Scalable Gold-Catalyzed Synthesis of High-Purity Phosphoenyl Esters for Pharmaceutical Applications
Introduction to Next-Generation Phosphoenyl Ester Synthesis
The landscape of fine chemical manufacturing is undergoing a significant transformation driven by the urgent need for safer, more sustainable, and cost-effective synthetic methodologies. A pivotal advancement in this domain is detailed in patent CN110437277B, which discloses a highly selective method for synthesizing phosphoenyl ester compounds, also known as alkenyl phosphates. These versatile intermediates are critical building blocks in the development of complex pharmaceutical agents, agrochemicals, and functional materials, serving as precursors for further elaboration into biologically active molecules. The core innovation lies in the strategic replacement of traditional, hazardous catalytic systems with a novel bifunctional gold catalyst, marking a substantial leap forward in process safety and efficiency. By leveraging the unique electronic and steric properties of gold, this technology enables the direct hydrophosphorylation of alkynes under remarkably mild conditions, thereby addressing long-standing challenges related to toxicity and energy consumption in the production of high-value olefinic phosphates.
For research and development directors and procurement strategists alike, the implications of this patent extend far beyond the laboratory bench. The ability to access complex alkenyl phosphate scaffolds with high stereoselectivity and purity opens new avenues for drug discovery and process optimization. Historically, the synthesis of these motifs has been plagued by the use of stoichiometric toxic reagents or precious metals that demand rigorous purification protocols. The methodology described in CN110437277B circumvents these bottlenecks by utilizing a catalytic system that is not only less toxic but also operates at significantly lower temperatures, reducing the thermal load on manufacturing infrastructure. This report provides a deep technical and commercial analysis of this breakthrough, offering actionable insights for stakeholders seeking to secure a reliable supply chain for advanced chemical intermediates while adhering to increasingly stringent environmental and safety regulations.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to the advent of this gold-catalyzed technology, the industrial synthesis of alkenyl phosphates was largely dependent on two primary methodologies, both of which present severe drawbacks for modern manufacturing. The first conventional approach involves the use of ruthenium catalysts to mediate the stereoselective addition of phosphoric acid to alkynes. While effective, this method typically necessitates extremely high reaction temperatures, often reaching up to 140°C, which imposes significant energy costs and limits the compatibility with thermally sensitive functional groups commonly found in pharmaceutical intermediates. The second prevalent method relies on mercury catalysis, frequently employing mercury acetate in conjunction with boron trifluoride etherate. This route is fundamentally flawed from a safety and environmental perspective due to the extreme toxicity of mercury compounds, which creates massive liabilities regarding worker safety, waste disposal, and product contamination. Furthermore, mercury-catalyzed reactions often require elevated temperatures around 80°C and generate hazardous waste streams that are costly and difficult to remediate, making them increasingly untenable in a regulatory environment focused on green chemistry principles.
The Novel Approach
In stark contrast to these legacy methods, the novel approach disclosed in the patent utilizes a sophisticated bifunctional gold catalyst to drive the hydrophosphorylation reaction with exceptional efficiency and selectivity. This new catalytic system operates under mild conditions, typically between 25°C and 75°C, with an optimal temperature of 50°C, representing a drastic reduction in energy consumption compared to ruthenium or mercury protocols. The catalyst itself is designed with a specific ligand architecture that stabilizes the gold center and facilitates the activation of the alkyne substrate without the need for harsh promoters. As illustrated in the catalyst structures below, the system employs a biphenyl-phosphine ligand modified with an amide side chain, which plays a crucial role in enhancing the catalytic activity and stereocontrol. This structural innovation allows for the use of significantly lower catalyst loadings, ranging from 0.01 to 0.05 equivalents, thereby minimizing the residual metal content in the final product and simplifying downstream purification processes.
