Advanced Rhodium-Catalyzed Synthesis of Z-3-Vinyl Isoindolinones for Pharmaceutical Applications
Introduction to Patent CN115057808A: A Breakthrough in Stereoselective Synthesis
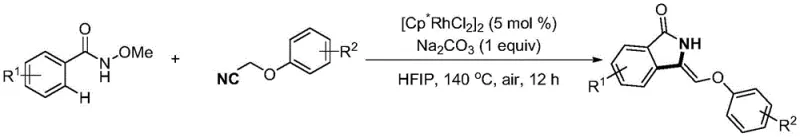
The pharmaceutical and fine chemical industries are constantly seeking more efficient pathways to access complex heterocyclic scaffolds, particularly those with specific stereochemical configurations that are difficult to obtain. Patent CN115057808A introduces a transformative synthetic methodology for preparing Z-3-vinyl substituted isoindolinone compounds, a structural motif prevalent in bioactive molecules and drug candidates. This innovation leverages a rhodium-catalyzed tandem cyclization reaction between N-methoxybenzamides and phenoxyacetonitriles, offering a distinct advantage over prior art by exclusively targeting the Z-isomer. The process is notable for its operational simplicity, utilizing air as the oxidant and eliminating the need for expensive ligands or Lewis acid additives. For R&D directors and procurement specialists, this represents a significant opportunity to streamline the supply chain for high-purity pharmaceutical intermediates while reducing the environmental footprint associated with traditional oxidative coupling reactions.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 3-vinyl substituted isoindolinones has been heavily biased towards the formation of the E-isomer due to its thermodynamic stability. Conventional transition metal-catalyzed methods often struggle to overcome this energetic preference, resulting in mixtures that require difficult and yield-loss-inducing separation processes. Furthermore, many existing protocols rely on harsh reaction conditions, including the use of stoichiometric amounts of toxic oxidants, specialized phosphine ligands, or moisture-sensitive Lewis acids. These requirements not only increase the cost of goods sold (COGS) but also complicate waste management and safety protocols in a manufacturing setting. The inability to selectively access the Z-configuration limits the chemical space available for medicinal chemists exploring structure-activity relationships (SAR), potentially hindering the development of novel therapeutics that rely on this specific geometric arrangement for biological activity.
The Novel Approach
The methodology disclosed in CN115057808A fundamentally shifts the paradigm by enabling the direct, one-step construction of the Z-3-vinyl isoindolinone skeleton with excellent stereoselectivity. By employing a [Cp*RhCl2]2 catalyst system in hexafluoroisopropanol (HFIP), the reaction proceeds through a unique mechanistic pathway that favors the kinetic Z-product. This approach bypasses the need for pre-functionalized substrates or multi-step sequences, thereby enhancing step economy. The use of simple, stable starting materials such as N-methoxybenzamides and phenoxyacetonitriles ensures a robust supply chain, while the reliance on air as the oxidant significantly reduces reagent costs and hazardous waste generation. This green chemistry approach aligns perfectly with modern sustainability goals in chemical manufacturing.
Mechanistic Insights into Rhodium-Catalyzed C-H Activation and Cyclization
The core of this technological advancement lies in the sophisticated interplay between the rhodium catalyst and the substrates. The reaction initiates with the coordination of the rhodium(III) species to the oxygen atom of the N-methoxybenzamide, acting as a weakly coordinating directing group. This facilitates the activation of the ortho C-H bond through a concerted metalation-deprotonation (CMD) mechanism, forming a stable five-membered rhodacycle intermediate. Unlike traditional cross-coupling reactions that might require pre-halogenated substrates, this C-H activation strategy directly functionalizes the inert carbon-hydrogen bond, representing a significant leap in atom economy. The subsequent nucleophilic attack of this organometallic intermediate onto the cyano group of the phenoxyacetonitrile is the key step that establishes the new carbon-carbon bond and sets the stage for ring closure.
Following the nucleophilic addition, the reaction undergoes a tandem cyclization and elimination sequence to release the final Z-3-vinyl isoindolinone product. The choice of HFIP as the solvent is critical; its strong hydrogen-bond donating ability likely stabilizes the transition states and intermediates, particularly the anionic species generated during the base-assisted deprotonation steps. Moreover, the specific steric environment created by the pentamethylcyclopentadienyl (Cp*) ligand on the rhodium center plays a pivotal role in enforcing the Z-stereoselectivity, preventing isomerization to the more stable E-form. This precise control over the reaction trajectory ensures high purity of the final product, minimizing the formation of regioisomeric or stereoisomeric impurities that are often challenging to remove in downstream processing.
