Advanced Synthesis of 2-Bromopyrene: Technical Breakthroughs for OLED Material Manufacturing
Advanced Synthesis of 2-Bromopyrene: Technical Breakthroughs for OLED Material Manufacturing
The rapid expansion of the organic light-emitting diode (OLED) industry has placed immense pressure on the supply chain for high-performance intermediate materials, specifically demanding higher purity and lower production costs for key building blocks like 2-bromopyrene. Patent CN109280011B, published in mid-2021, introduces a transformative synthesis methodology that addresses the longstanding inefficiencies associated with traditional pyrene functionalization. This technical disclosure outlines a robust pathway starting from 1-aminopyrene, utilizing a strategic bromination followed by a diazotization-deamination sequence to yield 2-bromopyrene with exceptional purity levels exceeding 99.5%. For R&D directors and procurement specialists in the electronic chemicals sector, this patent represents a critical pivot point away from low-yield, multi-step legacy processes toward a streamlined, industrially viable manufacturing protocol that promises significant operational efficiencies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of 2-bromopyrene has been plagued by complex reaction sequences that severely impact overall throughput and economic feasibility. The conventional wisdom relied on a tedious three-step transformation involving the hydrogenation of pyrene, followed by bromination, and finally a difficult aromatization step to restore the aromatic system. As illustrated in the reaction scheme below, this legacy approach suffers from compounding yield losses at every stage, with individual step yields often plummeting to less than or equal to 20 percent. Such inefficiencies not only drive up the cost of goods sold (COGS) but also create substantial waste management challenges due to the accumulation of byproducts and the need for extensive purification between each discrete operation. Furthermore, the difficulty in isolating pure 2-bromopyrene from the complex reaction mixtures generated by these harsh conditions has traditionally acted as a bottleneck for scaling production to meet the surging demand from the display and lighting sectors.
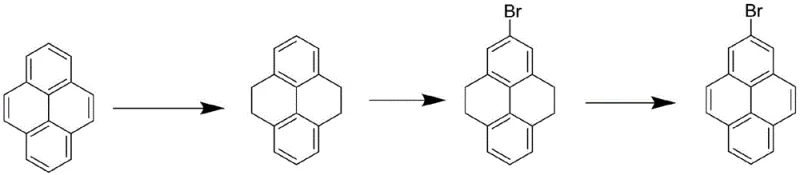
The Novel Approach
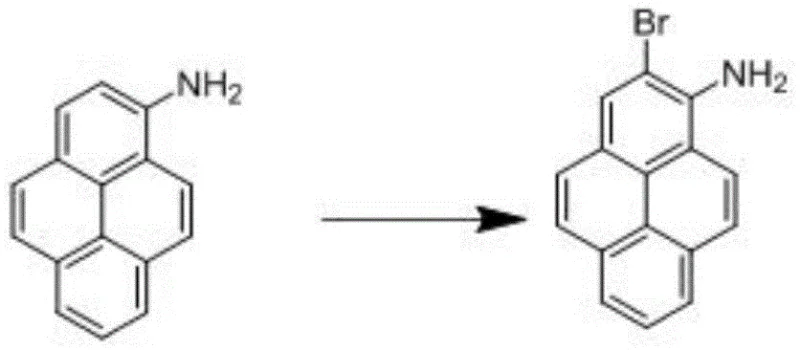
In stark contrast to the cumbersome legacy routes, the novel approach detailed in the patent data leverages the inherent reactivity of the amino group to facilitate a much more direct and controlled functionalization. By initiating the synthesis with 1-aminopyrene, a readily available and cost-effective precursor, the process bypasses the need for initial hydrogenation and subsequent difficult aromatization. The strategy employs a highly selective electrophilic substitution to install the bromine atom at the desired position, followed by a clean removal of the amino directing group via diazotization. This shift in synthetic logic fundamentally alters the economic landscape of 2-bromopyrene manufacturing, offering a reliable OLED material supplier pathway that drastically simplifies the operational workflow. The ability to achieve high yields in excess of 80 percent for the final deamination step, coupled with the high efficiency of the initial bromination, demonstrates a clear superiority in atom economy and process mass intensity compared to the prior art.
