Advanced Scandium Catalysis for Efficient Synthesis of Sulfur-Containing Pharmaceutical Intermediates
Advanced Scandium Catalysis for Efficient Synthesis of Sulfur-Containing Pharmaceutical Intermediates
The development of efficient methodologies for constructing carbon-sulfur bonds is a cornerstone of modern medicinal chemistry, particularly for the synthesis of bioactive sulfur-containing natural products and pharmaceutical agents. A significant breakthrough in this domain is detailed in patent CN109096162B, which discloses a novel scandium-catalyzed nucleophilic addition reaction of mercaptans to o-methylene benzoquinones. This technology addresses critical bottlenecks in traditional synthetic routes by utilizing trivalent scandium catalysts to facilitate thia-Michael additions under remarkably mild and neutral conditions. For R&D directors and process chemists, this represents a paradigm shift away from harsh acidic or basic protocols that often compromise sensitive functional groups. The ability to generate o-methylene benzoquinone intermediates in situ and immediately trap them with thiols offers a streamlined pathway to complex molecular architectures, ensuring high purity and operational simplicity that is essential for reliable pharmaceutical intermediate supplier operations.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the preparation and utilization of o-methylene benzoquinone compounds have been fraught with synthetic challenges that hinder efficient manufacturing. Traditional methods for generating these reactive intermediates often rely on pyrolysis, photochemical reactions, or the use of strong acidic and basic conditions, all of which impose severe limitations on process scalability and substrate tolerance. Specifically, conventional thia-Michael addition reactions frequently necessitate the use of expensive precious metal catalysts such as gold or ruthenium complexes, or harsh Brønsted acids and bases that require rigorous pH control. These aggressive conditions are incompatible with acid- or base-sensitive functional groups, leading to decomposition or side reactions that drastically reduce yield. Furthermore, the post-reaction processing in traditional methods is cumbersome, often requiring extensive neutralization steps to bring the system to neutrality before isolation, which increases waste generation and operational costs in cost reduction in API manufacturing initiatives.
The Novel Approach
In stark contrast, the methodology described in the patent introduces a robust scandium-catalyzed system that operates effectively under neutral conditions, thereby preserving the integrity of sensitive functional groups on the substrate. By employing Sc(III) catalysts, such as scandium trifluoromethanesulfonate, the reaction proceeds smoothly at moderate temperatures ranging from 30°C to 70°C, eliminating the need for energy-intensive heating or cryogenic cooling. This approach not only simplifies the reaction setup but also significantly enhances the safety profile of the manufacturing process. The in situ generation of the o-methylene benzoquinone intermediate ensures that the highly reactive species is immediately consumed by the nucleophilic thiol, minimizing polymerization or degradation pathways. This results in consistently high yields, reported between 82% and 95%, and facilitates a much cleaner reaction profile that reduces the burden on downstream purification processes, making it an attractive option for commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Scandium-Catalyzed Nucleophilic Addition
The core of this technological advancement lies in the unique Lewis acidity of the trivalent scandium ion, which activates the hydroxyl group of the 2-(hydroxy(phenyl)methyl)phenol precursor to facilitate dehydration and the formation of the electrophilic o-methylene benzoquinone (o-QM) species. Unlike traditional Brønsted acids that protonate the substrate indiscriminately, the scandium catalyst coordinates specifically to the oxygen atoms, lowering the activation energy for the elimination of water without creating a highly acidic bulk environment. Once the o-QM intermediate is generated, the scandium center continues to play a pivotal role by activating the quinone methide double bond towards nucleophilic attack. This dual activation mode ensures that the thiol nucleophile adds efficiently to the exocyclic methylene group, forming the new carbon-sulfur bond with high regioselectivity. The catalytic cycle is sustained as the scandium species is regenerated upon product release, allowing for low catalyst loadings (0.05 to 0.2 equivalents) to drive the reaction to completion.
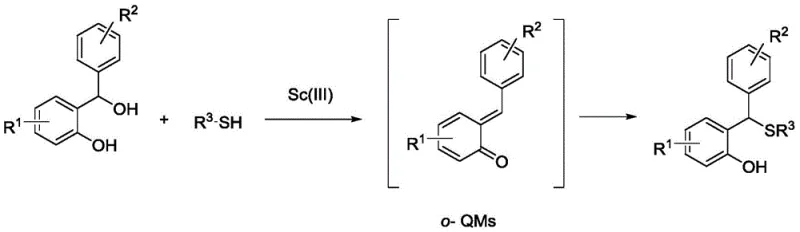
Understanding the impurity profile is crucial for R&D teams aiming to implement this chemistry for high-purity OLED material or API synthesis. The neutral nature of the reaction medium significantly suppresses common side reactions such as self-polymerization of the o-QM intermediate or hydrolysis of sensitive ester or amide groups that might be present on the aromatic rings. In traditional acid-catalyzed pathways, these side reactions often lead to complex impurity spectra that are difficult to separate. However, the scandium-catalyzed route produces a cleaner crude reaction mixture, where the primary byproduct is simply water. This simplifies the impurity landscape, allowing for straightforward purification via standard silica gel column chromatography using non-polar eluents like petroleum ether. The result is a product with superior chemical purity, meeting the stringent specifications required for regulatory submissions and reducing the risk of batch failures due to trace impurities.
