Advanced Photocatalytic Synthesis of N-(1-phenyl-2-(phenylseleno)ethyl)aniline Derivatives for Commercial Scale-up
Advanced Photocatalytic Synthesis of N-(1-phenyl-2-(phenylseleno)ethyl)aniline Derivatives for Commercial Scale-up
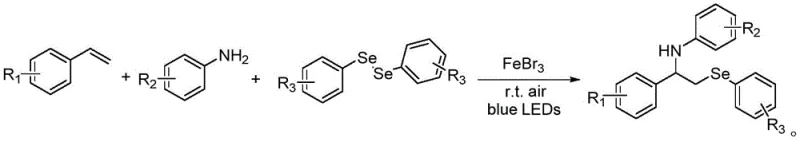
The pharmaceutical and fine chemical industries are constantly seeking greener, more efficient pathways to access complex organic scaffolds, particularly those containing heteroatoms like selenium which impart significant biological activity. Patent CN110305054B introduces a groundbreaking methodology for the preparation of double-substituted styrene derivatives, specifically focusing on the synthesis of N-(1-phenyl-2-(phenylseleno)ethyl)aniline derivatives. This technology represents a paradigm shift from traditional thermochemical processes to a visible-light-driven protocol that operates under remarkably mild conditions. By leveraging the unique properties of iron-based photocatalysis, this invention addresses critical pain points in modern organic synthesis, including the reliance on toxic heavy metals, excessive energy consumption, and multi-step procedures that erode overall process efficiency. For R&D directors and procurement specialists alike, this patent offers a compelling value proposition: a streamlined, one-step approach that transforms readily available olefins, anilines, and diselenides into high-value intermediates with impressive atom economy.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the functionalization of olefins to introduce both nitrogen and selenium functionalities has been fraught with significant technical and economic challenges. Traditional synthetic routes often necessitate the use of stoichiometric amounts of hazardous reagents or rely on precious metal catalysts such as palladium, platinum, or rhodium, which not only drive up raw material costs but also introduce stringent requirements for residual metal removal in pharmaceutical applications. Furthermore, these conventional methods frequently demand harsh reaction conditions, including elevated temperatures and high pressures, which increase the risk of thermal runaway and limit the compatibility of sensitive functional groups. The multi-step nature of many legacy syntheses results in cumulative yield losses and generates substantial volumes of chemical waste, creating a heavy burden on environmental compliance and waste treatment infrastructure. Additionally, the poor selectivity often observed in thermal radical reactions can lead to complex impurity profiles that are difficult and costly to separate, ultimately compromising the purity specifications required for downstream drug development.
The Novel Approach
In stark contrast to these archaic methodologies, the technology disclosed in CN110305054B utilizes a visible-light-mediated strategy that operates at room temperature, fundamentally altering the energy profile of the synthesis. By employing blue LEDs as the energy source, the process activates the selenium-selenium bond in diphenyl diselenide through a photocatalytic cycle mediated by Iron(III) bromide (FeBr3), a cheap and abundant earth metal. This approach eliminates the need for external heating, thereby drastically reducing the carbon footprint and operational hazards associated with high-temperature reactors. The reaction is conducted in environmentally benign solvents like ethyl acetate, further enhancing the green chemistry credentials of the process. Most importantly, this method achieves the simultaneous formation of C-Se and C-N bonds in a single operational step, bypassing the need for protecting group strategies or sequential functionalization. This consolidation of steps not only accelerates the timeline from bench to kilogram scale but also significantly improves the overall mass balance of the production process.
Mechanistic Insights into FeBr3-Catalyzed Visible Light Selenylation
The core of this innovation lies in the intricate interplay between the iron catalyst and the photon flux provided by the blue LEDs. Upon irradiation, the FeBr3 catalyst facilitates the homolytic cleavage of the relatively weak Se-Se bond in the diphenyl diselenide precursor, generating highly reactive phenylselenyl radicals. These radicals add regioselectively to the electron-rich double bond of the styrene derivative, forming a stabilized benzylic radical intermediate. This transient species is then intercepted by the nucleophilic aniline component, leading to the formation of the final C-N bond and regenerating the catalytic species or closing the redox cycle. The use of iron as a Lewis acid also helps in activating the amine nucleophile, ensuring high conversion rates even with less reactive aniline derivatives. This mechanistic pathway avoids the high-energy transition states typical of thermal radical initiators, allowing the reaction to proceed with exceptional chemoselectivity. The mildness of the radical generation prevents unwanted side reactions such as olefin polymerization or over-oxidation, which are common pitfalls in traditional free-radical chemistry.
From an impurity control perspective, the room temperature operation is a decisive factor in maintaining a clean reaction profile. High-temperature processes often promote degradation pathways and the formation of tarry by-products that complicate purification. By keeping the thermal energy input minimal, the patent ensures that the primary reaction channel dominates, resulting in crude products with high purity levels, often exceeding 99% as demonstrated in the experimental examples. The specific choice of ethyl acetate as a solvent further aids in impurity management; it provides excellent solubility for the organic reactants while being easily removable during the workup phase. The subsequent purification via silica gel column chromatography using a petroleum ether and ethyl acetate gradient is highly effective at separating any minor regioisomers or unreacted starting materials. This robust control over the impurity spectrum is critical for pharmaceutical intermediates, where strict limits on genotoxic impurities and heavy metals are enforced by regulatory bodies globally.
