability or price fluctuations without compromising reaction performance. This flexibility is crucial for maintaining production schedules and meeting tight delivery deadlines for downstream API synthesis, thereby enhancing the reliability of the supply chain for critical pharmaceutical intermediates.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces new safety and environmental challenges, but this technology is inherently designed for scalability. The absence of high-pressure gas eliminates the risk of catastrophic vessel failure, making it safer to scale from kilogram to tonne quantities. From an environmental standpoint, the use of water generates benign byproducts (zinc oxides/hydroxides) that are easier to treat and dispose of compared to the acidic or toxic waste streams generated by formic acid or borohydride reductions. This alignment with green chemistry principles facilitates easier regulatory approval and reduces the environmental compliance burden on manufacturing sites, making it an attractive option for companies striving to meet sustainability goals and reduce their carbon footprint in fine chemical production.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this rhodium-catalyzed synthesis method. These answers are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing clarity for stakeholders evaluating this technology for adoption. Understanding these nuances is essential for making informed decisions about process integration and resource allocation.
Q: What are the advantages of using water as a hydrogen source in this synthesis?
A: Using water as a hydrogen source eliminates the need for high-pressure hydrogen gas equipment, significantly enhancing operational safety and reducing capital expenditure on specialized reactors. Furthermore, water is inexpensive and environmentally benign, contributing to a greener synthesis profile with strong atom economy compared to traditional formic acid or hydrogen gas methods.
Q: What is the optimal catalyst loading for this rhodium-catalyzed reduction?
A: The patent data indicates that a molar ratio of quinoxaline substrate to rhodium metal between 1:0.025 and 1:0.05 yields optimal results. Specifically, a ratio of 1:0.025 has been demonstrated to provide high yields, such as 91% for the unsubstituted tetrahydroquinoxaline, balancing catalytic efficiency with cost-effectiveness for large-scale production.
Q: Can this method tolerate various substituents on the quinoxaline ring?
A: Yes, the method exhibits broad substrate scope, successfully reducing quinoxalines substituted with alkyl groups, halogens (fluorine, chlorine, bromine), and alkoxy groups. Examples in the patent show successful conversion of 6-methyl, 6-chloro, 6-fluoro, and 6-bromo quinoxalines into their corresponding tetrahydro-derivatives with yields ranging from moderate to excellent.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tetrahydroquinoxaline Supplier
The technological potential of rhodium-catalyzed transfer hydrogenation for producing tetrahydroquinoxaline derivatives is immense, offering a cleaner, safer, and more economical route to these valuable building blocks. At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods like this are seamlessly translated into robust industrial processes. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs that utilize advanced analytical techniques to verify the identity and purity of every batch, guaranteeing that our clients receive materials that meet the highest standards required for pharmaceutical applications.
We invite you to collaborate with us to leverage this advanced synthesis technology for your next project. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and purity needs. We encourage you to contact our technical procurement team to request specific COA data for our tetrahydroquinoxaline inventory and to discuss route feasibility assessments for your custom synthesis projects. By partnering with us, you gain access to a supply chain that is not only reliable and compliant but also driven by continuous innovation in process chemistry.
