Advanced Synthesis of 2-Iodo-3-Amino Naphthoquinones for Pharmaceutical Innovation
Advanced Synthesis of 2-Iodo-3-Amino Naphthoquinones for Pharmaceutical Innovation
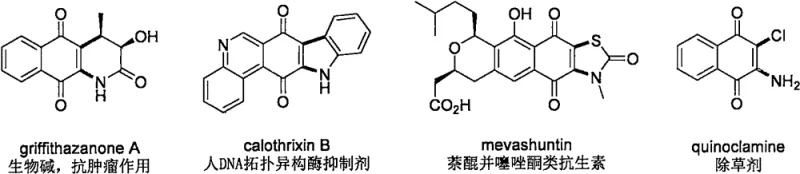
The landscape of pharmaceutical intermediate manufacturing is constantly evolving, driven by the need for more efficient, linear, and cost-effective synthetic routes for complex bioactive scaffolds. A significant breakthrough in this domain is documented in patent CN113372298B, which details a novel preparation method for 2-iodo-3-amino naphthoquinone compounds. This technology addresses a critical gap in organic synthesis, offering a robust pathway to access a core skeleton widely found in natural products with potent biological activities. These structures serve as the backbone for a variety of high-value therapeutic agents, ranging from antibiotics like mevashuntin to antitumor candidates such as griffithazanone A and human DNA topoisomerase inhibitors like calothrixin B. The ability to synthesize these motifs efficiently is paramount for accelerating drug discovery pipelines and securing the supply chain for next-generation medicines.
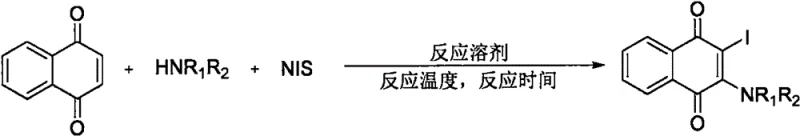
For R&D directors and process chemists, the introduction of this methodology represents a paradigm shift from traditional, step-intensive approaches to a streamlined, direct bifunctionalization strategy. The patent highlights that prior art methods often relied heavily on the manipulation of pre-functionalized synthetic building blocks, which inherently limited functional group compatibility and increased the number of synthetic steps. By contrast, the disclosed invention utilizes an in-situ generated iodoamination reagent derived from N-iodosuccinimide (NIS) and secondary amines. This radical series reaction not only simplifies the operational complexity but also enhances the overall atom economy and purity of the final product, making it an attractive candidate for commercial scale-up in the production of high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-amino-3-substituted naphthoquinones has been fraught with challenges that hinder efficient large-scale production. Conventional strategies typically depend on the use of pre-functionalized synthetic building blocks, which necessitates multiple protection and deprotection steps, thereby elongating the synthetic route and reducing overall yield. These traditional methods often suffer from poor functional group compatibility, meaning that sensitive moieties present in complex drug molecules might be degraded or altered during the harsh conditions required for naphthoquinone functionalization. Furthermore, the reliance on specialized starting materials increases the cost of goods sold (COGS) and introduces supply chain vulnerabilities, as these precursors may not be readily available from bulk chemical suppliers. The cumulative effect of these limitations is a bottleneck in the late-stage modification of drug molecules, restricting the ability of medicinal chemists to rapidly explore structure-activity relationships (SAR) around this privileged scaffold.
The Novel Approach
The innovative method described in patent CN113372298B overcomes these historical barriers through a direct, catalytic-free bifunctionalization of the naphthoquinone core. By employing N-iodosuccinimide and secondary amines as reaction raw materials in an organic solvent under nitrogen conditions, the process generates the target 2-iodo-3-amino naphthoquinone compounds via a radical series reaction. This approach eliminates the need for pre-functionalized blocks, effectively linearizing the synthesis and reducing the number of unit operations required. The reaction proceeds under remarkably mild conditions, specifically at room temperature (25°C) and completes within a mere 2 minutes, which is a testament to its kinetic efficiency. This drastic reduction in reaction time and thermal energy input translates directly into lower operational expenditures and higher reactor throughput, providing a compelling value proposition for procurement managers seeking cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into Radical-Mediated Iodoamination
From a mechanistic perspective, this transformation is driven by the in-situ generation of an electrophilic iodoamination species. The interaction between N-iodosuccinimide (NIS) and the secondary amine creates a reactive intermediate that facilitates the simultaneous introduction of iodine and amino groups across the naphthoquinone double bond. This radical series reaction pathway is highly selective, ensuring that the substitution occurs specifically at the 2 and 3 positions of the naphthoquinone ring system. The use of a radical mechanism rather than a transition-metal catalyzed process is particularly advantageous for pharmaceutical applications, as it avoids the potential contamination of the final product with heavy metal residues, thereby simplifying the purification process and ensuring compliance with stringent ICH Q3D guidelines for elemental impurities. The robustness of this mechanism is further evidenced by its tolerance to a wide range of secondary amines, including cyclic amines like morpholine and piperidine derivatives.
Impurity control is a critical aspect of this synthesis, especially given the reactivity of quinones. The mild reaction conditions play a pivotal role in minimizing side reactions such as over-oxidation or polymerization, which are common pitfalls in quinone chemistry. The patent data indicates that the reaction yields are consistently high, often exceeding 90% for various substrates, which suggests a clean reaction profile with minimal byproduct formation. The subsequent workup procedure, involving simple dilution with ethyl acetate and column chromatography, is sufficient to achieve high purity levels suitable for downstream coupling reactions. This high level of purity is essential for R&D teams who require reliable materials for biological testing and for supply chain heads who need to ensure batch-to-batch consistency in commercial production. The ability to achieve such purity without complex crystallization or extensive recrystallization steps further underscores the process efficiency.