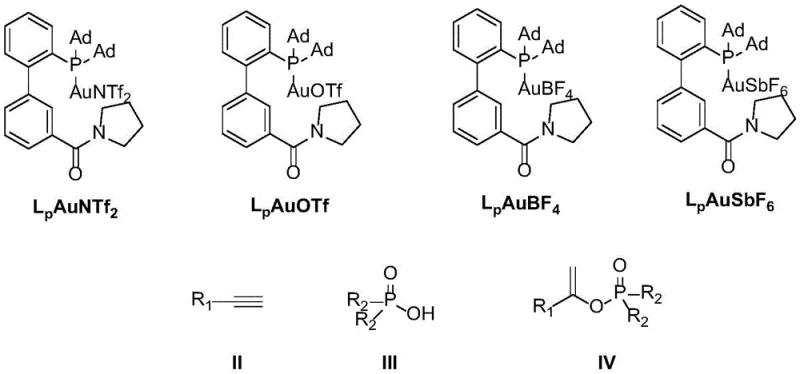
The versatility of this gold-catalyzed method is further evidenced by its broad substrate scope, accommodating a wide array of alkynes and phosphoric acid derivatives. Unlike previous methods that struggled with sterically hindered substrates or specific electronic environments, this system demonstrates robust performance across diverse chemical landscapes. The replacement of toxic mercury and expensive ruthenium with a low-toxicity gold catalyst not only aligns with global sustainability goals but also offers a tangible economic advantage by reducing the costs associated with hazardous material handling and waste treatment. For supply chain managers, this transition represents a de-risking of the production process, ensuring greater continuity and compliance with international safety standards.
Mechanistic Insights into Gold-Catalyzed Hydrophosphorylation
The success of this synthetic methodology hinges on the unique mechanistic pathway enabled by the bifunctional gold catalyst. The reaction proceeds through the activation of the alkyne triple bond by the cationic gold species, which acts as a soft Lewis acid to polarize the pi-system and render it susceptible to nucleophilic attack by the phosphoric acid. The specific design of the ligand, featuring bulky adamantyl groups and a hydrogen-bonding amide moiety, creates a tailored microenvironment around the gold center that directs the approach of the nucleophile. This spatial arrangement is critical for achieving the high stereoselectivity observed in the formation of the alkenyl phosphate products, favoring the generation of specific E or Z isomers depending on the substrate and reaction conditions. The mild reaction temperatures prevent side reactions such as polymerization or decomposition, which are common pitfalls in high-temperature olefin synthesis, thereby ensuring a cleaner reaction profile and higher isolated yields.
Furthermore, the mechanism benefits from the stability of the gold-phosphine complex, which resists decomposition under the reaction conditions, allowing for sustained catalytic turnover. The use of non-coordinating counterions, such as NTf2, OTf, BF4, or SbF6, further enhances the electrophilicity of the gold center, facilitating the activation of less reactive alkyne substrates. This mechanistic robustness translates directly to process reliability, as the reaction can be consistently reproduced with high fidelity across different batches. For R&D teams, understanding this mechanism provides a framework for further optimization, such as tuning the electronic properties of the phosphoric acid or modifying the alkyne substituents to access novel chemical space. The ability to control the stereochemistry of the resulting double bond is particularly valuable in medicinal chemistry, where the geometric configuration of an olefin can profoundly influence the biological activity and pharmacokinetic properties of the final drug candidate.
How to Synthesize Phosphoenyl Ester Efficiently
The practical implementation of this gold-catalyzed synthesis is straightforward and amenable to standard laboratory and pilot-plant equipment. The general procedure involves dissolving the gold catalyst, the alkyne substrate, and the phosphoric acid compound in a suitable organic solvent under an inert nitrogen atmosphere to prevent oxidation or moisture interference. The reaction mixture is then stirred in a Schlenk tube or similar vessel at a controlled temperature, typically 50°C, for a duration of 3 to 10 hours. Following the completion of the reaction, the workup process is simplified by the absence of toxic heavy metals; the solvent is removed under reduced pressure, and the crude product is purified via standard silica gel column chromatography. This operational simplicity reduces the technical barrier for adoption and facilitates rapid scale-up from gram to kilogram quantities.
- Dissolve the bifunctional gold catalyst (e.g., LpAuNTf2), alkyne substrate, and phosphoric acid compound in an organic solvent such as toluene or dichloroethane under a nitrogen atmosphere.
- Stir the reaction mixture in a Schlenk tube at a mild temperature range of 25-75°C (optimally 50°C) for 3 to 10 hours to ensure complete conversion.
