Advanced Room-Temperature Synthesis of Benzopyran Intermediates for Scalable Pharma Manufacturing
Introduction to Next-Generation Benzopyran Synthesis
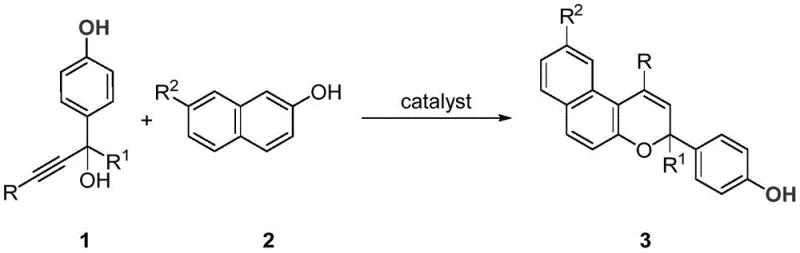
The pharmaceutical and fine chemical industries are constantly seeking more efficient, sustainable, and cost-effective pathways to access complex heterocyclic scaffolds. A significant breakthrough in this domain is detailed in Chinese Patent CN112079808A, published in December 2020, which discloses a novel method for preparing benzopyran compounds based on the in-situ generation of alkynyl-substituted p-methylene benzoquinone. This technology represents a paradigm shift from traditional high-energy synthetic routes, offering a streamlined one-pot procedure that operates under remarkably mild conditions. By leveraging specific Brønsted acid catalysts, this method enables the direct coupling of 4-hydroxyaryl-substituted propargyl alcohols with 2-naphthol derivatives at room temperature. For R&D directors and process chemists, this patent offers a compelling solution to the longstanding challenges of synthesizing benzopyran cores, which are ubiquitous in bioactive natural products and drug molecules. The elimination of harsh oxidants and expensive metal catalysts not only simplifies the operational workflow but also aligns perfectly with modern green chemistry principles, making it an attractive candidate for industrial adoption.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the benzopyran skeleton has relied heavily on cyclization reactions involving propargyl alcohols and phenols or naphthols under the influence of transition metal catalysts. These conventional methodologies, while effective in certain contexts, suffer from significant drawbacks that hinder their utility in large-scale manufacturing. Typically, these reactions require elevated temperatures and the presence of stoichiometric or catalytic amounts of precious metals, which introduces the risk of heavy metal contamination in the final active pharmaceutical ingredient (API). Furthermore, the need for external oxidants to drive the oxidative cyclization adds complexity to the process, increasing both the material costs and the environmental burden associated with waste disposal. The operational difficulty is compounded by the sensitivity of some substrates to high thermal stress, often leading to decomposition, polymerization, or the formation of complex impurity profiles that are difficult to separate. Consequently, the overall yield is frequently compromised, and the purification steps become labor-intensive and costly, creating a bottleneck for the reliable supply of high-purity pharmaceutical intermediates.
The Novel Approach
In stark contrast to these legacy methods, the technology described in Patent CN112079808A introduces a transformative approach that bypasses the need for metals and heat. This novel strategy utilizes a Brønsted acid-catalyzed tandem reaction that proceeds efficiently at room temperature. The core innovation lies in the in-situ generation of a reactive alkynyl-substituted p-methylene benzoquinone intermediate, which subsequently undergoes a smooth cyclization with the naphthol partner. This method boasts an exceptionally broad scope of functional group tolerance, accommodating halogens, alkyls, and alkoxy groups without the need for protective group strategies. The operational simplicity is unparalleled; reactants are simply mixed with a catalyst such as (±)-binaphthol phosphate in a common solvent like dichloromethane, and the reaction proceeds to completion with high conversion. This eliminates the need for specialized high-pressure equipment or rigorous temperature control systems, thereby drastically reducing the capital expenditure required for implementation. For procurement managers, this translates to a robust supply chain for benzopyran derivatives that is less susceptible to the volatility of precious metal markets and energy costs.
