Revolutionizing Chiral Amine Production: Catalytic Asymmetric Reduction Technology for Commercial Scale
Revolutionizing Chiral Amine Production: Catalytic Asymmetric Reduction Technology for Commercial Scale
The landscape of chiral amine synthesis has undergone a significant transformation with the disclosure of patent CN101547890A, which introduces a robust method for the catalytic asymmetric synthesis of primary amines via borane reduction of oxime ethers. This technology addresses a long-standing challenge in the pharmaceutical industry: the efficient production of enantiopure primary amines without the prohibitive costs associated with stoichiometric chiral auxiliaries. By utilizing stable chiral spiroboronic esters derived from non-racemic 1,2-amino alcohols, this process achieves complete conversion and exceptional enantioselectivity, reaching up to 99% ee in optimized conditions. For R&D directors and process chemists, this represents a pivotal shift from labor-intensive resolution methods to direct, catalytic asymmetric synthesis, enabling faster route scouting and more sustainable manufacturing pathways for complex drug intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
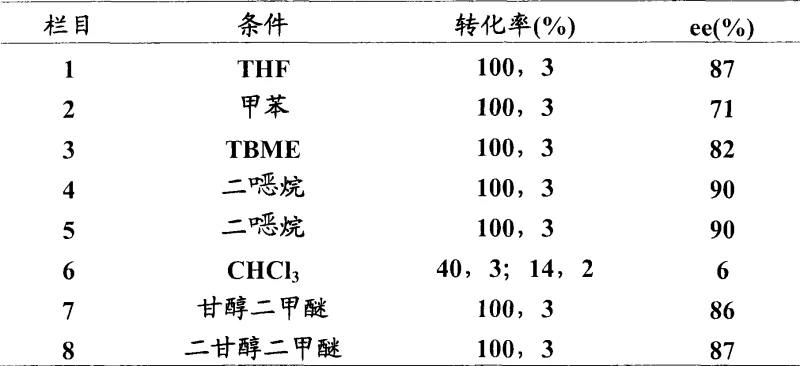
Historically, the asymmetric reduction of oxime ethers to prepare chiral amines has relied heavily on chiral catalysts such as 1,3,2-oxazaborolidines. However, these conventional methods suffer from significant inefficiencies that hinder large-scale adoption. A major drawback is the requirement for stoichiometric or near-stoichiometric amounts of expensive chiral amino alcohols to generate the active reducing species in situ. For instance, prior art methods often necessitated 2.5 equivalents of (S)-diphenylvalinol to achieve high selectivity, which drastically inflates the raw material costs and complicates the downstream purification process due to the presence of excess chiral reagents. Furthermore, many existing catalytic protocols fail to drive the reaction to completion, often resulting in mixtures containing unreacted O-benzyloxyamine intermediates, which compromises the overall yield and purity profile required for GMP manufacturing of active pharmaceutical ingredients.
The Novel Approach
The novel approach detailed in the patent overcomes these barriers by employing pre-formed, stable spiroboronic acid esters as true catalysts. Unlike transient species generated in situ, these spiroboronates can be isolated, stored for extended periods under inert conditions, and used at significantly lower loadings, typically around 10 mol%. This transition from stoichiometric reagents to genuine catalysis fundamentally alters the economic equation of chiral amine production. The method utilizes readily available borane sources like BH3·DMS or BH3·THF in common solvents such as dioxane or toluene. Optimization studies revealed that by fine-tuning the temperature and borane equivalents, the process achieves quantitative conversion of the oxime ether to the primary amine. This eliminates the need for difficult separations of partially reduced byproducts and ensures a cleaner reaction profile, making it highly attractive for cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into Spiroboronate-Catalyzed Borane Reduction
The core of this technological advancement lies in the unique structural properties of the chiral spiroboronic esters, specifically those derived from enantiopure 1,2-amino alcohols like diphenyl valinol. These catalysts function by forming a rigid chiral environment around the boron center, which facilitates the highly stereoselective transfer of hydride to the prochiral oxime ether substrate. The spirocyclic framework prevents racemization and ensures that the reduction proceeds through a well-defined transition state, leading to the preferential formation of one enantiomer over the other. Structural analysis indicates that the steric bulk of the phenyl groups on the catalyst backbone plays a crucial role in discriminating between the enantiotopic faces of the imine double bond. This precise spatial arrangement allows for excellent enantiocontrol even at elevated temperatures, providing a level of robustness that is often lacking in more sensitive organometallic catalytic systems.
