Advanced Synthesis of 8-Alkoxyquinolone Intermediates via Novel Boron Chelation Technology
Advanced Synthesis of 8-Alkoxyquinolone Intermediates via Novel Boron Chelation Technology
The pharmaceutical industry continuously seeks robust synthetic pathways for fluoroquinolone antibiotics, particularly those featuring sensitive 8-alkoxy substituents which enhance biological activity but pose significant synthetic challenges. Patent CN1031795C introduces a groundbreaking methodology for preparing (6,7-substituted-8-alkoxy-1-cyclopropyl-1,4-dihydro-4-oxo-3-quinolinecarboxylic acid-O3,O4) bis(acyloxy-O)borates. This technology represents a paradigm shift in pharmaceutical intermediates manufacturing by replacing hazardous fluoroboric acid protocols with a safer, more efficient boron chelation strategy. For R&D directors and procurement specialists, this innovation offers a compelling solution to long-standing issues regarding yield, purity, and equipment corrosion, positioning it as a critical asset for reliable quinolone intermediate supplier networks aiming to optimize their supply chains.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 8-alkoxyquinolone derivatives has been plagued by significant chemical and engineering hurdles. As detailed in prior art such as Japanese Patent Publication Show 62-252772, the presence of an electron-donating alkoxy group at the 8-position drastically reduces the reactivity of the adjacent 7-position. Consequently, mild reaction conditions fail to drive the substitution to completion, resulting in negligible yields of the desired derivative. Conversely, attempting to force the reaction through severe conditions often triggers the dealkylation of the sensitive 8-methoxy or 8-ethoxy groups, converting them into hydroxyl groups and ruining the product profile. Furthermore, alternative methods utilizing fluoroboric acid chelation, while effective in yield, introduce severe operational liabilities.
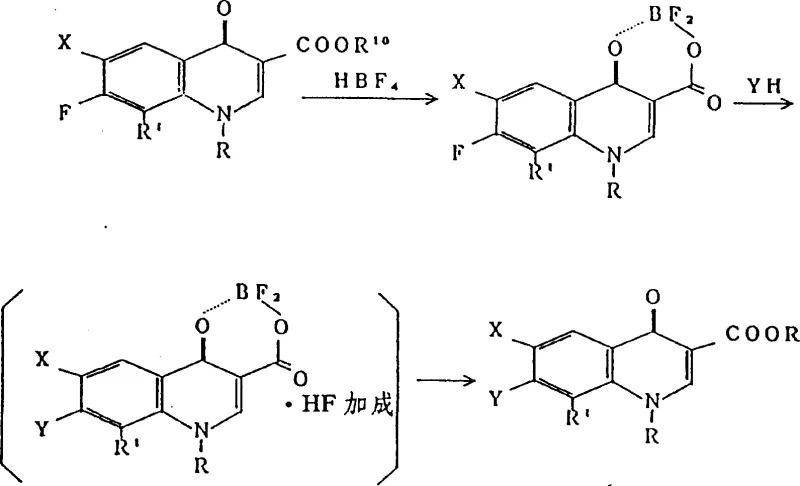
The reliance on fluoroboric acid creates a cascade of downstream problems for cost reduction in pharmaceutical manufacturing. The generation of hydrogen fluoride (HF) as a stoichiometric byproduct necessitates the use of exotic, corrosion-resistant reactor materials like Hastelloy, which exponentially increases capital expenditure. Moreover, the handling of HF requires rigorous safety protocols and waste treatment systems, adding substantial operational overhead. These factors collectively render traditional fluoroboric acid routes economically unviable for large-scale commercial scale-up of complex pharmaceutical intermediates, driving the urgent need for the safer alternative presented in the patent data.
