Advanced Diacid Catalysis for High-Purity Triaryl Ethylene Manufacturing and Scale-Up
Advanced Diacid Catalysis for High-Purity Triaryl Ethylene Manufacturing and Scale-Up
The landscape of organic synthesis for high-value pharmaceutical intermediates is constantly evolving, driven by the need for more efficient, selective, and scalable processes. A significant breakthrough in this domain is detailed in patent CN112811986A, which introduces a novel method for synthesizing triaryl ethylene compounds using a dual-acid catalytic system. Triaryl ethylenes are not merely academic curiosities; they form the structural backbone of critical therapeutics such as (Z)-tamoxifen, a cornerstone drug in breast cancer treatment, and possess high optical activity valuable for OLED device fabrication. The traditional reliance on harsh conditions and limited substrate scope has long been a bottleneck for procurement and R&D teams seeking reliable supply chains for these complex molecules. This new methodology addresses those pain points directly by leveraging a synergistic combination of Lewis and Bronsted acids to drive the reaction with exceptional precision.
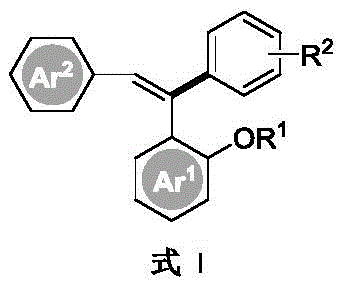
The core innovation lies in the ability to construct the triaryl ethylene scaffold with unprecedented control over both regioselectivity and stereoselectivity. Unlike older methods that struggle with isomeric mixtures, this dual-catalyst approach ensures the formation of the desired Z-isomer with high fidelity. For a Chief Technology Officer or a Process Development Lead, this translates to a drastic reduction in downstream purification costs and waste generation. The patent outlines a robust protocol where diaryl acetylenes react with substituted benzenes under inert atmospheres, yielding products that meet stringent purity specifications required for API manufacturing. By mastering this chemistry, manufacturers can secure a competitive edge in the supply of high-purity pharmaceutical intermediates, ensuring continuity for life-saving medications and advanced electronic materials.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of triaryl ethylene compounds via Friedel-Crafts type reactions has been plagued by significant technical hurdles that impact both cost and quality. Conventional protocols typically demand aromatic hydrocarbons possessing multiple electron-donating substituents to facilitate the electrophilic attack, severely restricting the diversity of accessible chemical space. This limitation forces R&D departments to design synthetic routes around available reactive substrates rather than optimal therapeutic candidates, often leading to longer, more expensive linear syntheses. Furthermore, existing methods frequently suffer from poor regioselectivity and stereoselectivity, resulting in complex mixtures of E/Z isomers and regioisomers that are notoriously difficult to separate. The energy intensity of these traditional processes is also a concern, as they often require elevated temperatures or stoichiometric amounts of corrosive reagents, creating safety hazards and increasing the environmental footprint of the manufacturing facility.
The Novel Approach
The methodology disclosed in the patent represents a paradigm shift by employing a cooperative catalytic system comprising both a protonic acid and a Lewis acid. This dual-activation strategy activates the alkyne moiety sufficiently to react with simple substituted benzenes, including those with only single substituents, thereby vastly expanding the substrate scope. The reaction proceeds under remarkably mild conditions, typically between 0 and 60 degrees Celsius, which preserves sensitive functional groups that would otherwise degrade under harsher traditional conditions. 
This approach not only simplifies the operational complexity but also enhances the economic viability of the process by reducing catalyst loading to mere mole percentages. The result is a streamlined workflow that delivers high yields with excellent stereocontrol, effectively solving the selectivity issues that have hindered the field for decades. For procurement managers, this means access to a more versatile and cost-effective supply source for complex aryl ethylene derivatives, reducing the risk of supply chain disruptions caused by difficult-to-source precursors.
Mechanistic Insights into Dual-Acid Catalyzed Hydroarylation
The success of this synthesis hinges on the precise interplay between the Lewis acid and the protonic acid within the reaction medium. The Lewis acid, such as scandium trifluoromethanesulfonate, coordinates with the alkyne triple bond, increasing its electrophilicity and rendering it susceptible to nucleophilic attack by the aromatic ring. Simultaneously, the protonic acid, such as trifluoromethanesulfonic acid, likely stabilizes the developing carbocation intermediate or assists in the proton transfer steps necessary to restore aromaticity. This synergistic activation lowers the activation energy barrier significantly, allowing the reaction to proceed rapidly at room temperature or slightly elevated temperatures. The specific choice of catalyst pair is critical; the patent highlights combinations like Sc(OTf)3 with TfOH as particularly effective, suggesting a hard-soft acid-base matching that optimizes the transition state geometry for the desired Z-isomer formation.