Mechanistic Insights into Diazotization Deamination and Bromination
The core of this technological advancement lies in the precise control of reaction conditions during the two critical transformation stages. The first stage involves the bromination of 1-aminopyrene, where the amino group acts as a powerful activating and ortho/para-directing group. By conducting this reaction in a polar aprotic solvent like DMF at strictly controlled low temperatures of 0-5 °C, the process ensures mono-bromination occurs selectively at the 2-position. The patent highlights the versatility of brominating agents, successfully demonstrating the use of N-bromosuccinimide (NBS), elemental bromine, or dibromohydantoin, all of which deliver yields greater than 95 percent. This high selectivity minimizes the formation of poly-brominated impurities, which are notoriously difficult to separate later in the process, thereby simplifying the downstream purification burden and enhancing the overall quality of the intermediate stream entering the second stage.
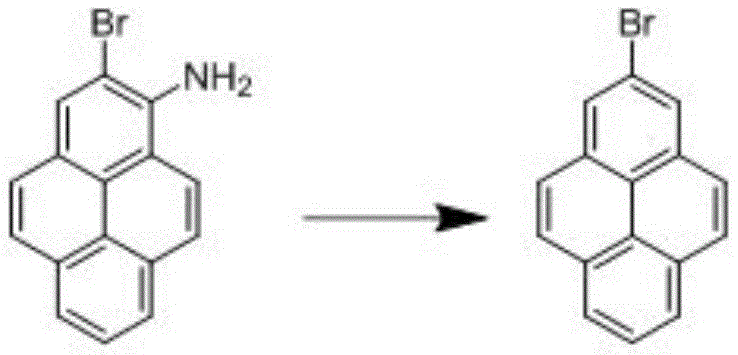
The second mechanistic pillar is the diazotization-deamination sequence, which serves as the definitive step to reveal the final 2-bromopyrene structure. In this phase, the 1-amino-2-bromopyrene is dissolved in a concentrated sulfuric acid solution, a critical choice of acid medium that ensures complete solubility of the substrate, unlike hydrochloric acid which leads to precipitation and poor reaction kinetics. Upon the dropwise addition of sodium nitrite at 0-5 °C, the unstable diazonium salt is formed in situ. This intermediate is immediately subjected to reduction using hypophosphorous acid, which acts as a mild yet effective reducing agent to replace the diazonium group with a hydrogen atom. The reaction scheme below depicts this clean conversion, which avoids the use of toxic heavy metal catalysts often found in alternative deamination methods. The rigorous temperature control throughout this exothermic process is paramount for safety and for preventing the decomposition of the diazonium species into phenolic byproducts, ensuring the final crude product is of sufficient quality for simple recrystallization.
How to Synthesize 2-Bromopyrene Efficiently
The implementation of this synthesis route requires strict adherence to the thermal and stoichiometric parameters defined in the patent to guarantee reproducibility and safety on a commercial scale. The process is designed to be operationally simple, relying on standard unit operations such as cooling, filtration, and recrystallization, which facilitates easy technology transfer from the laboratory to the pilot plant. Operators must pay particular attention to the exothermic nature of the diazotization and the subsequent reduction, maintaining the reaction mass between 0-5 °C to prevent runaway scenarios. The following guide outlines the standardized operational procedure derived from the patent examples, providing a clear roadmap for process engineers to establish a robust manufacturing line for this high-value electronic chemical.
- Brominate 1-aminopyrene using NBS or dibromohydantoin in DMF at 0-5 °C to obtain 1-amino-2-bromopyrene.
- Perform diazotization in sulfuric acid with sodium nitrite at 0-5 °C, followed by reduction with hypophosphorous acid.
- Purify the crude product via recrystallization in ethyl acetate to achieve purity levels exceeding 99.5%.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthesis methodology offers compelling strategic benefits that extend far beyond simple chemical yield improvements. The shift to a 1-aminopyrene based route fundamentally restructures the cost basis of 2-bromopyrene production by eliminating the need for expensive hydrogenation catalysts and high-pressure equipment associated with the older pyrene reduction methods. This simplification of the process infrastructure translates directly into reduced capital expenditure (CAPEX) for new production lines and lower operating expenditures (OPEX) for existing facilities, creating a more resilient supply chain capable of withstanding market volatility. Furthermore, the high selectivity of the reactions reduces the load on waste treatment facilities, aligning production with increasingly stringent environmental regulations and avoiding potential shutdowns due to compliance issues.
