Revolutionizing Biaryl Synthesis with Stable Meta-Carborane Iron Complexes for Commercial Scale Production
Revolutionizing Biaryl Synthesis with Stable Meta-Carborane Iron Complexes for Commercial Scale Production
The landscape of fine chemical synthesis is undergoing a significant transformation driven by the urgent need for sustainable, cost-effective, and scalable catalytic technologies. A groundbreaking development detailed in patent CN114716484A introduces a novel trivalent iron complex containing a meta-carborane methylpyridine structure, which represents a paradigm shift in the construction of pyrazine-thiazole biaryl scaffolds. These scaffolds are critical motifs found in numerous pharmaceutical agents exhibiting anti-inflammatory, antibacterial, and anticancer activities. The innovation lies not merely in the molecular architecture of the catalyst itself, but in its ability to facilitate oxidative coupling reactions under exceptionally mild conditions, specifically at room temperature and utilizing atmospheric oxygen as the terminal oxidant. This eliminates the reliance on expensive noble metals and energy-intensive heating protocols that have historically plagued this sector. For global procurement and R&D teams, this technology offers a tangible pathway to decarbonize synthetic routes while simultaneously enhancing the economic viability of producing high-value heterocyclic intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of pyrazine-thiazole biaryl compounds has been dominated by methodologies relying on palladium catalysis or stoichiometric metal organic bases, both of which present substantial logistical and financial burdens for large-scale manufacturing. Palladium, while effective, is a precious metal subject to volatile market pricing and supply chain constraints, making cost forecasting difficult for long-term projects. Furthermore, conventional palladium-catalyzed cross-couplings often necessitate rigorous exclusion of air and moisture, requiring specialized equipment and inert gas infrastructure that increases capital expenditure. Even recent advancements using simpler iron salts like FeCl3 have fallen short of ideal efficiency, typically requiring high-temperature reflux conditions to drive the reaction to completion. These elevated temperatures not only consume excessive energy but also promote the formation of thermal degradation byproducts and impurities, complicating downstream purification and reducing overall yield to approximately 60%. Such inefficiencies create bottlenecks in production schedules and inflate the cost of goods sold, rendering many potential drug candidates economically unfeasible to develop.
The Novel Approach
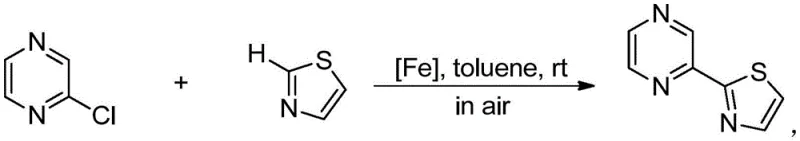
In stark contrast to these legacy methods, the novel approach utilizing the meta-carborane methylpyridine iron complex achieves high-efficiency catalysis under ambient conditions, fundamentally altering the process economics. The unique ligand environment provided by the meta-carborane cage stabilizes the iron center, allowing it to activate substrates effectively without the need for external heating. This capability enables the reaction to proceed at room temperature in an open vessel, leveraging air as a free and abundant oxidant rather than expensive chemical oxidants. The result is a streamlined process that drastically simplifies operational complexity and reduces utility consumption. As illustrated in the reaction scheme below, the coupling of halogenated pyrazines with thiazoles proceeds smoothly to afford the desired biaryl products with impressive yields ranging from 86% to 95%. This leap in performance demonstrates that the new catalyst overcomes the kinetic barriers that previously necessitated harsh conditions, offering a robust solution for the synthesis of diverse heterocyclic libraries.
Mechanistic Insights into FeCl3-Catalyzed Oxidative Coupling
The exceptional performance of this catalyst can be attributed to the sophisticated interplay between the electron-deficient carborane cluster and the nitrogen-donor pyridine ligands, which creates a highly stable yet reactive coordination sphere around the iron center. The meta-carborane moiety acts as a robust structural anchor, imparting significant thermal and chemical stability to the complex, thereby preventing catalyst decomposition during the reaction cycle. Mechanistically, the iron center likely facilitates a single-electron transfer (SET) process that activates the carbon-halogen bond of the pyrazine substrate, generating a radical intermediate that subsequently couples with the thiazole ring. The presence of the bulky carborane ligand may also play a crucial role in modulating the steric environment, preventing the aggregation of iron species into inactive clusters, which is a common deactivation pathway for simple iron salts. This stabilization ensures that the catalytic turnover number remains high throughout the reaction duration, allowing for the use of extremely low catalyst loadings relative to the substrate.
