Scalable Metal-Free Synthesis of Fluorinated Indenoindoles for Advanced Pharmaceutical Applications
Scalable Metal-Free Synthesis of Fluorinated Indenoindoles for Advanced Pharmaceutical Applications
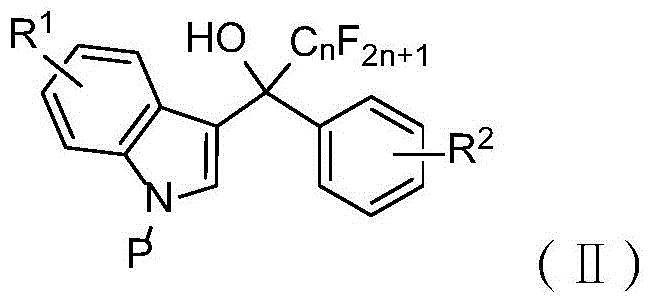
The development of efficient synthetic routes for fluorinated heterocycles remains a critical challenge in modern medicinal chemistry and material science. Patent CN116063223A introduces a groundbreaking methodology for the synthesis of 10-perfluoroalkyl-5,10-dihydroindeno[1,2-b]indole compounds, a privileged scaffold known for its potent biological activities ranging from anticancer properties to kinase inhibition. This technology leverages a metal-free Bronsted acid catalytic system that fundamentally shifts the paradigm from traditional transition-metal dependent processes to a more sustainable and cost-effective approach. By utilizing trifluoromethanesulfonic acid (TfOH) in hexafluoroisopropanol (HFIP), the process achieves exceptional atom economy and operational simplicity. The structural versatility of these compounds, as illustrated in the general formula below, allows for extensive functionalization, making them invaluable building blocks for next-generation therapeutics and organic electronic materials.
![General chemical structure of 10-perfluoroalkyl-5,10-dihydroindeno[1,2-b]indole compounds showing variable R groups and perfluoroalkyl chains](/insights/img/perfluoroalkyl-indenoindole-synthesis-pharma-supplier-20260303020256-02.png)
For procurement specialists and supply chain managers, the implications of this patent extend beyond mere chemical novelty; it represents a tangible opportunity for cost reduction in pharmaceutical intermediate manufacturing. The elimination of precious metal catalysts not only lowers raw material costs but also drastically simplifies the downstream purification process by removing the need for rigorous heavy metal scavenging steps. Furthermore, the reaction conditions are remarkably mild, typically proceeding at room temperature, which reduces energy consumption and enhances safety profiles in large-scale reactors. The robustness of this synthetic route is evidenced by its wide substrate tolerance, accommodating electron-withdrawing and electron-donating groups alike, thereby ensuring a reliable supply of diverse analogues for drug discovery pipelines without the need for complex process re-optimization for each new derivative.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the indeno[1,2-b]indole core has relied heavily on transition-metal catalyzed cyclizations, such as palladium or rhodium-mediated processes, which present significant logistical and economic hurdles for industrial adoption. These conventional methods often require stringent anhydrous conditions, elevated temperatures, and expensive ligand systems that drive up the overall cost of goods sold (COGS). Moreover, the use of heavy metals introduces severe regulatory burdens regarding residual metal limits in active pharmaceutical ingredients (APIs), necessitating additional purification stages that reduce overall yield and increase waste generation. Traditional approaches like the Japp–Klingemann reaction or nitro-promoted reductions often suffer from poor atom economy and generate stoichiometric amounts of hazardous by-products, complicating waste disposal and environmental compliance. The multi-step nature of many prior art syntheses also increases the lead time for high-purity pharmaceutical intermediates, creating bottlenecks in the supply chain that can delay critical clinical trials.
The Novel Approach
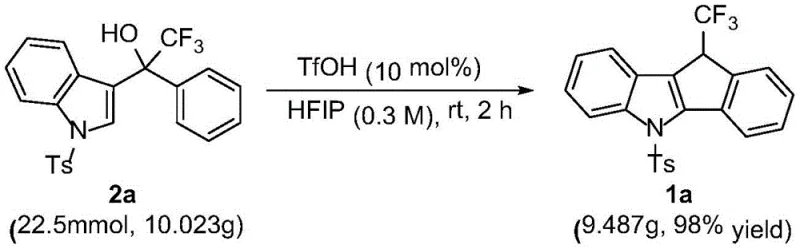
In stark contrast, the methodology disclosed in CN116063223A utilizes a simple yet highly effective Bronsted acid catalysis strategy that circumvents the drawbacks of metal-based systems. The core transformation involves the intramolecular cyclization of perfluoroalkyl 3-indole methanol precursors, driven by the strong acidity of TfOH in a specialized fluorinated solvent environment. As depicted in the reaction scheme below, this single-step process converts readily available starting materials into the target fused polycyclic structure with water as the sole by-product, exemplifying the principles of green chemistry. The use of HFIP as a solvent is particularly ingenious, as its unique hydrogen-bonding donor ability stabilizes cationic intermediates and enhances the electrophilicity of the reaction center, facilitating rapid cyclization under ambient conditions. This novel approach not only streamlines the synthetic pathway but also ensures high purity profiles directly from the reactor, minimizing the need for extensive chromatographic purification.