How to Synthesize 2-(Phenyl(phenylthio)methyl)phenol Derivatives Efficiently
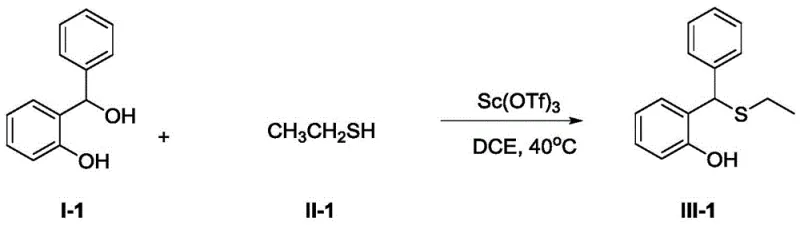
To implement this synthesis effectively, process chemists should focus on optimizing the stoichiometry and solvent choice to maximize throughput while maintaining safety. The patent outlines a general procedure where the phenolic substrate and the thiol are dissolved in a solvent such as 1,2-dichloroethane (DCE) or DMF, followed by the addition of the scandium catalyst. The reaction is then stirred at mild temperatures, typically around 30°C to 50°C, for a duration of 5 to 10 hours. This operational window provides flexibility for scaling, as the exotherm is manageable and the reaction time is compatible with standard batch cycles. For a concrete example of this protocol in action, consider the synthesis of the ethyl-thio derivative shown below, which achieved a 92% isolated yield under optimized conditions.
- Dissolve the 2-(hydroxy(phenyl)methyl)phenol substrate and the thiol compound in a suitable solvent such as 1,2-dichloroethane or DMF.
- Add the Scandium(III) catalyst, preferably Scandium Triflate, to the reaction mixture maintaining a molar ratio of approximately 1: 2:0.1.
- Stir the reaction at mild temperatures between 30°C and 70°C for 5 to 10 hours, followed by standard aqueous workup and silica gel chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this scandium-catalyzed methodology offers tangible strategic benefits beyond mere chemical efficiency. The shift away from precious metal catalysts like gold or ruthenium, which are subject to volatile market pricing and supply constraints, to earth-abundant scandium salts represents a significant stabilization of raw material costs. While scandium is a rare earth element, the catalyst loadings required are extremely low, and the potential for catalyst recovery and recycling in continuous flow setups further enhances the economic viability. Moreover, the elimination of harsh acids and bases removes the need for expensive corrosion-resistant equipment and reduces the consumption of neutralizing agents, leading to substantial cost savings in waste treatment and disposal. This aligns perfectly with global trends towards greener chemistry and sustainable manufacturing practices.
- Cost Reduction in Manufacturing: The economic argument for adopting this technology is compelling when analyzing the total cost of ownership. By replacing expensive precious metal catalysts with more affordable scandium salts and eliminating the multi-step neutralization and washing procedures associated with strong acid/base catalysis, the overall processing time and resource consumption are drastically reduced. The high yields (82-95%) directly translate to better atom economy and less raw material waste, which is a critical factor in reducing the cost of goods sold (COGS) for high-value intermediates. Additionally, the simplified workup procedure reduces labor hours and solvent usage, contributing to a leaner and more cost-effective production model.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of readily available and stable starting materials. The 2-(hydroxy(phenyl)methyl)phenol precursors and various alkyl or benzyl thiols are commodity chemicals with robust global supply networks, minimizing the risk of shortages. The mild reaction conditions also mean that the process is less sensitive to minor fluctuations in utility supplies, such as steam or chilled water, ensuring consistent production output. This reliability is essential for maintaining just-in-time delivery schedules for downstream pharmaceutical customers who depend on uninterrupted supply chains for their own manufacturing timelines.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, this method is exceptionally well-suited for large-scale production. The absence of hazardous reagents and the generation of minimal waste simplify the regulatory compliance landscape, making it easier to obtain necessary permits for expansion. The reaction's tolerance to a wide range of functional groups means that the same platform technology can be applied to synthesize a diverse library of analogs without needing to re-validate entirely new processes. This flexibility accelerates time-to-market for new drug candidates and supports the agile manufacturing needs of modern pharmaceutical development.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this scandium-catalyzed technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a clear understanding of the process capabilities and limitations for potential partners.
Q: What are the advantages of using Scandium catalysts over traditional acid-base catalysts for this reaction?
A: Scandium catalysts operate under neutral conditions, avoiding the harsh acidic or basic environments that can degrade sensitive functional groups. This eliminates the need for complex neutralization steps during workup, simplifying purification and improving overall yield.
Q: Can this method be scaled for commercial production of API intermediates?
A: Yes, the reaction utilizes mild temperatures (30-70°C) and common solvents like DCE or DMF, which are highly compatible with large-scale reactor operations. The high yields (82-95%) and simple workup procedures make it ideal for commercial scale-up.
Q: What types of thiol substrates are compatible with this Scandium-catalyzed protocol?
A: The protocol demonstrates broad substrate scope, successfully reacting with various alkyl thiols (ethyl, propyl, isobutyl) and benzyl thiols. This versatility allows for the synthesis of diverse sulfur-containing scaffolds required in drug discovery.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-(Phenyl(phenylthio)methyl)phenol Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic technologies like the scandium-mediated thia-Michael addition in accelerating drug discovery and development. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from bench-scale innovation to industrial reality is seamless. Our state-of-the-art facilities are equipped to handle air- and moisture-sensitive catalytic reactions with precision, and our rigorous QC labs enforce stringent purity specifications to guarantee that every batch meets the highest international standards. We are committed to leveraging this efficient synthetic route to deliver high-quality sulfur-containing intermediates that empower your pharmaceutical pipelines.
We invite you to collaborate with us to explore how this technology can optimize your specific project requirements. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your target molecule, demonstrating exactly how this method can improve your margins. Please contact our technical procurement team today to request specific COA data for our catalog compounds or to discuss route feasibility assessments for your custom synthesis needs. Let us be your partner in turning complex chemical challenges into commercial successes.
