Advanced Photocatalytic Synthesis of 3-Alkyl Quinoxalin-2(1H)-ones for Commercial Scale-Up
Introduction to Next-Generation Quinoxalinone Synthesis
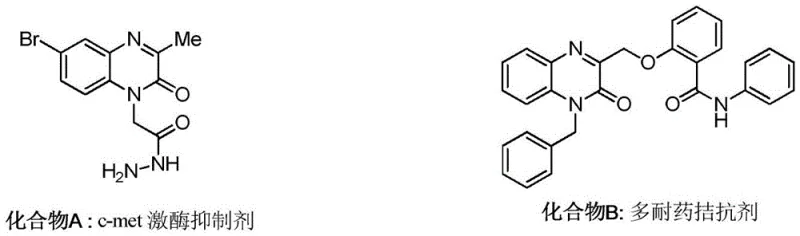
The development of efficient synthetic routes for nitrogen-containing heterocycles remains a cornerstone of modern pharmaceutical research, particularly for scaffolds exhibiting potent biological activity. Patent CN110117260B introduces a transformative methodology for the preparation of 3-alkyl quinoxalin-2(1H)-one compounds, a class of structures pivotal in the design of c-met kinase inhibitors and multidrug resistance antagonists. This innovation leverages visible-light photocatalysis to achieve direct C3-alkylation, bypassing the limitations of traditional thermal methods. By utilizing environmentally benign hypervalent iodine reagents and a robust ruthenium-based photocatalyst, this process offers a sustainable alternative to hazardous peroxide-mediated oxidations. The technology enables the construction of complex molecular architectures under exceptionally mild conditions, ensuring high purity and structural integrity essential for downstream drug development applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the functionalization of the quinoxalinone core at the C3 position has been fraught with significant chemical and operational challenges. Prior art methodologies often relied on iron-catalyzed systems requiring elevated temperatures or the use of tert-butyl hydroperoxide, which poses inherent explosion risks during scale-up. Furthermore, existing protocols were frequently restricted to specific types of alkylation, such as oxyalkylation or cyanoalkylation, failing to provide a general solution for introducing simple alkyl groups like methyl, ethyl, or cyclohexyl moieties. These limitations not only constrained the chemical space accessible to medicinal chemists but also introduced severe safety hazards and waste disposal issues associated with heavy metal residues and unstable oxidants. The inability to tolerate sensitive functional groups further narrowed the utility of these older methods in the synthesis of advanced pharmaceutical intermediates.
The Novel Approach
The patented technology revolutionizes this landscape by employing a visible-light-driven radical mechanism that operates efficiently at room temperature. Instead of dangerous peroxides, the process utilizes diacyloxyiodobenzenes generated in situ from inexpensive carboxylic acids and iodobenzene diacetate. This shift allows for the precise generation of alkyl radicals that selectively attack the C3 position of the quinoxalinone ring with high regioselectivity. The use of a ruthenium photocatalyst activated by low-power blue LEDs ensures energy efficiency while eliminating the need for thermal activation. This approach dramatically expands the substrate scope, enabling the incorporation of diverse aliphatic chains, including those with terminal alkynes and ester groups, thereby providing a versatile platform for the rapid diversification of bioactive quinoxalinone libraries.

Mechanistic Insights into Visible-Light Photoredox Catalysis
The efficacy of this synthesis relies on a sophisticated photoredox catalytic cycle initiated by the excitation of the ruthenium complex. Upon absorption of visible light photons, the ground-state photocatalyst transitions to an excited state, possessing sufficient reducing power to engage with the hypervalent iodine species. This single-electron transfer (SET) event triggers the fragmentation of the diacyloxyiodobenzene, releasing carbon dioxide and generating a highly reactive alkyl radical alongside an iodobenzene byproduct. The transient alkyl radical then undergoes addition to the electron-deficient C3 position of the quinoxalinone substrate, forming a distinct radical intermediate. Subsequent oxidation of this intermediate by the oxidized form of the photocatalyst regenerates the ground-state catalyst and yields a cationic species, which finally undergoes deprotonation to restore aromaticity and afford the stable 3-alkylated product. This elegant cycle ensures catalytic turnover with minimal waste generation.
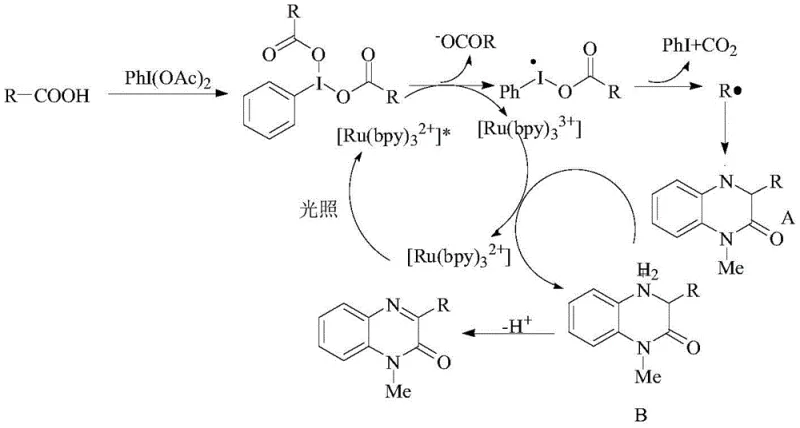
From an impurity control perspective, the mildness of the radical generation mechanism is paramount. Unlike thermal radical initiators that produce a broad distribution of radical species leading to complex byproduct profiles, this photochemical method generates alkyl radicals in a controlled, steady-state manner. The specificity of the hypervalent iodine fragmentation minimizes side reactions such as homocoupling of the alkyl radicals or over-oxidation of the sensitive quinoxalinone core. Furthermore, the use of PEG-200 as a solvent provides a stabilizing environment that suppresses non-productive radical pathways. The result is a reaction profile characterized by high conversion rates and exceptional selectivity, significantly reducing the burden on downstream purification processes and ensuring that the final API intermediate meets stringent regulatory specifications for residual solvents and related substances.