From an impurity control perspective, the high stereoselectivity observed (often Z/E > 20:1) indicates that the transition state is highly organized, likely due to steric interactions directed by the catalyst coordination sphere. This minimizes the formation of the thermodynamically stable but undesired E-isomer, which is a common byproduct in non-catalyzed thermal reactions. The ability to tolerate diverse functional groups, including halogens like bromine and chlorine as well as methoxy groups, further underscores the chemoselectivity of the system. 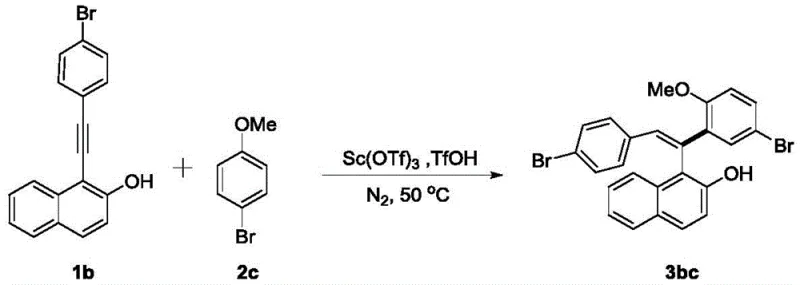
For the R&D Director, understanding this mechanism provides confidence in the robustness of the process during scale-up, as the catalyst system appears resilient to minor variations in substrate electronics. The mild conditions also imply that side reactions such as polymerization or over-alkylation are effectively suppressed, leading to a cleaner crude profile and simplifying the final purification steps.
How to Synthesize Triaryl Ethylene Efficiently
The practical execution of this synthesis is designed for reproducibility and ease of handling in a standard laboratory or pilot plant setting. The procedure involves a straightforward sequential addition of reagents under an inert atmosphere to prevent moisture sensitivity issues associated with strong Lewis acids. Following the reaction, the workup is uncomplicated, relying on standard silica gel chromatography which is familiar to process chemists worldwide.
- Under an inert gas atmosphere, sequentially add diaryl acetylene, substituted benzene, protonic acid, and Lewis acid into a reactor.
- Stir the reaction mixture at a controlled temperature ranging from 0 to 60 degrees Celsius for a duration of 5 to 300 minutes.
- Upon completion, purify the crude mixture using column chromatography with a petroleum ether and ethyl acetate solvent system to isolate the pure triaryl ethylene product.
Commercial Advantages for Procurement and Supply Chain Teams
Adopting this dual-acid catalytic technology offers profound strategic benefits for supply chain optimization and cost management in the fine chemical sector. The shift from stoichiometric reagents to catalytic amounts of commercially available Lewis and Bronsted acids fundamentally alters the cost structure of the synthesis. By eliminating the need for expensive, pre-functionalized starting materials that were previously required to drive the reaction, the raw material costs are significantly reduced. Furthermore, the mild reaction temperatures translate directly into lower energy consumption for heating and cooling systems, contributing to a greener and more sustainable manufacturing profile that aligns with modern corporate responsibility goals.
- Cost Reduction in Manufacturing: The utilization of low-loading catalysts eliminates the need for costly transition metals or harsh reagents that require specialized disposal. The high atom economy and reduced waste generation mean that the overall cost per kilogram of the active pharmaceutical ingredient precursor is drastically lowered. Additionally, the simplified purification process reduces solvent consumption and labor hours associated with complex separation techniques, driving further operational efficiencies.
- Enhanced Supply Chain Reliability: The broad substrate scope allows for the use of commodity chemicals like substituted benzenes and diaryl acetylenes which are readily available from multiple global suppliers. This diversification of the raw material base mitigates the risk of supply shortages that often plague niche intermediate markets. The robustness of the reaction conditions ensures consistent batch-to-batch quality, reducing the likelihood of production delays caused by failed runs or out-of-specification results.
- Scalability and Environmental Compliance: The absence of extreme temperatures and pressures makes this process inherently safer and easier to scale from gram to ton quantities without requiring specialized high-pressure reactors. The reduced generation of hazardous byproducts simplifies wastewater treatment and waste disposal compliance, lowering the environmental overhead costs for the manufacturing site. This scalability ensures that the supply chain can respond agilely to market demand surges for critical drugs like tamoxifen.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for decision-making.
Q: What are the key advantages of the dual-acid catalytic system over traditional Friedel-Crafts methods?
A: The dual-acid system utilizing both Lewis and Bronsted acids significantly enhances regioselectivity and stereoselectivity compared to conventional methods which often require electron-rich substrates. It allows for the use of simpler aromatic hydrocarbons with single substituents and operates under much milder conditions.
Q: What is the typical stereoselectivity (Z/E ratio) achieved in this synthesis?
A: The method demonstrates excellent stereoselectivity, consistently achieving Z/E ratios greater than 19:1, and in many optimized examples exceeding 20:1, ensuring high purity of the desired isomer.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process is highly scalable due to its mild reaction temperatures (0-60°C), low catalyst loading, and straightforward post-treatment involving standard column chromatography, making it viable for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Triaryl Ethylene Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the development of life-saving therapies and advanced materials. Our technical team has extensively analyzed the dual-acid catalytic pathway described in CN112811986A and possesses the expertise to implement this cutting-edge chemistry at an industrial scale. We boast extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and reliability. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of triaryl ethylene intermediate meets the exacting standards required for pharmaceutical registration.
We invite you to collaborate with us to leverage this innovative synthesis route for your specific project requirements. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume needs, demonstrating how this efficient catalytic method can optimize your budget. Please contact us today to request specific COA data and route feasibility assessments, and let us partner with you to accelerate your drug development timeline while maximizing value.
