Advanced Synthesis of Sulfonamide Imidazolium Salts for Photocatalytic Applications
The pharmaceutical and fine chemical industries are constantly seeking more efficient and environmentally benign methods for constructing complex molecular architectures. Patent CN111187219A introduces a groundbreaking advancement in this domain with the disclosure of a novel sulfonamide imidazole salt compound and its preparation method. This technology addresses critical challenges in organic synthesis by providing a stable, easy-to-handle precursor for generating carbon free radicals under photocatalytic conditions. Unlike traditional radical sources that often suffer from instability or require harsh activation conditions, these imidazolium salts offer a controlled release of reactive species, facilitating diversified addition reactions on free radical acceptors such as alkenes, alkynes, and aromatic compounds. For R&D directors and process chemists, this represents a significant leap forward in accessing chemical space that was previously difficult to navigate due to compatibility issues with sensitive functional groups.
The core innovation lies in the unique structural design of the sulfonamide imidazole salt, which combines the stability of an ionic salt with the reactivity of a latent radical source. The preparation method outlined in the patent is remarkably straightforward, involving a two-step sequence that begins with the sulfonation of an imidazole compound followed by alkylation. This approach not only ensures high chemical purity but also simplifies the downstream processing required to isolate the final product. By integrating this technology into existing synthetic workflows, manufacturers can achieve cost reduction in pharmaceutical intermediate manufacturing through improved yields and reduced waste generation. The ability to selectively generate various carbon free radicals by simply modifying the imidazole backbone provides a versatile platform for the synthesis of high-value drug candidates and agrochemical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the generation of carbon free radicals for organic synthesis has relied heavily on stoichiometric oxidants, reductants, or transition metal catalysts, which present significant drawbacks in an industrial setting. Traditional methods often involve the use of toxic heavy metals that require rigorous and expensive removal processes to meet regulatory standards for pharmaceutical products. Furthermore, many conventional radical precursors are thermally unstable or highly sensitive to moisture and air, making their storage, transportation, and handling hazardous and logistically challenging. The reliance on harsh reaction conditions, such as high temperatures or strong acids, can lead to the decomposition of sensitive substrates, resulting in low yields and complex impurity profiles that are difficult to separate. These limitations not only increase the overall cost of production but also pose environmental risks due to the generation of substantial chemical waste, conflicting with the industry's growing emphasis on green chemistry principles.
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes a mild, two-step synthesis to create stable sulfonamide imidazole salts that serve as efficient radical precursors. The process begins with the reaction of an imidazole compound with sulfonic anhydride or sulfonyl fluoride in the presence of a base like triethylamine at a controlled temperature of 0°C. This is followed by alkylation with alkyl trifluoromethanesulfonate to form the final salt. This methodology eliminates the need for hazardous reagents during the preparation of the precursor itself. The resulting salts are chemically stable solids that can be easily purified by simple washing or recrystallization, avoiding the need for complex chromatographic separations. 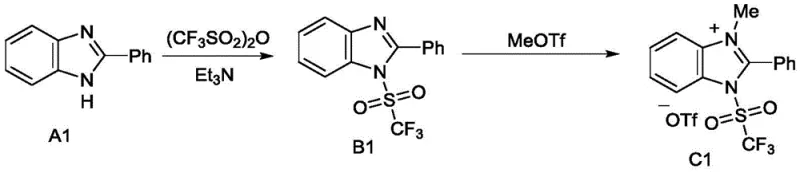 This streamlined workflow significantly enhances operational safety and efficiency, making it an ideal candidate for commercial scale-up of complex pharmaceutical intermediates. The subsequent use of these salts in photocatalytic reactions allows for the generation of radicals under visible light irradiation, further reducing energy consumption and environmental impact.
This streamlined workflow significantly enhances operational safety and efficiency, making it an ideal candidate for commercial scale-up of complex pharmaceutical intermediates. The subsequent use of these salts in photocatalytic reactions allows for the generation of radicals under visible light irradiation, further reducing energy consumption and environmental impact.
Mechanistic Insights into Photocatalytic Radical Generation
The mechanistic pathway enabled by these sulfonamide imidazole salts is a testament to the power of modern photocatalysis in organic synthesis. Upon exposure to visible light in the presence of a photocatalyst, such as the fac-Ir(ppy)3 complex mentioned in the test examples, the sulfonamide imidazole salt undergoes a single-electron transfer (SET) process. This triggers the cleavage of the N-S bond, leading to the extrusion of sulfur dioxide and the formation of a highly reactive carbon-centered free radical. This radical species is then free to engage in a variety of transformative reactions, including the hydrotrifluoromethylation and borylation reactions demonstrated in the patent's experimental section. The beauty of this system lies in its modularity; by altering the substituents on the imidazole ring (R1, R2, R3), chemists can tune the electronic and steric properties of the precursor, thereby controlling the nature of the generated radical. 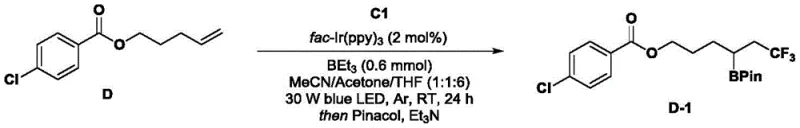 This level of control is crucial for high-purity pharmaceutical intermediate synthesis, where regioselectivity and chemoselectivity are paramount. The mechanism avoids the formation of metal-bound intermediates that can complicate purification, ensuring a cleaner reaction profile.