How to Synthesize N-(1-phenyl-2-(phenylseleno)ethyl)aniline Efficiently
The practical implementation of this synthesis is designed for ease of operation, making it highly attractive for process chemistry teams looking to rapidly prototype new analogs or scale up production. The protocol involves a straightforward mixing of reagents followed by a passive irradiation period, requiring minimal active intervention from laboratory personnel. The simplicity of the workup procedure, involving only solvent evaporation and standard chromatography, means that the technology can be transferred from R&D to pilot plant with minimal re-engineering. For detailed operational parameters, stoichiometry, and specific purification gradients, please refer to the standardized synthesis guide below which encapsulates the optimal conditions derived from the patent examples.
- Dissolve styrene derivatives, aniline compounds, diphenyl diselenide, and FeBr3 photocatalyst in ethyl acetate solvent at room temperature under nitrogen or oxygen atmosphere.
- Expose the reaction mixture to blue LED light irradiation for approximately 24 hours to facilitate the radical selenylation and amination process.
- Upon completion, remove the solvent via rotary evaporation and purify the crude product using silica gel column chromatography with a petroleum ether and ethyl acetate gradient.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this photocatalytic technology translates into tangible strategic advantages that extend beyond mere technical feasibility. The shift away from precious metal catalysts to iron-based systems represents a significant decoupling from the volatile pricing dynamics of the noble metal market. Furthermore, the simplification of the synthetic route reduces the number of unit operations required, directly correlating to lower labor costs and reduced equipment occupancy time. The ability to run reactions at ambient temperature also alleviates pressure on facility cooling and heating utilities, contributing to a leaner operational expenditure model. These factors combined create a resilient supply chain capable of responding quickly to market demands without being bottlenecked by complex manufacturing constraints or scarce raw materials.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts such as palladium or ruthenium removes a major cost driver from the bill of materials, while the use of commodity chemicals like styrene and aniline ensures stable raw material pricing. The one-step nature of the reaction significantly reduces solvent consumption and waste disposal costs associated with multi-step sequences, leading to substantial overall cost savings in the manufacturing of these complex organoselenium intermediates. Additionally, the energy efficiency gained by operating at room temperature rather than under reflux conditions further drives down the utility costs per kilogram of product produced.
- Enhanced Supply Chain Reliability: The starting materials required for this synthesis, including various substituted styrenes, anilines, and diphenyl diselenide, are commercially available in bulk quantities from multiple global suppliers, mitigating the risk of single-source dependency. The robustness of the reaction conditions means that production is less susceptible to disruptions caused by equipment failure or utility fluctuations, ensuring a consistent and reliable output of high-purity intermediates. This reliability is crucial for maintaining continuous supply to downstream pharmaceutical customers who require just-in-time delivery schedules for their drug development programs.
- Scalability and Environmental Compliance: The photochemical nature of the reaction is inherently scalable using modern flow chemistry reactors or large-scale batch photoreactors, allowing for seamless transition from gram-scale discovery to tonnage-level commercial production. The use of green solvents like ethyl acetate and the absence of toxic heavy metal residues simplify the environmental permitting process and reduce the burden on wastewater treatment facilities. This alignment with green chemistry principles not only future-proofs the manufacturing process against tightening environmental regulations but also enhances the corporate sustainability profile of the production entity.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this patented synthesis method. These answers are derived directly from the experimental data and claims within the patent documentation to provide accurate guidance for potential licensees or manufacturing partners. Understanding these nuances is essential for evaluating the fit of this technology within your existing portfolio of synthetic capabilities.
Q: What catalyst system is used in this patented synthesis?
A: The process utilizes Iron(III) bromide (FeBr3) as a cost-effective and earth-abundant photocatalyst, replacing expensive noble metals like palladium or ruthenium typically found in traditional cross-coupling reactions.
Q: What are the reaction conditions regarding temperature and light source?
A: The reaction proceeds efficiently at room temperature (r.t.) under the irradiation of blue LEDs, eliminating the need for high-temperature heating and significantly reducing energy consumption.
Q: What yields can be expected from this methodology?
A: Experimental data indicates variable but robust yields depending on substituents, ranging from moderate levels around 30% for sterically hindered substrates to excellent yields exceeding 80% for standard styrene and aniline combinations.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Disubstituted Styrene Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the photocatalytic synthesis described in CN110305054B for the production of high-value organoselenium intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from laboratory curiosity to market-ready product is seamless and efficient. Our state-of-the-art facilities are equipped with advanced photochemical reactors and rigorous QC labs capable of meeting the most stringent purity specifications required by the global pharmaceutical industry. We are committed to delivering not just a chemical product, but a comprehensive solution that optimizes your supply chain and accelerates your time to market.
We invite you to engage with our technical team to explore how this innovative iron-catalyzed route can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of switching to this greener methodology. Our experts are ready to provide specific COA data and route feasibility assessments to demonstrate how we can support your development goals with reliability and precision. Contact our technical procurement team today to initiate a discussion on scaling these valuable disubstituted styrene derivatives for your next commercial campaign.