The Novel Approach
The patented methodology circumvents these pitfalls by employing a novel di(acyloxy-O)borate chelating agent. Instead of relying on hazardous fluorine sources, this approach utilizes triacyloxyborate derivatives formed in situ from boric acid and organic anhydrides. This strategic shift eliminates the formation of corrosive hydrogen fluoride entirely, allowing the synthesis to proceed in standard stainless steel equipment without risk of reactor degradation. The resulting boron complex acts as a robust protective group, stabilizing the quinolone nucleus against thermal decomposition and unwanted side reactions during the critical 7-position substitution step.
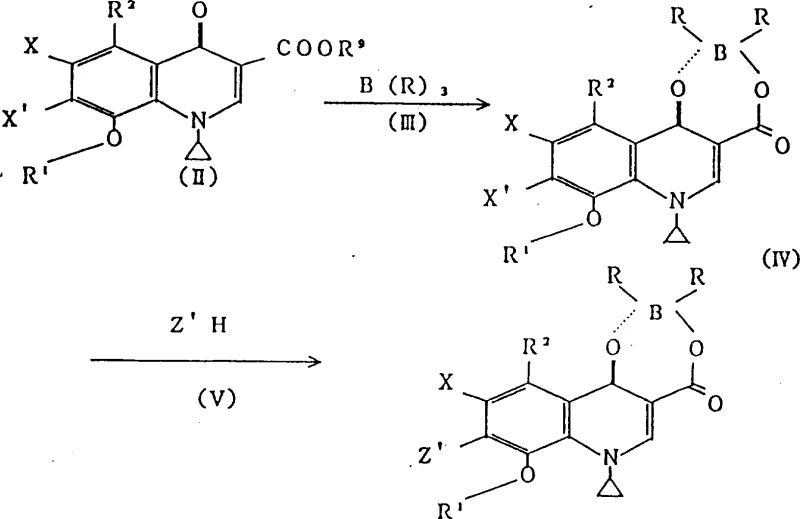
By integrating this boron chelation strategy, manufacturers can achieve high yields and exceptional purity levels previously unattainable with conventional methods. The process facilitates the smooth introduction of diverse cyclic amines at the 7-position while preserving the integrity of the 8-alkoxy moiety. This capability is essential for producing high-purity quinolone intermediates required for next-generation antibiotics. The versatility of the method allows for the use of various acyl groups and amine substrates, providing a flexible platform for synthesizing a wide array of analogues, thereby enhancing the strategic value for any reliable quinolone intermediate supplier looking to diversify their portfolio.
Mechanistic Insights into Boron-Chelated Cyclization
The core of this technological advancement lies in the formation and reactivity of the boron-chelated intermediate. The mechanism initiates with the reaction of the 8-alkoxyquinolone carboxylic acid ester with a triacyloxyborate derivative in an organic acid solvent, such as acetic or propionic acid. Under controlled temperatures ranging from 20°C to 200°C, the boron atom coordinates with the 4-oxo and 3-carboxyl oxygen atoms, forming a stable five-membered chelate ring. This coordination significantly alters the electronic distribution of the quinolone ring system, effectively activating the 7-position for nucleophilic attack while simultaneously shielding the sensitive 8-alkoxy group from acidic cleavage.
Following chelation, the subsequent nucleophilic substitution with cyclic amines proceeds with remarkable efficiency. The boron complex serves not merely as a protecting group but as an electronic modulator that enhances the leaving group ability of the halogen at the 7-position. Whether conducted in polar solvents like DMF or acetonitrile, or under solvent-free conditions, the reaction maintains high fidelity. The steric and electronic environment created by the boron ligand ensures that the incoming amine attacks exclusively at the 7-position, minimizing regio-isomeric impurities. This precise control over the reaction trajectory is paramount for meeting the stringent impurity specifications demanded by global regulatory bodies for API production.
How to Synthesize 8-Alkoxyquinolone Boron Complex Efficiently
The practical implementation of this synthesis involves a streamlined two-step sequence that balances reaction kinetics with operational safety. Initially, the triacyloxyborate reagent is generated by reacting boric acid with an organic acid anhydride in the presence of a catalyst like zinc chloride. This reagent is then introduced to the quinolone substrate in a heated organic acid medium to form the chelated intermediate. Subsequently, the isolated or crude chelate is reacted with the desired cyclic amine in the presence of a base such as triethylamine.
