Scalable Production of 2-Trifluoromethyl Quinazolinones via Iron Catalysis for Global Pharma Supply Chains
Introduction to Advanced Quinazolinone Synthesis
The pharmaceutical and agrochemical industries continuously demand robust synthetic methodologies for nitrogen-containing heterocycles, particularly quinazolinones, due to their pervasive presence in bioactive molecular scaffolds. As detailed in patent CN111675662B, a significant technological breakthrough has been achieved in the preparation of 2-trifluoromethyl substituted quinazolinone compounds. These structures are critical motifs found in numerous drug candidates exhibiting anti-cancer, anticonvulsant, and anti-inflammatory properties. The strategic introduction of the trifluoromethyl group enhances metabolic stability and lipophilicity, key parameters for oral bioavailability. This novel approach leverages a cost-effective iron-catalyzed system to construct these complex fused rings, addressing long-standing challenges in heterocyclic chemistry regarding reagent cost and reaction severity.
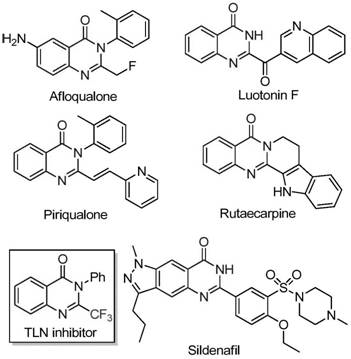
The significance of this invention lies in its ability to utilize readily available starting materials to access high-value intermediates. Unlike legacy methods that rely on scarce or hazardous reagents, this protocol employs trifluoroethylimidoyl chloride and isatin derivatives. The versatility of this method allows for the generation of diverse chemical libraries, enabling medicinal chemists to rapidly explore structure-activity relationships (SAR). For global supply chains, the shift towards base-metal catalysis represents a pivotal move towards sustainable and economically viable manufacturing processes, ensuring a steady supply of high-purity intermediates for downstream API production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of trifluoromethyl-substituted quinazolinones has been plagued by significant operational and economic hurdles. Traditional routes often depend on the cyclization of anthranilamides or isatoic anhydrides with trifluoroacetic anhydride or ethyl trifluoroacetate. These conventional synthons are frequently expensive, limiting their utility in large-scale commercial applications. Moreover, the reaction conditions required for these transformations are often severe, necessitating high temperatures or strong bases that can degrade sensitive functional groups. The narrow substrate scope of these older methods means that introducing diverse substituents often requires extensive re-optimization, leading to prolonged development timelines and increased R&D expenditures. Additionally, the use of stoichiometric amounts of activating agents generates substantial chemical waste, complicating environmental compliance and waste disposal protocols.
The Novel Approach
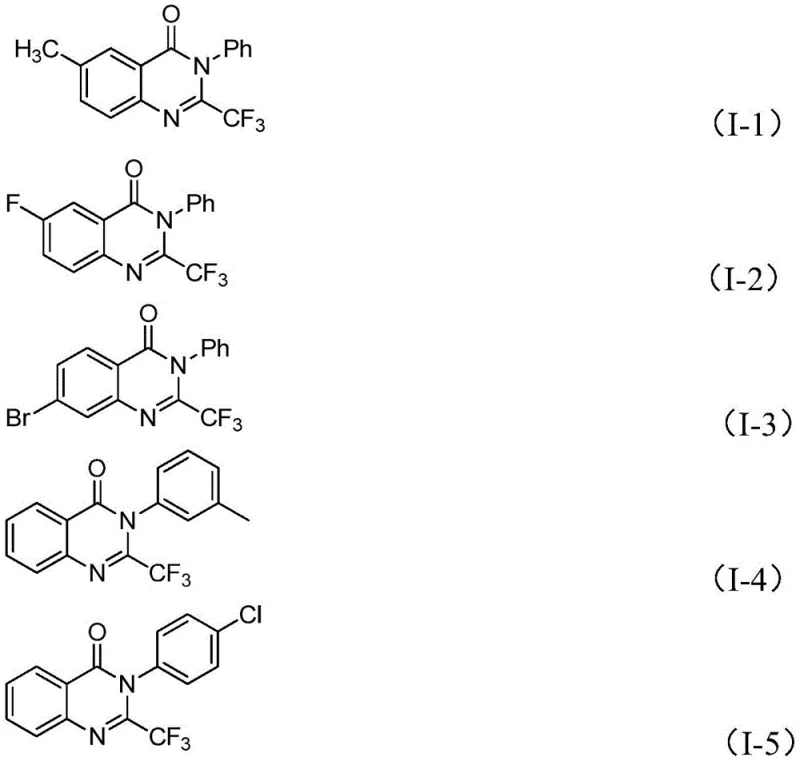
In stark contrast, the methodology disclosed in the patent utilizes a convergent strategy involving the condensation of trifluoroethylimidoyl chloride and isatin derivatives. This approach fundamentally alters the economic landscape of quinazolinone production by employing cheap, commercially available iron salts as catalysts instead of precious metals. The reaction proceeds through a tandem sequence involving carbon-nitrogen bond formation followed by decarbonylation and cyclization. This streamlined pathway not only improves atom economy but also significantly enhances the functional group tolerance of the process. By operating under relatively mild conditions with accessible reagents, this novel route minimizes the formation of by-products and simplifies the purification workflow. The ability to synthesize a wide array of derivatives, such as those shown in the structural examples below, underscores the robustness and adaptability of this new synthetic platform for industrial application.