How to Synthesize Z-3-Vinyl Isoindolinones Efficiently
To implement this synthesis in a laboratory or pilot plant setting, strict adherence to the optimized parameters is essential for reproducibility and yield maximization. The process involves mixing the N-methoxybenzamide derivative and the phenoxyacetonitrile coupling partner in a molar ratio of approximately 1:2, ensuring an excess of the nitrile substrate to drive the equilibrium forward. The catalyst loading is kept low at 5 mol%, demonstrating the high turnover number of the rhodium system, while sodium carbonate serves as a mild, inexpensive base to facilitate the C-H cleavage. Detailed standardized synthesis steps follow below to guide technical teams in replicating these results.
- Combine N-methoxybenzamide compound, phenoxyacetonitrile substrate, [Cp*RhCl2]2 catalyst (5 mol%), and sodium carbonate in hexafluoroisopropanol (HFIP) solvent.
- Heat the reaction mixture in an oil bath to 130-150°C under an air atmosphere and stir continuously for 12 hours to ensure complete conversion.
- Upon completion, cool the mixture, extract the organic phase, dry over sodium sulfate, and purify via silica gel column chromatography to isolate the Z-isomer product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this synthetic route offers compelling advantages that directly address the pain points of procurement managers and supply chain heads. The elimination of expensive and sensitive additives such as phosphine ligands or silver salts drastically simplifies the raw material sourcing strategy. Since the reaction utilizes air as the oxidant, there is no need to procure, store, or handle hazardous chemical oxidants, which reduces both inventory costs and safety compliance burdens. The robustness of the reaction conditions, specifically the tolerance for air and the use of a simple inorganic base, suggests that the process can be scaled up with minimal engineering modifications, ensuring a reliable supply of critical intermediates for drug development programs.
- Cost Reduction in Manufacturing: The economic viability of this process is underpinned by the use of commodity chemicals as starting materials. N-methoxybenzamides and phenoxyacetonitriles are readily available and inexpensive compared to halogenated precursors often used in palladium-catalyzed couplings. Furthermore, the removal of costly ligands and oxidants from the bill of materials leads to substantial savings in reagent costs. The simplified workup procedure, involving basic extraction and chromatography, reduces solvent consumption and labor time, contributing to a lower overall cost per kilogram of the active pharmaceutical ingredient (API) intermediate.
- Enhanced Supply Chain Reliability: Supply chain resilience is significantly improved by the stability and availability of the reactants. Unlike methods requiring freshly prepared organometallic reagents or unstable diazo compounds, the substrates here are shelf-stable solids or liquids that can be stocked in bulk without special storage conditions. The reaction's tolerance to air eliminates the need for rigorous inert gas purging systems (such as nitrogen or argon manifolds) in the production facility, allowing for more flexible scheduling and reduced downtime. This reliability ensures consistent delivery timelines for downstream customers who depend on these intermediates for their own synthesis campaigns.
- Scalability and Environmental Compliance: The green chemistry profile of this method facilitates easier regulatory approval and environmental compliance. By avoiding heavy metal oxidants and generating less hazardous waste, the process aligns with increasingly stringent environmental regulations governing chemical manufacturing. The use of HFIP, while a specialty solvent, allows for high efficiency which can offset its cost, and its recovery and reuse are feasible in a closed-loop system. The inherent safety of running the reaction under air at moderate temperatures (130-150°C) reduces the risk of thermal runaway incidents, making it a safer option for large-scale production compared to highly exothermic alternative routes.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this patented technology. These answers are derived directly from the experimental data and mechanistic understanding provided in the patent documentation, ensuring accuracy for technical decision-makers. Understanding these nuances is crucial for evaluating the feasibility of adopting this route for specific project needs.
Q: What distinguishes this synthesis method from conventional isoindolinone preparations?
A: Unlike traditional methods that predominantly yield the thermodynamically stable E-isomer, this rhodium-catalyzed protocol achieves high stereoselectivity for the Z-configuration. Furthermore, it operates without external oxidants, ligands, or Lewis acid additives, utilizing air as the terminal oxidant.
Q: What are the critical reaction conditions for optimal yield?
A: The process requires a specific combination of pentamethylcyclopentadienyl rhodium dichloride dimer as the catalyst and hexafluoroisopropanol (HFIP) as the solvent. The reaction must be maintained at 130-150°C for approximately 12 hours under an air atmosphere to drive the tandem cyclization effectively.
Q: Is this method scalable for industrial pharmaceutical intermediate production?
A: Yes, the method demonstrates excellent functional group compatibility and utilizes stable, commercially available substrates like N-methoxybenzamides and phenoxyacetonitriles. The absence of sensitive additives and the use of air oxidation simplify the engineering requirements for scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Z-3-Vinyl Isoindolinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of advanced synthetic methodologies like the one described in CN115057808A for accelerating drug discovery and development. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop to plant is seamless. Our facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of Z-3-vinyl isoindolinone intermediate meets the highest quality standards required by global regulatory bodies. We are committed to leveraging our technical expertise to optimize this rhodium-catalyzed process for your specific volume requirements.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the potential economic benefits tailored to your project scale. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing us to demonstrate our capability as your trusted partner in delivering high-performance pharmaceutical intermediates.