Mechanistic Insights into Brønsted Acid-Catalyzed Cyclization
The mechanistic elegance of this transformation is rooted in the activation of the propargylic alcohol by the Brønsted acid catalyst. Upon protonation, the hydroxyl group becomes a superior leaving group, facilitating the formation of a propargylic cation or a related reactive species. This activation triggers a cascade of events, beginning with the dehydration to form the key alkynyl-substituted p-methylene benzoquinone intermediate in situ. This highly electrophilic species is then intercepted by the electron-rich 2-naphthol nucleophile through a 1,8-conjugate addition. Following this initial C-C bond formation, the system undergoes an intramolecular 6-endo-dig cyclization to close the pyran ring, ultimately yielding the stable benzopyran framework. The choice of catalyst is critical; while various acids like camphorsulfonic acid or trifluoroacetic acid can promote the reaction, chiral phosphoric acids like (±)-binaphthol phosphate have been shown to offer superior efficiency and potentially enable enantioselective variants in future iterations. Understanding this mechanism allows process chemists to fine-tune reaction parameters, such as catalyst loading and solvent polarity, to maximize throughput and minimize side reactions.
From an impurity control perspective, the mildness of the reaction conditions is a decisive factor. High-temperature processes often lead to thermal degradation of sensitive functional groups or non-selective polymerization of the reactive quinone methide intermediates. By maintaining the reaction at room temperature, the kinetic energy of the system is kept low enough to favor the desired cyclization pathway over competing decomposition routes. This results in a cleaner crude reaction profile, which significantly reduces the burden on downstream purification units. For quality control teams, this means that achieving stringent purity specifications becomes more predictable and less resource-intensive. The ability to tolerate diverse substituents, such as the bromo and chloro groups seen in specific examples, further underscores the robustness of this catalytic cycle, ensuring that the process remains viable even when synthesizing complex, halogenated intermediates required for downstream cross-coupling reactions.
How to Synthesize Benzopyran Compounds Efficiently
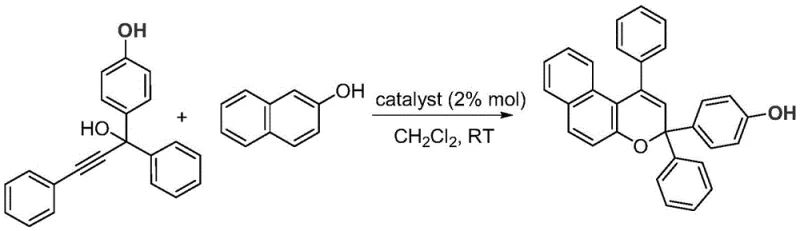
Implementing this synthesis in a laboratory or pilot plant setting is straightforward and requires minimal specialized equipment. The general protocol involves dissolving the 4-hydroxyaryl-substituted propargyl alcohol and the 2-naphthol derivative in dichloromethane, followed by the addition of the catalyst, typically at a loading of 2% mol relative to the substrate. The mixture is then stirred at ambient temperature, with reaction times ranging from as little as 20 minutes to several hours depending on the electronic nature of the substituents. Monitoring the reaction via thin-layer chromatography (TLC) allows for precise determination of the endpoint, preventing over-reaction. Once complete, the workup is remarkably simple; the solvent can be removed, and the crude product purified directly via silica gel column chromatography using a standard petroleum ether and ethyl acetate gradient. This streamlined workflow eliminates the need for aqueous quenching steps often required to remove metal catalysts, further enhancing the overall process efficiency and yield.
- Combine 4-hydroxyaryl-substituted propargyl alcohol, 2-naphthol derivative, and a Brønsted acid catalyst (e.g., (±)-binaphthol phosphate) in dichloromethane.
- Stir the reaction mixture at room temperature for 3.5 to 36 hours, monitoring progress via TLC or HPLC until completion.
- Purify the crude reaction mixture directly using silica gel column chromatography with a petroleum ether and ethyl acetate eluent system to isolate the pure benzopyran product.
