Advanced Rhodium-Catalyzed Synthesis of C2-Sulfonylamino Indole Derivatives for Commercial Scale-Up
Advanced Rhodium-Catalyzed Synthesis of C2-Sulfonylamino Indole Derivatives for Commercial Scale-Up
The pharmaceutical industry continuously seeks efficient pathways to access complex heterocyclic scaffolds, particularly indole derivatives which serve as privileged structures in numerous bioactive molecules. Patent CN112500393B introduces a groundbreaking methodology for the synthesis of C2-sulfonylamino indole derivatives, addressing critical bottlenecks in traditional synthetic routes. This innovation leverages a robust Rh(III)-catalyzed C-H activation strategy, enabling the direct coupling of N-pyridine-α-indole aryl alcohols with sulfonyl azides. By shifting away from stoichiometric strong bases and hazardous reagents, this technology offers a paradigm shift towards greener, more sustainable chemical manufacturing. For R&D directors and process chemists, this represents a vital opportunity to streamline the production of high-value pharmaceutical intermediates while maintaining rigorous purity standards essential for drug development pipelines.
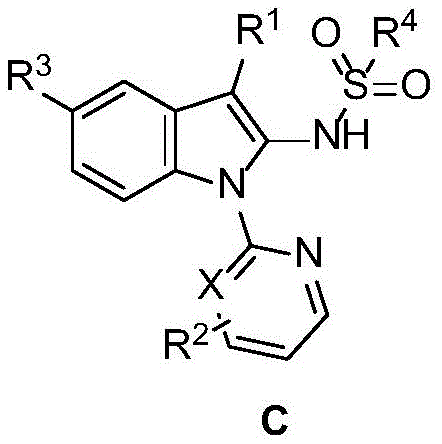
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of C2-aminated indole skeletons has been fraught with significant synthetic challenges that hinder large-scale adoption. Traditional approaches often rely on the use of aggressive strong bases, such as sodium hydride, to facilitate nucleophilic substitutions or eliminations, which poses severe safety risks in industrial settings due to hydrogen gas evolution and pyrophoric nature. Furthermore, existing methods frequently suffer from poor atom economy, requiring pre-functionalized starting materials that add unnecessary steps and cost to the overall process. Many prior art techniques also struggle with regioselectivity, leading to complex mixtures of isomers that are difficult and expensive to separate. These inefficiencies result in prolonged lead times and inflated manufacturing costs, making it difficult for procurement teams to secure reliable supplies of high-purity intermediates for clinical and commercial programs.
The Novel Approach
In stark contrast, the novel methodology disclosed in the patent utilizes a transition metal-catalyzed C-H functionalization strategy that fundamentally redefines the synthetic landscape for these compounds. By employing a Cp*Rh(III) catalyst, the process achieves direct activation of the inert C2-H bond on the indole ring, bypassing the need for pre-halogenation or other activating groups. This approach operates under remarkably mild conditions, typically at temperatures between 80-90°C, and exhibits impressive tolerance to air and moisture, eliminating the need for stringent inert atmosphere protocols. The use of sulfonyl azides as the nitrogen source is particularly advantageous, as the extrusion of nitrogen gas serves as a thermodynamic driving force, pushing the reaction to completion with high efficiency. This streamlined operation not only reduces the number of unit operations but also significantly lowers the environmental footprint of the manufacturing process.
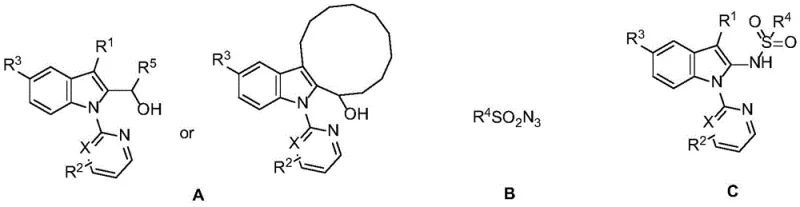
Mechanistic Insights into Rh(III)-Catalyzed C-H Activation
The core of this technological advancement lies in the sophisticated catalytic cycle mediated by the pentamethylcyclopentadienyl rhodium(III) complex. The mechanism initiates with the coordination of the pyridine or pyrimidine directing group on the indole substrate to the electrophilic Rh(III) center. This coordination facilitates the concerted metalation-deprotonation (CMD) process, selectively activating the proximal C2-H bond to form a stable five-membered rhodacycle intermediate. Subsequently, the sulfonyl azide undergoes coordination and subsequent nitrene transfer to the metal center, likely involving the extrusion of molecular nitrogen. This generates a high-valent Rh-nitrenoid species which then undergoes reductive elimination or insertion to forge the new C-N bond at the C2 position. The catalyst is finally regenerated through protonolysis or oxidation, ready to enter the next catalytic turnover. This precise mechanistic pathway ensures exceptional regioselectivity, minimizing the formation of unwanted byproducts and simplifying the downstream purification burden.
From an impurity control perspective, this catalytic system offers distinct advantages over radical-based or base-mediated alternatives. The directed nature of the C-H activation ensures that functionalization occurs exclusively at the C2 position, preventing random substitution on the benzene ring or the nitrogen atom. Furthermore, the mild reaction conditions prevent the decomposition of sensitive functional groups often present in complex drug candidates, such as esters, halides, or ethers. The patent data indicates that the process is highly robust, accommodating a wide range of electronic and steric environments on both the indole core and the sulfonyl azide partner. This versatility allows for the synthesis of diverse analogues with consistent quality, a critical factor for medicinal chemistry teams exploring structure-activity relationships (SAR) during lead optimization phases.