- React the 8-alkoxyquinolone precursor with a triacyloxyborate derivative in an organic acid solvent at elevated temperatures to form the boron chelate intermediate.
- Perform nucleophilic substitution by reacting the boron chelate intermediate with a cyclic amino compound in a polar solvent or under solvent-free conditions.
- Isolate the final product through filtration, washing, and drying, optionally followed by hydrolysis to remove the boron protecting group if necessary.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this boron-complex mediated synthesis offers transformative economic and logistical benefits. The primary advantage stems from the complete elimination of fluoroboric acid and the associated hydrogen fluoride byproduct. This change drastically simplifies the engineering requirements for production facilities, removing the need for specialized corrosion-resistant alloys and complex scrubbing systems designed for HF containment. Consequently, the barrier to entry for manufacturing these high-value intermediates is significantly lowered, fostering a more competitive and resilient supply market.
- Cost Reduction in Manufacturing: The replacement of expensive fluoroboric acid with readily available boric acid and organic anhydrides results in substantial raw material cost savings. Furthermore, the ability to utilize standard glass-lined or stainless steel reactors instead of exotic nickel-based alloys reduces capital depreciation and maintenance costs. The simplified waste treatment process, devoid of hazardous fluoride streams, further lowers the operational expenditure, making the overall cost structure much more favorable for long-term contracts.
- Enhanced Supply Chain Reliability: By mitigating the risks associated with handling highly corrosive and toxic HF, the process enhances overall plant safety and operational continuity. Reduced equipment corrosion leads to longer campaign runs and fewer unplanned shutdowns for maintenance, ensuring a steady flow of materials. The robustness of the boron chelate intermediate also allows for potential isolation and storage, providing flexibility in production scheduling and inventory management to buffer against market fluctuations.
- Scalability and Environmental Compliance: The process demonstrates excellent scalability, with reaction conditions that are easily transferable from pilot to commercial scale. The absence of persistent fluorinated waste streams aligns perfectly with modern green chemistry principles and increasingly strict environmental regulations. This compliance reduces the regulatory burden and potential liability for manufacturers, securing the long-term viability of the supply chain for commercial scale-up of complex pharmaceutical intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this boron chelation technology. Understanding these details is crucial for stakeholders evaluating the feasibility of adopting this route for their specific product pipelines.
Q: Why is the boron complex method superior to traditional fluoroboric acid methods?
A: Traditional methods using fluoroboric acid generate hazardous hydrogen fluoride (HF) as a byproduct, requiring specialized corrosion-resistant equipment. The novel boron complex method eliminates HF generation, allowing the use of standard industrial reactors and significantly reducing safety risks and equipment costs.
Q: What are the key purity advantages of this synthesis route?
A: The boron chelation stabilizes the quinolone nucleus during the harsh nucleophilic substitution step, preventing unwanted dealkylation of the sensitive 8-alkoxy group. This results in a cleaner impurity profile and higher overall yield compared to direct substitution methods.
Q: Can this process be scaled for commercial API production?
A: Yes, the process utilizes common organic solvents like acetic acid and acetonitrile and operates at moderate temperatures (20-200°C). The absence of corrosive HF and the use of stable intermediates make it highly suitable for large-scale commercial manufacturing from kilograms to metric tons.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 8-Alkoxyquinolone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of advanced synthetic methodologies in delivering high-quality pharmaceutical ingredients. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the innovative boron chelation process described in patent CN1031795C can be seamlessly integrated into your supply chain. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 8-alkoxyquinolone intermediate meets the exacting standards required for global API registration.
We invite you to collaborate with us to leverage this cost-effective and safe technology for your fluoroquinolone projects. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our expertise can drive efficiency and reliability in your manufacturing operations.