How to Synthesize 3-Alkyl Quinoxalinone Efficiently
The operational simplicity of this protocol makes it highly attractive for both laboratory discovery and process chemistry teams. The procedure begins with the straightforward preparation of the hypervalent iodine reagent, followed by a one-pot photocatalytic reaction that requires no specialized high-pressure equipment. The reaction proceeds smoothly under ambient air conditions, eliminating the need for rigorous inert atmosphere techniques often required for sensitive organometallic transformations. Detailed standardized synthesis steps, including precise stoichiometry, workup procedures, and purification parameters, are provided in the technical guide below to ensure reproducibility across different manufacturing sites.
- Preparation of Diacyloxyiodobenzene: React aliphatic carboxylic acid with iodobenzene diacetate in chloroform at 50°C under reduced pressure to form the hypervalent iodine precursor.
- Photocatalytic Reaction Setup: Mix the quinoxalinone substrate, the prepared diacyloxyiodobenzene, and Ru(bpy)3Cl2·6H2O (1 mol%) in PEG-200 solvent.
- Visible Light Irradiation: Stir the mixture under 3W blue LED irradiation at room temperature (25°C) for 6-12 hours until TLC indicates completion.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement specialists and supply chain managers, the adoption of this photocatalytic technology offers compelling strategic benefits centered on cost stability and operational safety. The reliance on commodity chemicals such as fatty acids and iodobenzene diacetate insulates the supply chain from the volatility associated with scarce precious metal catalysts or specialized oxidants. Moreover, the elimination of hazardous peroxides reduces the regulatory burden and insurance costs associated with storing and handling explosive materials. The ability to run reactions at room temperature significantly lowers energy consumption compared to thermal processes requiring reflux or cryogenic cooling, contributing to a lower overall carbon footprint and reduced utility costs for large-scale production facilities.
- Cost Reduction in Manufacturing: The economic model of this process is driven by the use of low-loading catalysts and inexpensive, readily available starting materials. By avoiding expensive transition metals like palladium or platinum and replacing them with a ruthenium complex used at merely 1 mol%, the direct material cost is substantially minimized. Additionally, the simplified workup procedure, which avoids complex aqueous quenches required for reactive metal hydrides, reduces labor time and solvent usage. The high yields reported across various substrates mean less raw material is wasted, directly improving the cost-of-goods-sold (COGS) for the final pharmaceutical intermediate.
- Enhanced Supply Chain Reliability: Sourcing reliability is critical for continuous manufacturing, and this method excels by utilizing building blocks that are commercially abundant. Fatty acids and quinoxalinone derivatives are produced globally at multi-ton scales, ensuring that raw material shortages are unlikely to disrupt production schedules. The robustness of the reaction conditions means that the process is less sensitive to minor variations in reagent quality or environmental factors, leading to consistent batch-to-batch performance. This reliability allows for more accurate forecasting and inventory management, reducing the need for excessive safety stock and freeing up working capital.
- Scalability and Environmental Compliance: Scaling photochemical reactions has historically been challenging, but the use of low-power LEDs and transparent reactors makes this technology highly adaptable to flow chemistry or large batch vessels. The process generates benign byproducts such as iodobenzene and carbon dioxide, simplifying waste treatment and ensuring compliance with increasingly strict environmental regulations. The use of PEG-200, a non-volatile and biodegradable solvent, further enhances the green credentials of the process, facilitating easier permitting and reducing the environmental impact of solvent emissions and disposal.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this photocatalytic alkylation technology. These insights are derived directly from the experimental data and optimization studies detailed in the patent documentation, providing clarity on substrate compatibility and process parameters. Understanding these nuances is essential for project managers evaluating the feasibility of integrating this route into existing production pipelines.
Q: What are the advantages of this photocatalytic method over traditional metal catalysis?
A: This method avoids toxic heavy metal catalysts and hazardous peroxides, operating instead under mild room temperature conditions with visible light, significantly improving safety and environmental compliance.
Q: Can this synthesis accommodate diverse alkyl groups at the C3 position?
A: Yes, the protocol demonstrates excellent functional group tolerance, successfully introducing linear, branched, cyclic, and functionalized alkyl chains including those with ester and alkyne moieties.
Q: Is the PEG-200 solvent recyclable for industrial scale-up?
A: PEG-200 is a green, non-volatile solvent that simplifies product isolation and can potentially be recovered and reused, aligning with green chemistry principles for large-scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Alkyl Quinoxalinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced synthetic methodologies play in accelerating drug development timelines. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from benchtop discovery to industrial manufacturing is seamless. We are committed to delivering high-purity 3-alkyl quinoxalinone intermediates that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our expertise in photocatalysis allows us to optimize this specific route for maximum efficiency and cost-effectiveness tailored to your project needs.
We invite you to collaborate with us to leverage this cutting-edge technology for your next pharmaceutical program. Please contact our technical procurement team to request a Customized Cost-Saving Analysis specific to your target molecule. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our manufacturing capabilities can support your supply chain goals and drive your project forward with confidence.