Commercial Advantages for Procurement and Supply Chain Teams
For stakeholders focused on the bottom line and supply chain resilience, this patented methodology offers substantial strategic benefits. The shift away from transition metal catalysis removes a major cost driver and supply risk associated with fluctuating prices of precious metals like palladium or gold. Furthermore, the elimination of external oxidants simplifies the reagent list, reducing inventory complexity and storage hazards. The ability to run reactions at room temperature significantly lowers energy consumption compared to refluxing conditions, contributing to a smaller carbon footprint and reduced utility costs. These factors combine to create a manufacturing process that is not only economically superior but also more sustainable and compliant with increasingly strict environmental regulations. The high yields reported in the patent examples suggest that material throughput can be maximized, minimizing waste and improving the overall atom economy of the production line.
- Cost Reduction in Manufacturing: The primary economic advantage stems from the replacement of expensive transition metal catalysts with inexpensive, commercially available Brønsted acids. This substitution drastically lowers the raw material cost per kilogram of the final product. Additionally, the simplified workup procedure, which avoids complex metal scavenging steps, reduces the consumption of auxiliary materials and labor hours. The high reaction efficiency means less starting material is wasted, directly improving the cost of goods sold (COGS) for the final benzopyran intermediate. This makes the process highly competitive for the production of cost-sensitive generic drug intermediates.
- Enhanced Supply Chain Reliability: The starting materials for this reaction, specifically propargyl alcohols and naphthols, are commodity chemicals that are widely available from multiple global suppliers. This diversity in sourcing mitigates the risk of supply disruptions that can occur with specialized or single-source reagents. Moreover, the robustness of the reaction conditions ensures consistent batch-to-batch quality, which is critical for maintaining long-term contracts with pharmaceutical clients. The stability of the process at room temperature also reduces the risk of thermal runaway incidents, enhancing facility safety and ensuring uninterrupted production schedules.
- Scalability and Environmental Compliance: Scaling this reaction from gram to ton scale is facilitated by the absence of exothermic hazards associated with strong oxidants or high-temperature operations. The use of dichloromethane, a common industrial solvent, allows for easy integration into existing solvent recovery systems. From an environmental standpoint, the reduction in heavy metal waste and the avoidance of stoichiometric oxidants significantly lower the E-factor of the process. This alignment with green chemistry principles simplifies regulatory filings and environmental impact assessments, accelerating the time to market for new drug candidates utilizing this scaffold.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this benzopyran synthesis technology. These answers are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing a clear understanding of the process capabilities and limitations. Whether you are evaluating this route for a specific project or assessing its fit within your broader manufacturing portfolio, these insights will help clarify the operational realities of adopting this novel methodology.
Q: What are the key advantages of this benzopyran synthesis method over traditional metal-catalyzed routes?
A: This method operates at room temperature without requiring expensive transition metal catalysts or external oxidants. It utilizes cheap, commercially available Brønsted acid catalysts like (±)-binaphthol phosphate, resulting in significantly lower production costs and simplified purification processes compared to high-temperature metal-catalyzed cyclizations.
Q: What is the substrate scope and functional group tolerance for this reaction?
A: The protocol demonstrates excellent functional group tolerance, accommodating various substituents on both the propargyl alcohol and the naphthol components. Halogens (bromo, chloro), alkyl groups, alkoxy groups, and even sensitive moieties like cyclopropyl rings are well-tolerated, yielding products with high purity and minimal side reactions.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the process is highly scalable due to its mild operating conditions (room temperature) and the use of common solvents like dichloromethane. The absence of hazardous oxidants and the high yields (up to 99% in lab scale) indicate strong potential for cost-effective commercial scale-up with reduced safety risks and waste generation.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Benzopyran Compounds Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this acid-catalyzed synthesis route for the production of high-value benzopyran intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent and reliable supply of critical building blocks. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of benzopyran compound delivered meets the highest industry standards. We understand that transitioning to a new synthetic route requires confidence in both the chemistry and the manufacturer, and our track record of successful technology transfers speaks to our commitment to excellence.
We invite pharmaceutical companies and research institutions to collaborate with us to leverage this efficient synthesis technology for their specific projects. By partnering with us, you gain access to a Customized Cost-Saving Analysis tailored to your unique volume requirements and quality needs. We encourage you to contact our technical procurement team today to request specific COA data for our benzopyran portfolio and to discuss detailed route feasibility assessments. Let us help you optimize your supply chain and accelerate your drug development timeline with our advanced manufacturing capabilities.