Mechanistic Insights into FeCl3-Catalyzed Cyclization
The core of this technological advancement is the iron-catalyzed cascade reaction that efficiently constructs the quinazolinone core. The mechanism initiates with the activation of the trifluoroethylimidoyl chloride by the base, sodium hydride, facilitating a nucleophilic attack on the carbonyl carbon of the isatin derivative. This step forms a key trifluoroacetamidine intermediate. Subsequently, the ferric chloride catalyst plays a dual role: it promotes the decarbonylation of the isatin moiety and facilitates the intramolecular cyclization required to close the six-membered ring. The presence of 4A molecular sieves is crucial in this system, as they act as a water scavenger, driving the equilibrium towards product formation and preventing the hydrolysis of sensitive intermediates. This mechanistic pathway avoids the high-energy barriers associated with traditional thermal cyclizations, allowing the reaction to proceed with high efficiency at moderate temperatures.
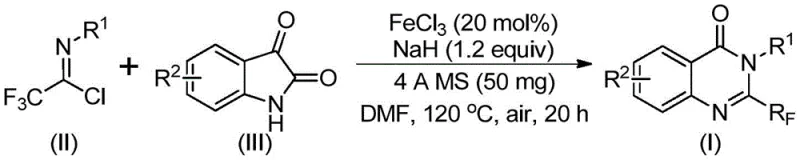
From an impurity control perspective, this mechanism offers distinct advantages. The selectivity of the iron catalyst ensures that the cyclization occurs regioselectively, minimizing the formation of isomeric by-products that are common in uncatalyzed thermal reactions. The use of DMF as a solvent further stabilizes the polar transition states involved in the decarbonylation step. Understanding this mechanistic nuance is vital for process chemists aiming to scale this reaction, as it highlights the importance of maintaining anhydrous conditions and precise stoichiometric control of the base. The result is a clean reaction profile that yields the target 2-trifluoromethyl quinazolinone with high purity, reducing the burden on downstream purification units and ensuring consistent quality for regulatory submissions.
How to Synthesize 2-Trifluoromethyl Quinazolinone Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires adherence to specific operational parameters to maximize yield and safety. The protocol is designed to be user-friendly, utilizing standard glassware and commercially sourced reagents. The reaction begins with the careful addition of sodium hydride to the reaction vessel containing the catalyst and molecular sieves, followed by the substrates in DMF. A two-stage heating profile is employed: an initial period at 40°C allows for the formation of the intermediate, followed by heating to 120°C to drive the cyclization to completion. Detailed standardized operating procedures for this synthesis are provided in the structured guide below, ensuring reproducibility across different batches and facilities.
- Combine ferric chloride (20 mol%), sodium hydride (1.2 equiv), 4A molecular sieves, trifluoroethylimidoyl chloride, and isatin derivative in DMF solvent within a Schlenk tube.
