Advanced Catalytic Strategy for Scalable 3-Allyl-3-Hydroxyoxindole Production
The pharmaceutical and fine chemical industries constantly seek robust synthetic routes for high-value heterocyclic scaffolds, particularly those serving as precursors to bioactive natural products. Patent CN102070509B introduces a significant advancement in the preparation of 3-allyl-3-hydroxyoxindoles, a critical class of intermediates used in the total synthesis of alkaloids such as Convolutamydine, Flustraminol, and Donaxaridine. This technology leverages a versatile Lewis acid or Brønsted acid catalytic system to facilitate the nucleophilic addition of allylsilanes to isatins. Unlike legacy methods that often suffer from harsh conditions or narrow substrate scope, this invention provides a generalized platform capable of accommodating diverse functional groups including halogens, nitro groups, and alkyl substituents. For R&D directors and procurement specialists, understanding the nuances of this catalytic allylation is essential for optimizing supply chains and reducing the cost of goods for complex API intermediates.
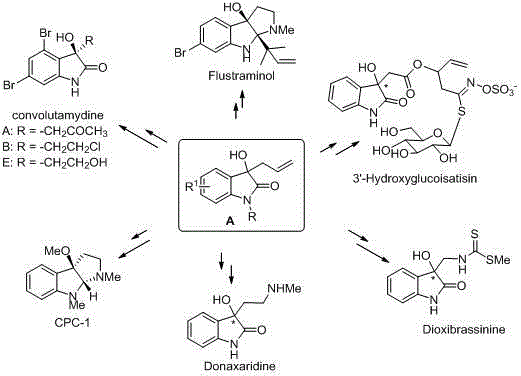
The strategic value of this patent lies in its ability to transform a traditionally difficult transformation into a operationally simple procedure. By utilizing commercially available allyltrimethylsilane and a wide array of metal salts ranging from mercury and indium to copper and iron, the method ensures that manufacturers are not locked into a single supply chain for exotic catalysts. This flexibility is paramount for maintaining continuity in the production of high-purity pharmaceutical intermediates. Furthermore, the reaction proceeds efficiently across a broad temperature window, from cryogenic conditions for sensitive substrates to elevated temperatures for sterically hindered systems, offering process chemists unprecedented control over reaction kinetics and selectivity.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 3-allyl-3-hydroxyoxindoles has been plagued by significant operational and economic hurdles that impede large-scale adoption. Traditional approaches, such as the Indium-mediated Barbier reaction, necessitate the use of stoichiometric amounts of indium powder, which is not only costly but also generates substantial metallic waste, complicating downstream purification and environmental compliance. Other methods relying on transition metal catalysis, such as palladium or iridium complexes, often demand rigorously anhydrous and oxygen-free environments, requiring expensive glovebox techniques or specialized Schlenk lines that increase capital expenditure and batch cycle times. Additionally, many prior art protocols exhibit poor functional group tolerance, failing to accommodate halogenated or nitro-substituted isatins without extensive protecting group manipulations, thereby lengthening the synthetic sequence and reducing overall atom economy.
The Novel Approach
The methodology disclosed in CN102070509B represents a paradigm shift by employing catalytic amounts of Lewis acids that are stable to air and moisture in many instances. This innovation eliminates the need for stoichiometric metal reductants and allows reactions to be conducted in common organic solvents like dichloromethane, THF, or even methanol without rigorous drying. The use of allyltrimethylsilane as a nucleophile is particularly advantageous due to its stability and ease of handling compared to reactive organometallic reagents like Grignard or organozinc species. This approach drastically simplifies the workup procedure; post-reaction processing involves standard aqueous acid quenching and extraction, avoiding the complex filtration steps required to remove bulk metal residues. Consequently, this novel route offers a direct path to cost reduction in pharmaceutical intermediate manufacturing by minimizing raw material costs and maximizing throughput efficiency.
Mechanistic Insights into Lewis Acid-Catalyzed Allylation
The core of this synthetic strategy relies on the activation of the electrophilic carbonyl group at the C3 position of the isatin ring by a Lewis acid catalyst. When a metal salt such as mercury(II) perchlorate or indium(III) triflate coordinates with the carbonyl oxygen, it significantly increases the electrophilicity of the carbon center, rendering it susceptible to nucleophilic attack by the soft allyl group of the silane reagent. This activation lowers the energy barrier for the addition step, allowing the reaction to proceed under mild thermal conditions. The subsequent desilylation and protonation steps yield the tertiary alcohol product with high regioselectivity. The versatility of the catalyst system allows for fine-tuning of the electronic environment; for instance, harder Lewis acids may be preferred for electron-deficient isatins, while softer metals might better accommodate sterically demanding substrates.
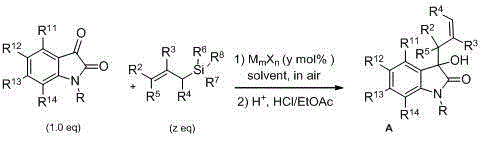
Furthermore, the patent elucidates the potential for asymmetric induction through the use of chiral ligands complexed with the Lewis acid center. By introducing chiral phosphines like BINAP or MOP, the catalyst creates a chiral pocket that differentiates the enantiotopic faces of the planar isatin carbonyl. This mechanistic feature is crucial for the synthesis of enantiomerically enriched intermediates required for modern drug development, where stereochemistry often dictates biological activity and safety profiles. The ability to access both racemic and chiral variants using the same fundamental reaction manifold provides a significant advantage in process development, allowing for rapid scale-up of screening hits without the need to develop entirely new synthetic routes for chiral targets. Impurity profiles are generally clean, with the major byproduct being hexamethyldisiloxane, which is easily removed during concentration or chromatography.
