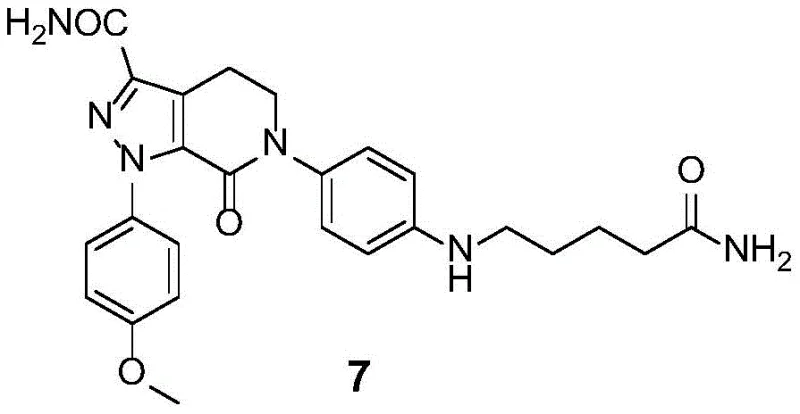
Advanced 4-Step Synthesis of Apixaban Impurity 7 for High-Purity Quality Control Standards
The pharmaceutical industry's relentless pursuit of drug safety and efficacy has placed impurity profiling at the forefront of regulatory compliance, particularly for potent anticoagulants like Apixaban. Patent CN107722007B introduces a groundbreaking preparation method for Apixaban Impurity 7, chemically defined as 6-(4-((5-amino-5-oxopentyl)amino)phenyl)-1-(4-methoxyphenyl)-7-oxo-4,5,6,7-tetrahydro-1H-pyrazolo[3,4-c]pyridine-3-carboxamide. This specific degradation product, formed during the alkaline decomposition of the active pharmaceutical ingredient, poses significant analytical challenges due to its structural similarity to the parent molecule. The patented technology offers a robust, four-step synthetic pathway that bypasses the limitations of isolation from drug substance, enabling the production of high-purity reference standards essential for rigorous quality control and method validation.
For R&D directors and quality assurance teams, the availability of such well-characterized impurities is not merely a regulatory checkbox but a critical component of patient safety strategies. The ability to synthesize this complex heterocyclic compound from simple, commercially available precursors ensures a consistent supply of reference materials, facilitating accurate quantification of trace impurities in final drug products. This technical advancement underscores the importance of developing orthogonal synthetic routes to access specific degradation products that are otherwise inaccessible through standard manufacturing processes.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, obtaining specific degradation impurities like Compound 7 involved subjecting the finished Apixaban API to forced degradation conditions, typically using high concentrations of alkali over extended periods. This approach is fraught with inefficiencies; the yield of the specific impurity is often negligible, and the reaction mixture becomes a complex soup of various degradation products. Separating the target impurity from the parent drug and other by-products is notoriously difficult because their physicochemical properties, such as polarity and solubility, are nearly identical. Standard purification techniques like recrystallization often fail, and preparative HPLC is cost-prohibitive for generating the milligram-to-gram quantities required for comprehensive toxicological and analytical studies.
The Novel Approach
The methodology disclosed in CN107722007B revolutionizes this process by constructing the molecule from the ground up rather than breaking it down. By starting with a substituted piperidinone and a halogenated ester, the synthesis builds the carbon skeleton precisely where needed. This bottom-up strategy allows for the introduction of functional groups in a controlled sequence, minimizing the formation of unwanted side products. The route employs standard organic transformations—alkylation, protection, condensation, and cyclization—that are well-understood and easily optimized. This shift from isolation to total synthesis transforms the production of reference standards from a bottleneck into a reliable, scalable operation.
Mechanistic Insights into the 4-Step Synthetic Cascade
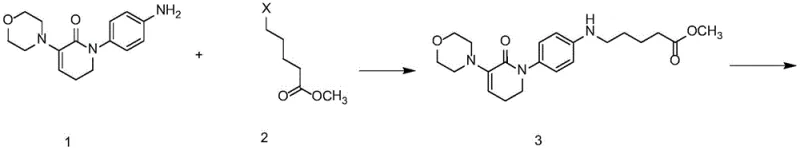
The synthetic journey begins with the N-alkylation of a tetrahydropyridinone derivative (Compound 1) using a halo-ester (Compound 2). This nucleophilic substitution is driven by a base such as triethylamine or cesium carbonate in polar aprotic solvents like DMF or acetonitrile. The choice of base and solvent is critical here; stronger bases like cesium carbonate can enhance the nucleophilicity of the nitrogen atom, ensuring complete conversion even with less reactive chloro-esters. Following this, the secondary amine generated in situ is immediately protected with a Boc group (di-tert-butyl dicarbonate). This protection step is mechanistically vital as it prevents the nitrogen from interfering in subsequent cyclization reactions, thereby directing the regioselectivity of the ring closure in later stages.