Mechanistic Insights into TfOH-Catalyzed Cyclization
The mechanistic elegance of this transformation lies in the activation of the hydroxyl group by the strong Bronsted acid, generating a stabilized carbocation intermediate that is uniquely suited for intramolecular electrophilic aromatic substitution. The perfluoroalkyl group, while strongly electron-withdrawing, plays a crucial role in stabilizing the adjacent positive charge through inductive effects, preventing premature decomposition or polymerization side reactions that often plague carbocation chemistry. The HFIP solvent further modulates the reaction landscape by forming a hydrogen-bond network that solvates the leaving water molecule and stabilizes the transition state, effectively lowering the activation energy barrier. This precise control over the reaction trajectory ensures that the cyclization occurs regioselectively at the desired position on the indole ring, avoiding the formation of constitutional isomers that would be difficult to separate. For R&D directors, understanding this mechanism is vital for troubleshooting and optimizing the process for novel substrates, as it highlights the delicate balance between acid strength, solvent polarity, and substrate electronics required for success.
Impurity control is inherently built into this mechanism due to the mildness of the reaction conditions and the specificity of the acid catalysis. Unlike harsh Lewis acids that might promote non-selective Friedel-Crafts alkylation or polymerization of the electron-rich indole moiety, TfOH in HFIP provides a controlled acidic environment that favors the desired cyclization pathway. The absence of transition metals eliminates the risk of metal-induced oxidative degradation or cross-coupling side reactions, resulting in a cleaner crude reaction profile. Additionally, the protocol demonstrates excellent functional group tolerance, meaning that sensitive moieties such as esters, nitriles, and halides remain intact throughout the synthesis, preserving the chemical integrity of complex molecular architectures. This high level of chemoselectivity reduces the burden on analytical teams to identify and quantify trace metal impurities or complex organic by-products, accelerating the release of materials for biological testing.
How to Synthesize 10-Perfluoroalkyl-5,10-dihydroindeno[1,2-b]indole Efficiently
The practical implementation of this synthesis is designed for ease of operation, requiring standard laboratory equipment and commercially available reagents without the need for specialized inert atmosphere techniques. The process begins with the dissolution of the protected perfluoroalkyl 3-indole methanol precursor in hexafluoroisopropanol, followed by the careful addition of the trifluoromethanesulfonic acid catalyst. Detailed standardized synthesis steps see the guide below.
- Dissolve the perfluoroalkyl 3-indole methanol precursor in hexafluoroisopropanol (HFIP) solvent.
- Add trifluoromethanesulfonic acid (TfOH) catalyst (10 mol%) to the reaction mixture at room temperature.
- Stir the reaction until completion monitored by TLC, then purify via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this technology offers compelling advantages that directly address the pain points of modern pharmaceutical sourcing and manufacturing. The shift away from precious metal catalysts translates into significant cost savings, not just in terms of reagent purchase but also in the reduced capital expenditure for metal recovery and waste treatment infrastructure. The simplicity of the work-up procedure, which often involves basic extraction and crystallization rather than complex chromatography, allows for faster batch turnover times and higher throughput in production facilities. This efficiency gain is critical for maintaining supply continuity in volatile markets where demand for specialized intermediates can fluctuate rapidly. Furthermore, the use of non-toxic by-products aligns with increasingly stringent environmental regulations, reducing the liability and compliance costs associated with hazardous waste disposal.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts such as palladium or rhodium removes a major cost driver from the bill of materials, while the high atom economy ensures that raw materials are converted efficiently into the final product. The mild reaction conditions also reduce energy consumption associated with heating and cooling, contributing to a lower overall carbon footprint and operational expense. By simplifying the purification process, manufacturers can reduce solvent usage and labor hours, further driving down the cost per kilogram of the active intermediate.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals like triflic acid and HFIP, rather than specialized custom-synthesized ligands or catalysts, mitigates the risk of supply chain disruptions. The robustness of the reaction across a wide range of substrates means that a single manufacturing platform can be adapted to produce multiple derivatives, increasing flexibility and responsiveness to customer needs. This adaptability ensures that procurement teams can secure a stable supply of critical intermediates even when facing fluctuations in raw material availability or geopolitical tensions affecting specialty chemical markets.
- Scalability and Environmental Compliance: The patent explicitly demonstrates the scalability of this process with a successful 10-gram scale-up experiment that maintained high yields, proving its viability for pilot and commercial plant operations. The generation of water as the only by-product significantly simplifies effluent treatment and reduces the environmental impact of the manufacturing process. This green chemistry profile facilitates easier regulatory approval and enhances the sustainability credentials of the final pharmaceutical product, which is increasingly valued by end-users and investors alike.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthetic technology. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation to ensure accuracy and relevance for industry stakeholders.
Q: What are the advantages of this metal-free synthesis method?
A: The method eliminates expensive transition metal catalysts, reduces heavy metal contamination risks, and operates under mild conditions with water as the only by-product, significantly simplifying purification.
Q: Is this process suitable for large-scale production?
A: Yes, the patent demonstrates successful scale-up to 10-gram quantities with maintained high yields (98%), indicating robust potential for industrial manufacturing.
Q: What is the substrate scope of this reaction?
A: The reaction tolerates a wide range of functional groups including halogens, esters, nitro groups, and various perfluoroalkyl chains (CF3 to C4F9), allowing for diverse derivative synthesis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 10-Perfluoroalkyl-5,10-dihydroindeno[1,2-b]indole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this metal-free synthesis technology for the development of advanced therapeutic agents and functional materials. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can seamlessly transition from benchtop discovery to full-scale manufacturing. Our state-of-the-art facilities are equipped to handle fluorinated chemistry safely and efficiently, adhering to stringent purity specifications and rigorous QC labs to guarantee the quality of every batch. We understand the critical nature of supply chain stability in the pharmaceutical industry and are committed to providing a reliable source of high-quality intermediates that meet your exacting standards.
We invite you to collaborate with us to leverage this innovative synthetic route for your next drug discovery program. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and timeline. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you accelerate your path to market with confidence and efficiency.