- Perform post-treatment by removing the solvent under reduced pressure and purifying the crude product via silica gel column chromatography to isolate the high-purity alkenyl phosphate.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this gold-catalyzed technology offers compelling advantages that directly impact the bottom line and supply chain resilience. The elimination of mercury and the reduction of ruthenium usage address two of the most significant cost drivers in fine chemical manufacturing: hazardous waste disposal and precious metal recovery. By switching to a low-toxicity gold catalyst that operates at low loadings, manufacturers can drastically simplify their waste management protocols and reduce the regulatory burden associated with handling toxic substances. This shift not only lowers operational expenditures but also mitigates the risk of production stoppages due to environmental compliance issues. Furthermore, the mild reaction conditions reduce energy consumption, contributing to a smaller carbon footprint and aligning with corporate sustainability targets that are increasingly important to global partners.
- Cost Reduction in Manufacturing: The transition from expensive ruthenium or toxic mercury catalysts to a low-loading gold system results in substantial cost savings. The removal of heavy metal contaminants is often a multi-step, expensive process involving specialized scavengers; by using a cleaner gold catalyst, these downstream purification costs are significantly minimized. Additionally, the high atom economy and selectivity of the reaction reduce the formation of byproducts, leading to higher overall yields and better utilization of raw materials. The qualitative improvement in process efficiency means that less solvent and energy are required per unit of product, driving down the variable cost of goods sold and improving margin potential for high-volume production runs.
- Enhanced Supply Chain Reliability: The reliance on readily available starting materials and a robust catalytic system enhances the reliability of the supply chain. Unlike methods that require specialized, hard-to-source reagents or extreme conditions that strain equipment, this process uses common solvents like toluene and standard heating capabilities. This accessibility ensures that production can be maintained consistently without the risk of bottlenecks caused by reagent shortages or equipment failures. The stability of the catalyst and the simplicity of the operation also mean that the process is less prone to batch-to-batch variability, ensuring a steady supply of high-quality intermediates for downstream customers who depend on just-in-time delivery models.
- Scalability and Environmental Compliance: The mild nature of the reaction conditions makes this technology highly scalable from laboratory to commercial production. Operating at 50°C rather than 140°C reduces the thermal stress on reactors and allows for the use of standard glass-lined or stainless-steel equipment without the need for specialized high-temperature alloys. From an environmental standpoint, the absence of mercury eliminates the generation of persistent bio-accumulative toxins, simplifying the permitting process for new manufacturing lines. The reduced energy demand and safer waste profile position this method as a future-proof solution that meets the evolving regulatory landscape for green chemistry in the pharmaceutical and agrochemical sectors.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this gold-catalyzed synthesis. These answers are derived directly from the technical specifications and beneficial effects outlined in the patent documentation, providing clarity on the operational parameters and advantages of the technology. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this method into existing production workflows or for procurement officers assessing the quality and safety profile of the supplied intermediates.
Q: Why is the gold-catalyzed method superior to traditional mercury or ruthenium catalysis?
A: Traditional methods rely on highly toxic mercury salts or expensive ruthenium catalysts that require harsh conditions (up to 140°C). The patented gold-catalyzed method operates at mild temperatures (25-75°C), eliminates heavy metal toxicity risks, and offers superior stereoselectivity with lower catalyst loading.
Q: What represents the key innovation in the catalyst design for this synthesis?
A: The process utilizes a specialized bifunctional gold catalyst featuring a bulky biphenyl-phosphine ligand with an amide side chain. This unique structure enhances stability and selectivity, allowing for efficient activation of alkynes without the need for excessive heat or toxic promoters.
Q: Is this synthesis method suitable for large-scale commercial production?
A: Yes, the method is highly scalable due to its mild reaction conditions and simple workup procedures. The use of common organic solvents like toluene and the elimination of hazardous mercury waste streams significantly simplify the engineering requirements for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Phosphoenyl Ester Supplier
NINGBO INNO PHARMCHEM stands at the forefront of adopting advanced synthetic technologies to deliver superior chemical solutions to the global market. Our expertise in scaling diverse pathways from 100 kgs to 100 MT/annual commercial production ensures that we can meet the demanding volume requirements of multinational corporations while maintaining stringent purity specifications. We leverage our rigorous QC labs and deep process knowledge to guarantee that every batch of phosphoenyl ester produced via this gold-catalyzed route meets the highest standards of quality and consistency. Our commitment to innovation allows us to offer cutting-edge intermediates that empower our clients to accelerate their own drug discovery and development pipelines with confidence.
We invite potential partners to engage with our technical procurement team to discuss how this advanced synthesis method can optimize your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits of switching to this safer, more efficient protocol. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your project needs, ensuring a seamless integration of these high-value intermediates into your manufacturing operations.