- Stir the mixture at 40°C for 8-10 hours to initiate the reaction, then increase temperature to 120°C and react for an additional 18-20 hours under air.
- Upon completion, filter the reaction mixture, mix with silica gel, and purify via column chromatography to isolate the target 2-trifluoromethyl quinazolinone.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this iron-catalyzed methodology presents a compelling value proposition centered on cost stability and operational resilience. The shift from precious metal catalysts to abundant iron salts drastically reduces the raw material cost baseline, insulating the manufacturing process from the volatile price fluctuations often seen in the platinum group metals market. Furthermore, the simplicity of the workup procedure—filtration followed by chromatography—reduces the consumption of solvents and stationary phases, contributing to lower overall production costs. This efficiency translates directly into improved margins for the final active pharmaceutical ingredient, making the supply chain more competitive in a price-sensitive global market.
- Cost Reduction in Manufacturing: The elimination of expensive palladium or rhodium catalysts removes a significant cost driver from the bill of materials. Iron chloride is a commodity chemical with a stable supply chain, ensuring predictable pricing. Additionally, the high yields reported (up to 93%) minimize the loss of valuable starting materials, further enhancing the economic viability of the process. The reduced need for specialized equipment to handle air-sensitive precious metals also lowers capital expenditure requirements for manufacturing sites.
- Enhanced Supply Chain Reliability: The starting materials, specifically isatin derivatives and trifluoroethylimidoyl chlorides, are widely available from multiple global suppliers, reducing the risk of single-source bottlenecks. The robustness of the reaction conditions means that the process is less susceptible to minor variations in raw material quality or environmental factors, ensuring consistent batch-to-batch output. This reliability is critical for maintaining continuous API production schedules and meeting strict delivery commitments to downstream pharmaceutical partners.
- Scalability and Environmental Compliance: The use of DMF, while requiring careful handling, is a well-understood solvent in the industry with established recovery and recycling protocols. The absence of heavy metal residues in the final product simplifies the regulatory clearance process, as stringent limits on metal impurities do not require complex scavenging steps. The process generates less hazardous waste compared to traditional methods using stoichiometric activators, aligning with modern green chemistry principles and reducing the environmental footprint of the manufacturing facility.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and technical specifications outlined in the patent documentation. They are intended to provide clarity for R&D teams evaluating the feasibility of this route and for procurement specialists assessing the supply risks. Understanding these details is essential for making informed decisions about integrating this technology into existing production portfolios.
Q: What are the primary advantages of using FeCl3 over traditional catalysts for this synthesis?
A: The use of ferric chloride (FeCl3) offers a significant economic advantage as it is an abundant, non-precious metal catalyst compared to expensive palladium or rhodium systems. Furthermore, it demonstrates excellent functional group tolerance and operates effectively under aerobic conditions, simplifying the operational requirements for large-scale manufacturing.
Q: Can this method accommodate diverse substituents on the isatin and imidoyl chloride substrates?
A: Yes, the protocol exhibits broad substrate scope. It successfully tolerates various substituents including alkyl, halogen (F, Cl, Br, I), methoxy, and nitro groups at ortho-, meta-, and para-positions, allowing for the synthesis of a wide library of analogues for SAR studies without needing to optimize conditions for each variant.
Q: Is the purification process complex for industrial scale-up?
A: The post-treatment is straightforward and scalable. The process involves simple filtration to remove solids followed by standard silica gel column chromatography. The use of inexpensive reagents and the absence of difficult-to-remove transition metal residues streamline the downstream processing, making it highly suitable for commercial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Quinazolinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the drug development lifecycle. Our team of expert process chemists has extensively evaluated the iron-catalyzed route described in CN111675662B and confirmed its potential for robust commercial manufacturing. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can transition seamlessly from gram-scale discovery to multi-ton industrial supply. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 2-trifluoromethyl quinazolinone meets the highest international standards.
We invite you to collaborate with us to leverage this cost-effective technology for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our optimized manufacturing processes can enhance your supply chain efficiency and reduce your overall time to market.