This level of control is crucial for high-purity pharmaceutical intermediate synthesis, where regioselectivity and chemoselectivity are paramount. The mechanism avoids the formation of metal-bound intermediates that can complicate purification, ensuring a cleaner reaction profile.
Furthermore, the impurity control mechanism inherent in this process is robust. The byproducts of the radical generation step, primarily sulfur dioxide and the neutral imidazole derivative, are either gaseous or easily separable from the desired organic product. This contrasts sharply with metal-catalyzed processes where metal residues can persist and catalyze unwanted side reactions or degrade the final product over time. The use of a photocatalytic cycle also means that the catalyst loading can be kept very low (e.g., 2 mol%), minimizing the introduction of foreign substances into the reaction mixture. For supply chain managers, this translates to a more predictable and consistent product quality, reducing the risk of batch failures due to trace impurities. The ability to perform these reactions at room temperature under an inert atmosphere further protects sensitive functional groups from degradation, preserving the integrity of complex molecular scaffolds.
How to Synthesize Sulfonamide Imidazolium Salt Efficiently
The synthesis of these valuable reagents is designed for practicality and scalability, utilizing common laboratory equipment and readily available reagents. The process typically involves dissolving the chosen imidazole starting material in a chlorinated solvent like dichloromethane, followed by the sequential addition of base and sulfonylating agent. After the initial sulfonation, the intermediate is treated with an alkylating agent to form the final quaternary salt. The workup procedure is notably simple, relying on extraction and washing rather than column chromatography, which is a major advantage for large-scale production. For detailed operational parameters and specific stoichiometric ratios, please refer to the standardized guide below.
- Dissolve the imidazole compound in dichloromethane and react with triethylamine and sulfonic anhydride at 0°C to form the sulfonamide intermediate.
- Remove the solvent under reduced pressure, extract the organic phase, and dry to obtain the imidazole sulfonamide solution.
- Add alkyl trifluoromethanesulfonate to the solution, react for 8-12 hours, and wash the resulting crude product with diethyl ether to obtain the pure salt.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this sulfonamide imidazole salt technology offers compelling strategic advantages. The primary benefit stems from the drastic simplification of the synthesis route, which directly correlates to reduced manufacturing costs and shorter lead times. By eliminating the need for expensive and scarce transition metal catalysts in the radical generation step, and replacing them with a reusable photocatalytic system and a cheap organic salt, the overall cost of goods sold (COGS) can be significantly optimized. Additionally, the stability of the salt precursor allows for bulk purchasing and long-term storage without degradation, providing greater flexibility in inventory management and buffering against supply chain disruptions.
- Cost Reduction in Manufacturing: The process achieves cost efficiency through the use of inexpensive starting materials such as imidazoles and triflic anhydride, which are commodity chemicals available from multiple global suppliers. The elimination of chromatographic purification in favor of simple crystallization or washing steps reduces solvent consumption and labor costs associated with processing time. Furthermore, the high yields reported in the patent examples indicate minimal material wastage, maximizing the atom economy of the process and lowering the effective cost per kilogram of the final active ingredient.
- Enhanced Supply Chain Reliability: The robustness of the synthetic route ensures consistent production output, which is critical for maintaining uninterrupted supply to downstream customers. The reagents used are not subject to the same geopolitical restrictions or supply bottlenecks often associated with precious metal catalysts. This diversification of the supply base reduces risk and enhances the resilience of the manufacturing operation. The ability to store the intermediate salts for extended periods also allows manufacturers to build safety stock, ensuring reducing lead time for high-purity pharmaceutical intermediates when urgent orders arise.
- Scalability and Environmental Compliance: The reaction conditions are mild and operate at near-ambient temperatures, reducing the energy footprint of the manufacturing process. The avoidance of heavy metals simplifies waste treatment protocols, as the effluent does not require specialized processing to remove toxic metal ions. This aligns with increasingly stringent environmental regulations and corporate sustainability goals. The process is inherently scalable, as demonstrated by the straightforward workup procedures that can be easily adapted from gram-scale laboratory experiments to multi-ton industrial reactors without significant re-engineering.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this technology. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation, ensuring accuracy and relevance for potential partners and licensees.
Q: What are the stability characteristics of the sulfonamide imidazole salt?
A: The sulfonamide imidazole salt compounds described in the patent exhibit stable chemical properties, allowing for easy storage and handling compared to transient radical species. They serve as robust precursors that can be purified via recrystallization or washing.
Q: How does this method improve upon traditional radical generation techniques?
A: This method avoids the pollution associated with large amounts of oxidation-reduction agents and metal reagents. It utilizes photocatalysis under mild conditions to selectively generate carbon free radicals, which is particularly suitable for synthesizing sensitive molecules with multiple functional groups.
Q: Can the imidazole structure be modified for different applications?
A: Yes, the patent highlights that by changing the imidazole structure (R1, R2, R3 groups), various carbon free radicals can be selectively generated. This structural flexibility provides significant convenience for diverse organic chemistry and drug synthesis pathways.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Sulfonamide Imidazole Salt Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the sulfonamide imidazole salt technology in advancing modern drug discovery and process chemistry. As a leading CDMO partner, we possess the technical expertise and infrastructure to translate this innovative patent into commercial reality. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project moves seamlessly from the laboratory bench to full-scale manufacturing. We are committed to delivering products that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify every batch.
We invite you to collaborate with us to leverage this cutting-edge chemistry for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific synthesis needs. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our capabilities can accelerate your development timeline and optimize your supply chain efficiency.