How to Synthesize 2-Iodo-3-Amino Naphthoquinone Efficiently
The practical implementation of this synthesis route is designed for ease of operation, making it accessible for both laboratory-scale discovery and pilot-plant production. The standard protocol involves charging a reaction vessel with naphthoquinone, N-iodosuccinimide, and a selected secondary amine in a solvent such as acetonitrile. The mixture is then subjected to a nitrogen atmosphere to prevent oxidative degradation of the radical intermediates. Following a brief stirring period at ambient temperature, the reaction is quenched and worked up using standard extraction techniques. The detailed standardized synthesis steps, including specific molar ratios and purification parameters, are outlined in the guide below to ensure reproducibility and safety during scale-up operations.
- Combine naphthoquinone, N-iodosuccinimide (NIS), and a secondary amine in an organic solvent such as acetonitrile under a nitrogen atmosphere.
- Stir the reaction mixture at room temperature (25°C) for approximately 2 minutes to allow the radical series reaction to proceed via an in-situ generated iodoamination reagent.
- Dilute the reaction mixture with ethyl acetate, filter the organic phase, remove the solvent, and purify the crude product via column chromatography using petroleum ether and ethyl acetate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this synthetic methodology offers tangible strategic benefits beyond mere technical novelty. The shift towards a direct bifunctionalization route fundamentally alters the cost structure of producing these valuable intermediates. By utilizing commodity chemicals like naphthoquinone and NIS instead of specialized pre-functionalized blocks, the raw material costs are significantly reduced. Moreover, the elimination of transition metal catalysts removes the need for expensive scavenging resins and rigorous metal testing, which are often hidden cost drivers in API manufacturing. The simplified workflow also reduces the demand on labor and equipment time, allowing facilities to produce more batches within the same timeframe, thereby enhancing overall asset utilization and supply reliability.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the use of inexpensive, commercially available starting materials and the absence of precious metal catalysts. Traditional methods often require palladium or copper catalysts which not only add to the material cost but also necessitate costly removal steps to meet regulatory standards. By avoiding these metals entirely, the process achieves substantial cost savings in both raw material procurement and downstream purification. Additionally, the extremely short reaction time minimizes energy consumption for heating and cooling, contributing to a lower carbon footprint and reduced utility costs per kilogram of product produced.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the reliance on bulk chemicals that are widely sourced from multiple global suppliers. Naphthoquinone and common secondary amines like morpholine are produced at massive scales for various industries, ensuring that supply disruptions are unlikely to impact production schedules. The robustness of the reaction across different solvents, including acetonitrile and toluene, provides further flexibility; if one solvent faces availability issues, the process can be adapted to use alternatives without compromising yield. This adaptability is crucial for maintaining continuous supply lines for critical pharmaceutical intermediates in a volatile global market.
- Scalability and Environmental Compliance: Scaling this reaction from grams to tons is facilitated by its exothermic nature being easily managed at room temperature, removing the need for complex cryogenic or high-temperature reactors. The mild conditions reduce the risk of thermal runaway, a key safety consideration for large-scale operations. Furthermore, the high atom efficiency and minimal waste generation align with green chemistry principles, simplifying waste treatment and disposal. The ability to recycle solvents like ethyl acetate and acetonitrile further enhances the environmental profile, making it easier for manufacturers to comply with increasingly strict environmental regulations while maintaining high production volumes.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this naphthoquinone synthesis technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent literature, providing a clear understanding of the process capabilities and limitations for potential partners and licensees.
Q: What are the key advantages of this naphthoquinone synthesis method over traditional routes?
A: Unlike conventional methods that rely on cumbersome pre-functionalized synthetic building blocks with poor functional group compatibility, this novel approach utilizes a direct bifunctionalization strategy. It employs cheap and readily available raw materials like N-iodosuccinimide and secondary amines under mild conditions, resulting in significantly higher yields and purity while simplifying the synthetic linearity for late-stage drug modification.
Q: What are the optimal reaction conditions for scaling up this process?
A: The process operates efficiently at room temperature (25°C) with a remarkably short reaction time of just 2 minutes. While acetonitrile is the preferred solvent for optimal results, the reaction demonstrates robustness across various organic solvents including toluene and dichloroethane, offering flexibility for large-scale manufacturing and solvent recovery strategies.
Q: Can this method accommodate diverse amine substrates for library synthesis?
A: Yes, the method exhibits excellent substrate scope, successfully reacting with various secondary amines including morpholine, substituted piperidines, and protected piperazines. This versatility allows for the rapid generation of diverse 2-iodo-3-amino naphthoquinone derivatives, which are critical scaffolds for developing antibiotics, antitumor agents, and DNA topoisomerase inhibitors.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Iodo-3-Amino Naphthoquinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this advanced synthetic route for the pharmaceutical industry. As a leading CDMO partner, we possess the technical expertise and infrastructure to translate this laboratory-scale innovation into commercial reality. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project moves seamlessly from clinical trials to market launch. We are committed to delivering high-purity pharmaceutical intermediates that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify every batch.
We invite you to collaborate with us to leverage this cutting-edge technology for your drug development programs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our supply chain solutions can accelerate your timeline and optimize your manufacturing costs for complex naphthoquinone derivatives.