How to Synthesize C2-Sulfonylamino Indole Derivatives Efficiently
The practical implementation of this synthesis is designed for ease of execution in both laboratory and pilot plant environments. The protocol involves a straightforward one-pot procedure where the N-pyridine-α-indole aryl alcohol, sulfonyl azide, and the rhodium catalyst are combined in an organic solvent such as 1,2-dichloroethane. The reaction mixture is then heated to 85°C for a period ranging from 24 to 72 hours, depending on the specific substrate reactivity. Upon completion, the workup is minimal, requiring only the removal of the solvent followed by standard chromatographic purification. This simplicity translates directly into operational efficiency, reducing the demand for specialized equipment and highly trained personnel. Detailed standardized synthesis steps for specific high-yielding examples are provided in the guide below to assist technical teams in immediate replication.
- Mix N-pyridine-α-indole aryl alcohol compound, sulfonyl azide, and Cp*Rh(III) catalyst in anhydrous 1,2-dichloroethane.
- Heat the reaction mixture to 80-90°C (preferably 85°C) under argon atmosphere for 24-72 hours.
- Cool to room temperature, remove solvent under vacuum, and purify the crude product via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this Rh(III)-catalyzed technology presents a compelling value proposition centered on cost efficiency and supply reliability. The elimination of hazardous reagents like sodium hydride removes significant safety overheads and insurance costs associated with handling pyrophoric materials. Additionally, the use of readily available industrial raw materials, such as simple alcohols and commercially sourced sulfonyl azides, ensures a stable and resilient supply chain that is less susceptible to market volatility. The simplified purification process, which avoids complex crystallizations or distillations, drastically reduces solvent consumption and waste generation, aligning with modern green chemistry mandates and reducing disposal costs. These factors collectively contribute to a more sustainable and economically viable manufacturing model for high-value pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The transition to this catalytic method drives down production costs through multiple mechanisms. By utilizing a catalyst loading as low as 2.5-10%, the process minimizes the consumption of expensive precious metals while maintaining high turnover numbers. The avoidance of stoichiometric bases and the reduction in reaction steps eliminate the need for costly quenching procedures and extensive aqueous workups. Furthermore, the high selectivity of the reaction reduces the loss of valuable starting materials to side products, thereby improving the overall mass balance and yield efficiency. These cumulative savings allow for a more competitive pricing structure for the final API intermediate without compromising on quality.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route significantly mitigates supply chain risks. Since the reaction is insensitive to air and moisture, it does not require specialized glovebox facilities or rigorously dried solvents, making it easier to scale up in standard manufacturing plants. The broad substrate scope means that alternative raw materials can be sourced easily if supply disruptions occur, providing flexibility in vendor selection. Moreover, the simplified operational protocol reduces the likelihood of batch failures due to human error or equipment malfunction, ensuring consistent on-time delivery of critical intermediates to downstream customers.
- Scalability and Environmental Compliance: Scaling this process from gram to kilogram quantities is straightforward due to the absence of exothermic hazards associated with strong bases. The use of common organic solvents like dichloroethane facilitates easy solvent recovery and recycling, further enhancing the environmental profile of the process. The reduced generation of inorganic salts and hazardous waste streams simplifies compliance with increasingly stringent environmental regulations. This scalability ensures that the technology can seamlessly transition from early-stage drug discovery to commercial manufacturing, supporting the long-term growth of pharmaceutical projects.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this C2-sulfonylamino indole synthesis technology. These insights are derived directly from the experimental data and technical disclosures within the patent documentation, providing a clear understanding of the process capabilities and limitations. Understanding these details is crucial for project managers evaluating the feasibility of integrating this route into their existing production workflows.
Q: What are the key advantages of this Rh(III)-catalyzed method over traditional amination?
A: This method eliminates the need for harsh strong bases like sodium hydride and avoids complex multi-step sequences. It utilizes direct C-H activation at the C2 position, offering higher atom economy, milder reaction conditions (85°C), and insensitivity to air and moisture, which significantly simplifies operational safety and equipment requirements.
Q: What is the substrate scope for the sulfonyl azide component?
A: The process demonstrates excellent tolerance for diverse substituents on the sulfonyl azide. It successfully accommodates phenyl, substituted phenyl groups (including electron-donating methyl/methoxy and electron-withdrawing halogens/CF3), and heteroaromatic rings like thiophene, allowing for the rapid generation of diverse chemical libraries for SAR studies.
Q: How is the product purified after the reaction?
A: The workup procedure is remarkably simple. After the reaction completes, the solvent is removed via spin-drying under vacuum. The resulting crude material is then purified using standard silica gel column chromatography with a petroleum ether and ethyl acetate mixed solvent system, yielding high-purity products suitable for downstream applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable C2-Sulfonylamino Indole Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced C-H activation technologies in accelerating drug discovery and development. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project needs are met with precision and reliability. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications required for global regulatory submissions. We are committed to delivering high-purity C2-sulfonylamino indole derivatives that adhere to the highest quality standards, supporting your journey from preclinical research to market launch.
We invite you to collaborate with our technical team to explore how this innovative synthesis route can optimize your specific project requirements. Contact us today to request a Customized Cost-Saving Analysis tailored to your volume needs. Our experts are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate the tangible benefits of partnering with us. Let us help you secure a competitive edge in the pharmaceutical market through superior chemistry and supply chain excellence.
