Advanced Synthesis of Indolinotetrahydropyran Compounds for Commercial Pharmaceutical Applications
The pharmaceutical industry continuously seeks robust synthetic routes for complex heterocyclic scaffolds that serve as critical building blocks for new drug candidates. Patent CN115785110A introduces a groundbreaking methodology for the preparation of indolinotetrahydropyran compounds, a class of molecules characterized by their fused polycyclic structure and significant potential in oncology. This technology leverages a palladium-catalyzed dearomatization strategy, specifically targeting electron-poor 3-nitroindoles, which have historically been challenging substrates for such transformations. By overcoming the inherent low nucleophilicity of these systems, the disclosed method provides a reliable pathway to generate high-value pharmaceutical intermediates with exceptional stereocontrol. For R&D directors and procurement specialists, this represents a pivotal advancement in accessing diverse chemical space for antitumor drug discovery pipelines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional asymmetric dearomatization strategies have predominantly focused on electron-rich aromatic systems such as simple indoles or naphthols, relying on their inherent nucleophilicity to drive cycloaddition reactions. However, this reliance severely limits the structural diversity of accessible products, particularly when electron-withdrawing groups are present on the aromatic ring. Previous attempts to engage electron-poor heteroaromatics like 3-nitroindoles in palladium-catalyzed cycloadditions have been scarce, with only isolated reports of [3+2] reactions involving specific dipolar intermediates. The scarcity of [4+2] cycloaddition protocols for these substrates stems from the difficulty in activating the oxygen nucleophile relative to carbon or nitrogen, often leading to poor reactivity or complete failure of the dearomatization step. Consequently, the synthesis of complex indolinotetrahydropyran cores has remained a bottleneck, restricting the ability of medicinal chemists to explore this chemical space for therapeutic applications.
The Novel Approach
The technology disclosed in CN115785110A circumvents these historical limitations by employing a specialized palladium catalytic system capable of generating Π-allylpalladium 1,4-[O,C] dipolar active intermediates from alkenyl carbonates. This innovative approach successfully facilitates the first reported dearomatization [4+2] cycloaddition between these active intermediates and electron-poor 3-nitroindoles. The reaction proceeds under remarkably mild conditions, avoiding the harsh reagents often required to force unreactive substrates to couple. As illustrated in the specific optimization examples, the process demonstrates exceptional efficiency, converting starting materials into the desired fused tetracyclic products with high fidelity. This breakthrough not only expands the toolbox for organic synthesis but also provides a scalable route to compounds that were previously inaccessible or economically unviable to produce.
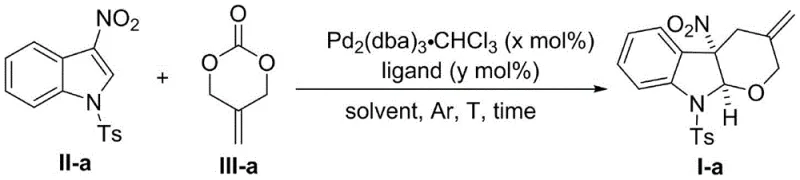
Mechanistic Insights into Pd-Catalyzed Dearomatization [4+2] Cycloaddition
The core of this technological advancement lies in the precise generation and reactivity of the Π-allylpalladium 1,4-[O,C] dipolar active intermediate. Unlike traditional Π-allylpalladium species that typically react through carbon or nitrogen centers, this system uniquely activates the oxygen atom of the alkenyl carbonate to initiate the nucleophilic attack on the 3-nitroindole ring. This mechanistic pathway is critical because it overcomes the thermodynamic barrier associated with the dearomatization of electron-deficient heterocycles. The palladium catalyst, coordinated with a phosphine ligand such as triphenylphosphine, stabilizes the transition state, allowing the reaction to proceed with high regioselectivity and diastereoselectivity. The result is the formation of a new C-O bond and a C-C bond simultaneously, constructing the tetrahydropyran ring fused to the indoline core in a single operational step.
Furthermore, the reaction design inherently supports high purity profiles, which is a paramount concern for pharmaceutical intermediate manufacturing. The use of mild temperatures ranging from 0°C to 50°C minimizes the formation of thermal degradation byproducts and polymerization side reactions that often plague high-energy cycloadditions. The high diastereomeric ratio (>20:1 dr) observed across multiple examples indicates a highly ordered transition state, reducing the burden on downstream purification processes. For supply chain managers, this mechanistic efficiency translates to reduced waste generation and simplified isolation procedures, as the crude products often require only standard column chromatography to achieve analytical purity. The general reaction scope is depicted below, highlighting the versatility of the method across various substituted indoles.