How to Synthesize 3-Allyl-3-Hydroxyoxindole Efficiently
The practical implementation of this chemistry is straightforward and amenable to standard laboratory and plant equipment. The general protocol involves charging a reaction vessel with the chosen catalyst, solvent, isatin substrate, and allyltrimethylsilane, followed by stirring at the optimized temperature until conversion is complete as monitored by TLC or HPLC. The reaction mixture is then quenched with dilute hydrochloric acid to decompose any silyl intermediates and protonate the alkoxide. Detailed standardized operating procedures regarding specific catalyst loadings, solvent volumes, and purification parameters are critical for ensuring batch-to-batch consistency and high purity specifications.
- Charge a reaction vessel with a Lewis acid catalyst (e.g., Hg(ClO4)2 or In(OTf)3), solvent, isatin substrate, and allyltrimethylsilane.
- Stir the mixture at a controlled temperature ranging from -78°C to 100°C depending on the catalyst activity until TLC indicates consumption of isatin.
- Quench the reaction with dilute hydrochloric acid, extract with ethyl acetate, wash with bicarbonate and brine, dry, and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a supply chain perspective, the adoption of this catalytic allylation method offers tangible benefits in terms of risk mitigation and operational efficiency. The reliance on commodity chemicals such as isatin and allyltrimethylsilane, both of which are produced globally in large quantities, ensures a stable supply base that is less susceptible to geopolitical disruptions or single-source bottlenecks. The robustness of the reaction conditions means that manufacturing can be performed in facilities with standard ventilation and safety systems, rather than requiring specialized inert atmosphere suites, thereby lowering the barrier to entry for contract manufacturing organizations and expanding the pool of potential suppliers.
- Cost Reduction in Manufacturing: The transition from stoichiometric metal reagents to catalytic systems fundamentally alters the cost structure of the synthesis. By reducing the catalyst loading to the mole percent level, the direct material cost per kilogram of product is significantly lowered. Moreover, the elimination of expensive protecting group steps and the simplification of the workup procedure reduce labor hours and solvent consumption. The ability to recycle solvents and the absence of heavy metal sludge disposal fees further contribute to substantial cost savings, making the final intermediate more price-competitive in the global market.
- Enhanced Supply Chain Reliability: The tolerance of the reaction to moisture and air for many catalyst variants reduces the risk of batch failures due to environmental excursions. This robustness translates to higher first-pass yields and more predictable delivery schedules. Since the method accommodates a wide range of substituted isatins, manufacturers can maintain a flexible inventory of raw materials and respond quickly to changes in demand for specific derivatives without requalifying entirely new processes. This agility is a critical asset in the fast-paced pharmaceutical sector where time-to-market is a key competitive differentiator.
- Scalability and Environmental Compliance: Scaling this reaction from gram to tonnage scale is facilitated by the exothermic nature of the reaction being manageable under the described conditions. The use of less toxic catalysts compared to alternatives like tin or lead reagents aligns with increasingly stringent environmental regulations regarding heavy metal residues in APIs. The simplified aqueous workup minimizes the generation of hazardous waste streams, supporting corporate sustainability goals and reducing the environmental footprint of the manufacturing process. This compliance advantage ensures long-term viability of the supply chain without the risk of regulatory shutdowns.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthetic technology. These insights are derived directly from the experimental data and claims within the patent documentation, providing a factual basis for decision-making. Understanding these details helps stakeholders evaluate the feasibility of integrating this method into their existing production portfolios.
Q: What are the advantages of this Lewis acid method over traditional Barbier reactions?
A: Unlike traditional Barbier reactions requiring stoichiometric indium powder and strict anhydrous conditions, this catalytic method uses low catalyst loading (mol%), tolerates moisture and air for many catalysts, and offers broader substrate compatibility including halogenated isatins.
Q: Can this method produce chiral 3-allyl-3-hydroxyoxindoles?
A: Yes, the patent describes the use of chiral ligands such as (S)-BINAP or (S)-MOP complexed with Lewis acids like mercury perchlorate to achieve enantioselective synthesis with moderate to good ee values.
Q: Is the process suitable for large-scale manufacturing?
A: The process is highly scalable due to the use of commercially available reagents, mild reaction conditions that do not require specialized inert atmosphere equipment for all variants, and simplified workup procedures involving standard aqueous washes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Allyl-3-Hydroxyoxindole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of drug development programs. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial manufacture is seamless. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 3-allyl-3-hydroxyoxindole meets the exacting standards required for downstream synthesis of complex bioactive molecules. Our commitment to quality assurance means that our clients can rely on consistent material performance, minimizing the risk of delays in their own clinical or commercial timelines.
We invite you to collaborate with us to leverage this advanced catalytic technology for your specific project needs. Our experts are ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements and purity targets. Please contact our technical procurement team to request specific COA data and route feasibility assessments. By partnering with us, you gain access to a supply chain that is not only cost-effective but also technically sophisticated, ensuring that your project remains on the cutting edge of pharmaceutical innovation.