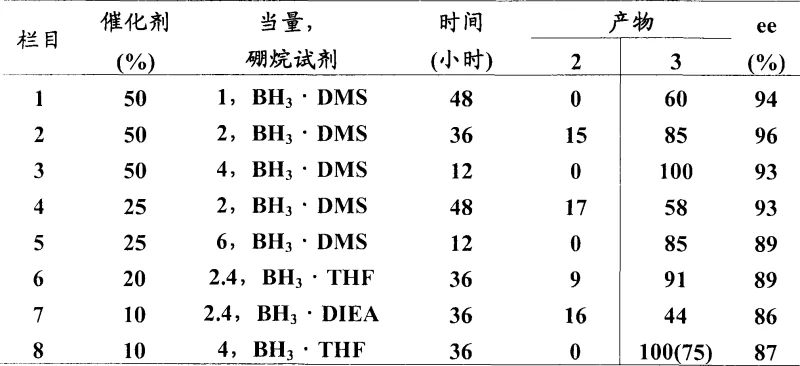
From an impurity control perspective, the mechanism ensures high chemical fidelity by promoting complete reduction of the N-O bond. In conventional reductions, incomplete conversion often leaves behind hydroxylamine derivatives which are structurally similar to the product and difficult to remove. The optimized catalytic cycle described here drives the reaction to full consumption of the starting material, particularly when using activated borane sources like BH3·THF in ether solvents. The data suggests that the electronic nature of the oxime ether substituent also influences the reaction kinetics; for example, electron-withdrawing groups on the benzyl moiety can enhance enantioselectivity, likely by increasing the electrophilicity of the imine carbon. This mechanistic understanding allows process chemists to predict outcomes for new substrates and tailor reaction conditions to minimize genotoxic impurities or heavy metal residues, aligning perfectly with stringent regulatory requirements for API synthesis.
How to Synthesize Enantiopure Primary Amines Efficiently
The synthesis protocol outlined in the patent provides a streamlined pathway for generating high-value chiral building blocks. The procedure begins with the preparation of the oxime ether substrate, followed by the addition of the pre-formed spiroboronate catalyst in an anhydrous solvent system. The reaction is typically initiated at low temperatures, such as 0°C, to maximize stereocontrol, before potentially warming to ensure complete conversion. This flexibility in temperature programming allows for the optimization of both rate and selectivity depending on the specific steric demands of the substrate. The workup involves standard acidic quenching followed by basification and extraction, avoiding complex filtration steps often required for heterogeneous catalysts. For detailed operational parameters and safety guidelines regarding borane handling, please refer to the standardized synthesis steps provided below.
- Preparation of the catalytic system by mixing chiral spiroboronic ester (e.g., Catalyst 5 derived from diphenyl valinol) with borane source (BH3·THF or BH3·DMS) in anhydrous solvent like dioxane under inert atmosphere.
- Controlled addition of the oxime ether substrate to the pre-formed catalyst-borane complex at optimized temperatures ranging from 0°C to room temperature to ensure high enantioselectivity.