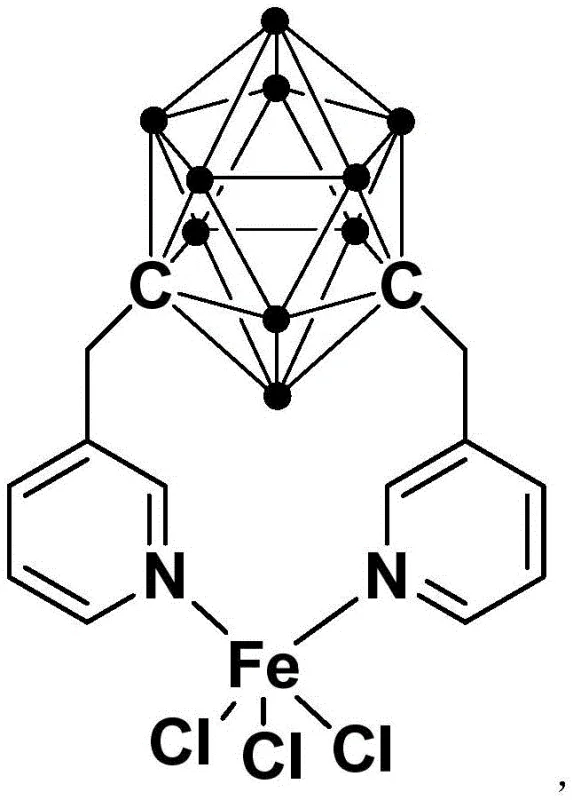
Furthermore, the mild reaction conditions inherent to this mechanistic pathway provide superior control over the impurity profile of the final product. In traditional high-temperature processes, thermal stress often leads to non-selective side reactions, such as homocoupling of the starting materials or decomposition of sensitive functional groups. By operating at room temperature, the new iron complex minimizes these thermal degradation pathways, resulting in a cleaner crude reaction mixture. This purity advantage translates directly into reduced burden on the purification team, as fewer chromatographic steps or recrystallizations are required to meet stringent pharmaceutical specifications. The ability to tolerate a wide range of electronic and steric variations in the substrate further underscores the versatility of this catalytic system, making it a powerful tool for medicinal chemists exploring structure-activity relationships without being constrained by synthetic limitations.
How to Synthesize Meta-Carborane Methylpyridine Iron Complex Efficiently
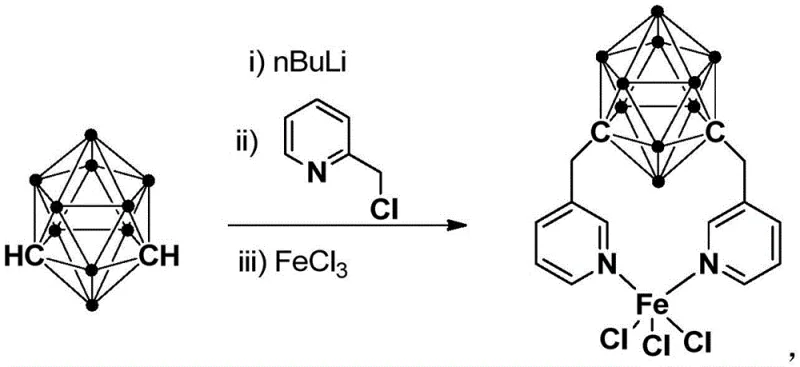
The preparation of this high-performance catalyst is designed for scalability and reproducibility, utilizing readily available starting materials and straightforward unit operations. The synthesis follows a logical three-step sequence involving lithiation, alkylation, and metallation, all of which can be performed in standard glass-lined reactors commonly found in fine chemical plants. The process begins with the controlled addition of n-butyllithium to a solution of meta-carborane at cryogenic temperatures to ensure selective deprotonation, followed by warming to ambient temperature to complete the activation. Subsequent addition of chloromethylpyridine allows for the formation of the carborane-pyridine ligand precursor, which is then immediately complexed with ferric chloride to generate the final active species. The entire protocol avoids the use of exotic reagents or hazardous high-pressure conditions, aligning perfectly with green chemistry principles and safety regulations.
- Lithiation of meta-carborane using n-BuLi at low temperature (-80°C to -75°C) followed by warming to room temperature.