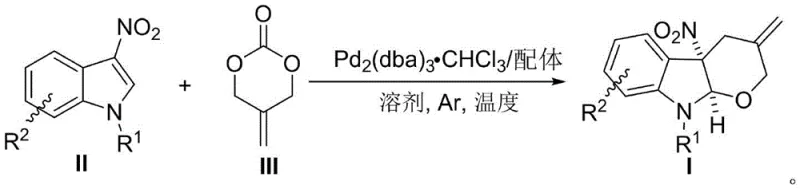
How to Synthesize Indolinotetrahydropyran Efficiently
The synthesis protocol outlined in the patent provides a clear and reproducible framework for producing these valuable intermediates. The process begins with the preparation of the catalytic species in a dry environment to prevent moisture-induced deactivation of the palladium complex. Following the complexation phase, the sequential addition of substrates ensures controlled reaction kinetics, preventing exothermic runaways and ensuring consistent batch quality. The detailed standardized synthesis steps are provided in the guide below, which covers reagent preparation, reaction monitoring, and workup procedures optimized for maximum recovery.
- Prepare the catalyst system by mixing Pd2(dba)3·CHCl3 and a phosphine ligand (e.g., PPh3) in a dry reaction vessel with solvent such as acetonitrile.
- Stir the mixture for approximately 5 minutes to ensure complete complexation of the palladium catalyst before adding substrates.
- Add 3-nitroindole and alkenyl carbonate sequentially, maintain reaction temperature between 0°C to 50°C, and purify the resulting indolinotetrahydropyran compound via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial manufacturing perspective, this synthetic route offers substantial advantages over legacy methods, particularly regarding cost structure and operational safety. The ability to utilize commercially available starting materials like 3-nitroindoles and alkenyl carbonates eliminates the need for custom-synthesized precursors that often carry high price tags and long lead times. Moreover, the reaction operates at near-ambient temperatures, significantly reducing the energy consumption associated with heating or cryogenic cooling typically required for sensitive organometallic transformations. This energy efficiency directly correlates to lower utility costs per kilogram of product, enhancing the overall economic viability of large-scale production campaigns.
- Cost Reduction in Manufacturing: The catalytic system is highly efficient, functioning effectively with palladium loadings as low as 1 mol%. This minimal catalyst requirement drastically reduces the consumption of expensive precious metals, which is a major cost driver in fine chemical synthesis. Additionally, the use of triphenylphosphine as a ligand offers a cost-effective alternative to specialized chiral ligands that can be prohibitively expensive. By eliminating the need for stoichiometric amounts of activators or harsh reagents, the process further lowers the raw material bill, allowing for significant margin improvement in the final API intermediate pricing.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions contributes to a more resilient supply chain. Since the process tolerates a range of solvents including acetonitrile and toluene, procurement teams have flexibility in sourcing materials based on market availability and price fluctuations. The high yields reported, reaching up to 99% in optimized examples, ensure that less starting material is wasted, maximizing the output from each batch. This efficiency reduces the frequency of production runs needed to meet demand, thereby lowering the risk of supply disruptions and ensuring a steady flow of material to downstream drug substance manufacturers.
- Scalability and Environmental Compliance: The simplicity of the operation, involving straightforward mixing and stirring without complex pressure or temperature cycling, makes this chemistry highly amenable to scale-up from laboratory to multi-ton production. The mild conditions also imply a safer working environment with reduced risk of thermal hazards, aligning with stringent modern safety standards. Furthermore, the high atom economy of the cycloaddition reaction minimizes waste generation, supporting environmental compliance goals and reducing the costs associated with waste disposal and treatment facilities.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and beneficial effects documented in the patent specification, providing clarity on performance metrics and application potential. Understanding these details helps stakeholders evaluate the feasibility of integrating this route into their existing manufacturing portfolios.
Q: What is the key innovation in this synthesis method compared to conventional dearomatization?
A: Unlike conventional methods that rely on electron-rich aromatics, this patent utilizes electron-poor 3-nitroindole in a Pd-catalyzed [4+2] cycloaddition, enabling access to novel indolinotetrahydropyran scaffolds with high diastereoselectivity.
Q: What are the optimal reaction conditions for maximizing yield?
A: The patent specifies that using acetonitrile as the solvent, triphenylphosphine as the ligand, and maintaining a temperature of 40°C with a catalyst loading as low as 1 mol% results in yields up to 99%.
Q: Do these compounds show biological activity?
A: Yes, preliminary cell activity assays indicate that these compounds exhibit significant inhibitory effects on human leukemia K562 cells, suggesting potential as lead compounds for antitumor drug development.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Indolinotetrahydropyran Supplier
NINGBO INNO PHARMCHEM stands at the forefront of translating advanced academic research into commercial reality. With extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, we possess the technical capability to adapt this patented Pd-catalyzed process for industrial requirements. Our facility is equipped with rigorous QC labs and adheres to stringent purity specifications, ensuring that every batch of indolinotetrahydropyran intermediate meets the exacting standards required for pharmaceutical development. We understand the critical nature of supply continuity and quality consistency in the global drug supply chain.
We invite procurement leaders and R&D directors to collaborate with us to leverage this cutting-edge technology for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume needs. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing expertise can accelerate your timeline to market while optimizing your budget.