- Quenching the reaction mixture with acid/base workup followed by extraction and purification via column chromatography to isolate the high-purity primary amine or its acetyl derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this catalytic technology offers profound strategic benefits beyond mere technical performance. The shift from stoichiometric chiral reagents to a catalytic regime drastically reduces the consumption of high-cost chiral pool materials. Since the catalyst loading is reduced from greater than one equivalent to as low as ten percent, the direct material cost per kilogram of product is significantly lowered. This reduction in raw material intensity not only improves the gross margin of the final intermediate but also mitigates the risk associated with the supply volatility of expensive chiral amino alcohols. Furthermore, the stability of the spiroboronate catalysts means they can be sourced in bulk and stored without degradation, allowing for better inventory management and reducing the frequency of just-in-time ordering which can disrupt production schedules.
- Cost Reduction in Manufacturing: The elimination of stoichiometric chiral auxiliaries removes a major cost driver from the bill of materials. By operating in a true catalytic mode, the process minimizes waste generation and reduces the burden on waste treatment facilities, leading to substantial cost savings in environmental compliance and disposal. Additionally, the simplified workup procedure reduces solvent consumption and processing time, further enhancing the overall economic efficiency of the manufacturing campaign without compromising on quality standards.
- Enhanced Supply Chain Reliability: The robustness of the catalyst system ensures consistent batch-to-batch reproducibility, which is critical for maintaining supply continuity to downstream API manufacturers. The use of commodity chemicals like borane-THF and common ether solvents means that the supply chain is not dependent on niche or single-source reagents. This diversification of raw material sources enhances resilience against market fluctuations and geopolitical disruptions, ensuring that production timelines are met reliably even in volatile market conditions.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated to work efficiently across various substrate classes including arylalkyl and pyridylalkyl ketoximes. The absence of transition metals in the catalyst system simplifies the purification process, as there is no need for expensive and time-consuming heavy metal scavenging steps to meet strict residual metal limits. This green chemistry attribute not only accelerates regulatory approval but also aligns with corporate sustainability goals by reducing the environmental footprint of the chemical synthesis.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this asymmetric reduction technology. These insights are derived directly from the experimental data and embodiments disclosed in the patent documentation, providing a reliable foundation for decision-making. Understanding these nuances helps stakeholders evaluate the feasibility of integrating this method into existing production lines or new drug development programs. For more specific technical data or custom feasibility studies, our team is available to provide detailed consultations tailored to your specific project requirements.
Q: What is the primary advantage of using spiroboronic esters over traditional oxazaborolidines?
A: Unlike traditional oxazaborolidines which often require stoichiometric amounts or in situ preparation, spiroboronic esters are stable, isolable solids that function effectively at true catalytic loadings (as low as 10%), significantly reducing material costs and simplifying handling.
Q: Can this methodology be applied to pyridyl-containing substrates?
A: Yes, the patent demonstrates successful application to pyridylalkyl ketoximes. While some optimization of temperature and catalyst loading (up to 30%) may be required due to the coordinating nature of the pyridine nitrogen, excellent enantioselectivities (>98% ee) are achievable.
Q: How does the solvent choice impact enantioselectivity in this reduction?
A: Solvent polarity and coordination play a critical role. Ether solvents like dioxane generally provide superior enantioselectivity (up to 97% ee) compared to toluene or chlorinated solvents, likely due to favorable stabilization of the transition state without competing coordination.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Primary Amines Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic technologies like the spiroboronate-mediated reduction described in CN101547890A. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory processes are seamlessly translated into robust industrial operations. Our facility is equipped with rigorous QC labs and stringent purity specifications capable of handling sensitive borane chemistry and chiral separations. We are committed to delivering high-purity pharmaceutical intermediates that meet the exacting standards of the global healthcare industry, leveraging our deep technical expertise to optimize yield and enantioselectivity for every client project.
We invite you to collaborate with us to unlock the full commercial potential of your chiral amine projects. Our technical procurement team is ready to assist you with a Customized Cost-Saving Analysis to quantify the economic benefits of switching to this catalytic route for your specific molecule. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our integrated manufacturing solutions can accelerate your time to market while optimizing your supply chain costs.