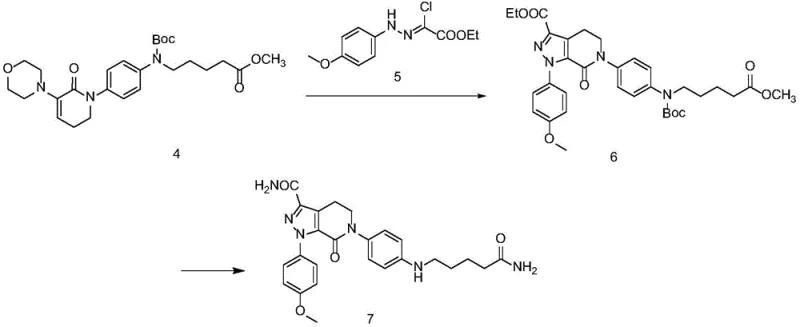
The core of the molecular architecture is assembled in the third step, where the protected intermediate reacts with a hydrazine derivative (Compound 5). This condensation reaction, facilitated by heating in toluene or ethyl acetate, constructs the fused pyrazolo[3,4-c]pyridine system. The mechanism likely involves the formation of a hydrazone intermediate followed by intramolecular cyclization and aromatization. Finally, the terminal carboxamide group is installed via a reaction with formamide in the presence of a metal alkoxide. This step effectively converts an ester or nitrile precursor into the primary amide functionality found in the final impurity, completing the synthesis of the complex heterocyclic scaffold with high fidelity.
How to Synthesize Apixaban Impurity 7 Efficiently
The patented process outlines a streamlined workflow that balances reaction efficiency with ease of purification. The initial alkylation is performed at elevated temperatures (80-90°C) to drive the reaction to completion, followed by a simple aqueous workup to isolate the intermediate. The subsequent protection and cyclization steps utilize mild conditions that preserve the integrity of the sensitive ester and amide functionalities. For the final amidation, the use of formamide serves a dual purpose as both a reagent and a solvent, simplifying the reaction setup. Detailed standardized synthetic steps see the guide below.
- Perform N-alkylation of the piperidinone precursor (Compound 1) with a halogenated ester (Compound 2) using a base like triethylamine or potassium carbonate in DMF or toluene to yield Compound 3.
- Protect the secondary amine of Compound 3 using di-tert-butyl dicarbonate (Boc2O) in ethyl acetate or dichloromethane to obtain the protected intermediate Compound 4.
- React Compound 4 with the hydrazine derivative (Compound 5) under basic conditions followed by acidification to construct the pyrazolo[3,4-c]pyridine core, yielding Compound 6.
- Execute the final amidation cyclization by reacting Compound 6 with formamide and a metal alkoxide base (e.g., sodium methoxide) to generate the target Apixaban Impurity 7.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, this synthetic route offers substantial advantages by decoupling the supply of reference standards from the supply of the expensive finished API. The starting materials, including substituted piperidinones and halo-esters, are commodity chemicals available from multiple global suppliers, reducing the risk of single-source dependency. Furthermore, the reaction conditions do not require exotic catalysts or cryogenic temperatures, allowing the synthesis to be performed in standard multipurpose reactors found in most CDMO facilities. This accessibility translates directly into lower manufacturing costs and more stable pricing for high-purity reference materials.
- Cost Reduction in Manufacturing: The elimination of expensive API degradation and complex chromatographic purification significantly lowers the cost basis. By using robust, high-yielding steps with inexpensive reagents like formamide and Boc anhydride, the overall process economics are optimized. The ability to purify intermediates via simple filtration or extraction before the final step reduces the load on the final purification stage, saving on silica gel and solvent consumption.
- Enhanced Supply Chain Reliability: Because the synthesis relies on stable, shelf-stable intermediates and common solvents, the supply chain is far more resilient to disruptions. There is no need to coordinate the degradation of batch-specific API, which can introduce variability and delays. The modular nature of the four-step synthesis allows for flexible production scheduling, ensuring that reference standards can be produced on demand to meet tight regulatory submission deadlines.
- Scalability and Environmental Compliance: The process utilizes solvents such as ethyl acetate, toluene, and ethanol, which are easier to recover and recycle compared to more hazardous alternatives. The absence of heavy metal catalysts simplifies waste treatment and disposal, aligning with modern green chemistry principles. The scalability of the alkylation and cyclization steps means that production can be easily ramped up from laboratory to pilot scale without significant re-engineering of the process parameters.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of Apixaban Impurity 7. These insights are derived directly from the experimental data and claims within the patent documentation, providing clarity on the feasibility and benefits of this novel synthetic approach for industry stakeholders.
Q: Why is a dedicated synthesis route preferred over isolating Impurity 7 from Apixaban degradation?
A: Isolating Impurity 7 from the alkaline decomposition of Apixaban is inefficient because the impurity levels are low and its structure is highly similar to the parent drug, making separation via chromatography or recrystallization extremely difficult. The dedicated synthetic route described in patent CN107722007B allows for the production of gram-to-kilogram quantities of high-purity material specifically for use as a qualified reference substance.
Q: What are the critical reaction conditions for the final cyclization step?
A: The final conversion of Compound 6 to Compound 7 requires heating with formamide in the presence of a strong metal alkoxide base, such as sodium methoxide or potassium tert-butoxide. The reaction is typically conducted in polar aprotic solvents like DMF or DMAc at moderate temperatures (50-60°C) to ensure complete cyclization and amidation without degrading the sensitive heterocyclic core.
Q: How does this synthesis method impact the cost of quality control materials?
A: By utilizing readily available starting materials like methyl 5-bromovalerate and standard protecting group chemistry, this method avoids the high costs associated with degrading expensive finished API. The use of common solvents and straightforward workup procedures, such as filtration and standard column chromatography, significantly lowers the operational expenditure for producing certified reference materials.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Apixaban Impurity 7 Supplier
At NINGBO INNO PHARMCHEM, we understand that the integrity of your analytical data depends on the quality of your reference standards. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that even complex impurities like Compound 7 are synthesized with the utmost precision. We operate stringent purity specifications and maintain rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify the identity and purity of every batch, guaranteeing that our materials meet the exacting demands of global regulatory agencies.
We invite you to collaborate with us to secure a stable supply of this critical reference material. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to support your drug development and quality control initiatives, ensuring your project moves forward without interruption.