- Alkylation by adding chloromethylpyridine to the lithiated carborane solution and reacting at room temperature for 6-8 hours.
- Metallation by adding FeCl3 to the mixture, reacting at room temperature for 3-5 hours, followed by filtration and column chromatography purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this meta-carborane iron complex technology offers compelling strategic advantages that extend far beyond simple reaction yield improvements. The shift from precious metal catalysts to an iron-based system fundamentally alters the cost structure of the manufacturing process, removing exposure to the volatile pricing of palladium and rhodium. Additionally, the elimination of high-temperature reflux requirements leads to a drastic reduction in energy consumption, lowering the utility footprint of the production facility. The use of air as an oxidant removes the need for purchasing, storing, and handling hazardous chemical oxidants, thereby simplifying inventory management and reducing safety compliance costs. These factors combine to create a leaner, more resilient supply chain that is less susceptible to external market shocks and regulatory changes regarding heavy metal residues in final drug products.
- Cost Reduction in Manufacturing: The transition to an iron-based catalyst system delivers substantial cost savings by replacing expensive noble metals with abundant and inexpensive iron salts. The process eliminates the need for costly ligand synthesis associated with palladium systems and removes the requirement for specialized scavengers to remove trace metal impurities from the final API. Furthermore, the ability to run reactions at room temperature significantly lowers energy bills associated with heating and cooling cycles, while the simplified workup procedure reduces solvent consumption and labor hours. These cumulative efficiencies result in a lower cost of goods sold, providing a competitive margin advantage in the marketplace.
- Enhanced Supply Chain Reliability: Relying on iron and basic organic building blocks like carboranes and chloromethylpyridines ensures a stable and secure supply of raw materials, as these commodities are not subject to the geopolitical constraints often affecting platinum group metals. The robustness of the catalyst, which is insensitive to air and moisture, simplifies logistics and warehousing requirements, allowing for bulk purchasing and long-term storage without degradation. This stability ensures consistent production schedules and reduces the risk of batch failures due to catalyst sensitivity, thereby guaranteeing reliable delivery timelines to downstream customers.
- Scalability and Environmental Compliance: The green nature of this synthesis, characterized by atom economy and the use of benign oxidants, positions manufacturers favorably against increasingly stringent environmental regulations. The absence of heavy metal waste streams simplifies effluent treatment and disposal, reducing environmental compliance costs. Moreover, the mild reaction conditions and high tolerance for substrate variation make the process highly scalable from kilogram to multi-ton production without the need for extensive re-optimization, facilitating rapid technology transfer from R&D to commercial manufacturing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this catalytic technology in industrial settings. These insights are derived directly from the experimental data and beneficial effects reported in the patent literature, providing a factual basis for decision-making. Understanding these details is crucial for evaluating the feasibility of integrating this new catalyst into existing production lines or new product development pipelines.
Q: What are the primary advantages of this iron complex over palladium catalysts?
A: Unlike traditional palladium catalysts which are expensive and often require harsh conditions, this iron complex utilizes abundant iron, operates at room temperature, and uses air as a green oxidant, significantly reducing raw material costs and environmental impact.
Q: Is the catalyst stable under industrial storage conditions?
A: Yes, the patent data indicates that the meta-carborane methylpyridine iron complex possesses high thermal stability and is insensitive to air and water, making it robust for long-term storage and handling in standard warehouse environments without special inert atmosphere requirements.
Q: What is the substrate scope for this catalytic system?
A: The catalyst demonstrates broad substrate versatility, effectively catalyzing the oxidative coupling of various halogenated pyrazines with thiazoles, accommodating different electronic effects and steric hindrance while maintaining high yields between 86% and 95%.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Meta-Carborane Iron Complex Supplier
As the demand for sustainable and efficient catalytic solutions continues to grow, NINGBO INNO PHARMCHEM stands ready to support your organization with high-quality meta-carborane iron complexes and related pharmaceutical intermediates. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from laboratory discovery to full-scale manufacturing is seamless and risk-free. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of catalyst meets the exacting standards required for GMP-compliant API synthesis. Our commitment to quality and consistency makes us a trusted partner for global enterprises seeking to optimize their synthetic routes.
We invite you to engage with our technical procurement team to discuss how this innovative iron complex can be tailored to your specific process needs. By requesting a Customized Cost-Saving Analysis, you can quantify the potential economic benefits of switching to this greener catalytic system for your specific application. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that drive value and efficiency in your chemical manufacturing operations.