- Cost Reduction in Manufacturing: The economic argument for this new process is anchored in the utilization of 1-aminopyrene, a raw material that is significantly more affordable and accessible than the specialized reagents required for the traditional three-step pyrene functionalization. By achieving yields greater than 95 percent in the bromination step and over 80 percent in the deamination step, the process minimizes raw material waste, which is a primary driver of cost in fine chemical synthesis. Additionally, the elimination of transition metal catalysts removes the necessity for expensive and time-consuming heavy metal scavenging steps, further streamlining the production timeline and reducing the consumption of auxiliary materials. This cumulative effect results in a substantially lower cost of goods, allowing suppliers to offer more competitive pricing structures to downstream OLED panel manufacturers without compromising on margin.
- Enhanced Supply Chain Reliability: From a logistics and sourcing perspective, the reliance on commodity chemicals such as sulfuric acid, sodium nitrite, and hypophosphorous acid enhances the robustness of the supply chain against disruptions. Unlike proprietary catalysts or exotic reagents that may have single-source suppliers and long lead times, the reagents used in this patent are widely available from multiple global vendors, ensuring continuity of supply even during periods of market stress. The simplicity of the synthesis, which avoids complex multi-stage telescoping or sensitive anhydrous conditions for the majority of the steps, also reduces the risk of batch failures due to operator error or equipment malfunction. This reliability is crucial for maintaining the just-in-time delivery schedules required by the fast-paced consumer electronics industry.
- Scalability and Environmental Compliance: The process is inherently scalable, utilizing standard batch reactor technologies that are common in fine chemical manufacturing facilities worldwide. The purification step, which relies on a straightforward recrystallization from ethyl acetate, is easily adaptable from kilogram to multi-ton scales without requiring complex chromatographic separations that are difficult to scale. Moreover, the aqueous workup and the use of relatively benign solvents contribute to a greener process profile, reducing the volume of hazardous organic waste generated per kilogram of product. This environmental efficiency not only lowers disposal costs but also supports the sustainability goals of major technology corporations who are increasingly demanding green chemistry credentials from their chemical suppliers.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology, derived directly from the experimental data and comparative examples provided in the patent documentation. Understanding these nuances is essential for process chemists and quality assurance teams evaluating the feasibility of adopting this route for commercial production. The answers reflect the specific advantages observed in the patent examples, particularly regarding solvent selection and acid media optimization.
Q: Why is sulfuric acid preferred over hydrochloric acid in the diazotization step?
A: According to patent CN109280011B, sulfuric acid ensures complete dissolution of the 1-amino-2-bromopyrene substrate. Comparative data shows that using hydrochloric acid results in poor solubility, leading to significantly lower yields (32%) and purity (55.7%) compared to the sulfuric acid method.
Q: What represents the primary cost advantage of this new synthesis route?
A: The primary cost advantage stems from utilizing 1-aminopyrene as a starting material, which is commercially more accessible and affordable than the multi-step pyrene hydrogenation route. Additionally, the high yield (>80%) and simplified purification reduce overall production costs.
Q: How is high purity (>99.5%) achieved in the final product?
A: High purity is achieved through a specific recrystallization protocol using ethyl acetate. The crude product is dissolved by heating to reflux and then cooled to 20 ± 2 °C, which effectively precipitates the pure 2-bromopyrene while leaving impurities in the solution.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Bromopyrene Supplier
As the global demand for high-performance OLED materials continues to accelerate, the ability to source critical intermediates like 2-bromopyrene from a technically proficient and scalable partner becomes a key differentiator for success. NINGBO INNO PHARMCHEM stands at the forefront of this sector, leveraging deep expertise in organic synthesis to translate complex patent methodologies like CN109280011B into reliable commercial realities. Our facility is equipped with extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the volumetric requirements of large-scale display manufacturing without compromising on the stringent purity specifications required for electronic grade materials. Our rigorous QC labs employ advanced analytical techniques to verify that every batch meets the >99.5% purity benchmark, guaranteeing consistent performance in your final OLED devices.
We invite industry leaders to engage with our technical procurement team to discuss how our optimized synthesis routes can drive value for your organization. By partnering with us, you gain access to a Customized Cost-Saving Analysis that quantifies the potential economic benefits of switching to our high-efficiency manufacturing protocols. We encourage you to contact us directly to request specific COA data and route feasibility assessments tailored to your specific project needs, ensuring a seamless integration of our high-purity 2-bromopyrene into your supply chain.
