Scalable Synthesis of Sulfonyl Thioesters via Halide-Promoted Oxidation for Global Pharma Supply Chains
Scalable Synthesis of Sulfonyl Thioesters via Halide-Promoted Oxidation for Global Pharma Supply Chains
The pharmaceutical and fine chemical industries are constantly seeking robust synthetic methodologies that balance high purity with economic viability, and the technology disclosed in patent CN113620764A represents a significant leap forward in the preparation of sulfonyl thioesters. This innovative approach utilizes readily available thiols as starting materials, employing hydrogen peroxide as a green oxidant in the presence of specific alkali or alkaline earth metal halide additives. By shifting away from traditional, hazardous reagents, this method not only simplifies the operational workflow but also ensures the production of intermediates with exceptional purity profiles, addressing the stringent quality requirements of modern drug development pipelines. The strategic integration of halide promoters allows for precise control over the oxidation state, minimizing side reactions and maximizing yield without the need for expensive transition metal catalysts that often contaminate the final product.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of sulfonyl thioesters has been plagued by significant technical and economic hurdles that hinder efficient large-scale manufacturing. Traditional routes often rely on the use of acyl chlorides as starting materials, which are highly corrosive and pose severe safety risks during handling and storage, necessitating specialized equipment and rigorous safety protocols. Furthermore, these legacy methods frequently require the addition of zinc powder to facilitate the reaction, resulting in moderate yields of only around 76% and generating substantial amounts of solid waste that complicate downstream processing. Alternatively, pathways utilizing disulfides as precursors involve expensive raw materials and aggressive oxidants like potassium permanganate or potassium hydrogen persulfate, which introduce heavy metal contaminants and generate excessive three-waste streams, thereby inflating production costs and environmental compliance burdens for chemical manufacturers.
The Novel Approach
In stark contrast to these cumbersome legacy processes, the novel methodology described in the patent introduces a streamlined oxidative coupling strategy that leverages the reactivity of thiols directly. By employing hydrogen peroxide, a benign oxidant that produces water as its only byproduct, the process drastically reduces the environmental footprint while maintaining high reaction efficiency. The inclusion of simple halide salts, such as potassium bromide or lithium chloride, serves as a powerful catalytic system that activates the oxidant under mild thermal conditions, typically between 30°C and 55°C. This gentle approach not only preserves the integrity of sensitive functional groups but also achieves product purities exceeding 99%, effectively eliminating the need for complex rectification steps and delivering a superior quality intermediate ready for immediate use in subsequent synthetic transformations.
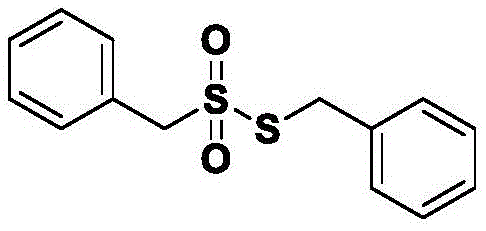
Mechanistic Insights into Halide-Promoted Thiol Oxidation
The core of this technological breakthrough lies in the synergistic interaction between the thiol substrate, hydrogen peroxide, and the halide additive, which creates a highly reactive yet controlled oxidative environment. Mechanistically, the halide ion likely facilitates the generation of a reactive hypohalous species in situ or acts as an electron transfer mediator that lowers the activation energy for the oxidation of the sulfur atom. This pathway avoids the formation of harsh radical species that typically lead to over-oxidation or decomposition, ensuring that the reaction proceeds selectively to the desired sulfonyl thioester state. The patent data indicates that maintaining the reaction pH within a narrow window of 6 to 8 is critical, as deviations into highly acidic or basic conditions can trigger hydrolysis or degradation of the product, underscoring the importance of precise process control in achieving optimal outcomes.
Furthermore, the stability profile of the synthesized compounds reveals that the sulfonyl thioester linkage is robust under neutral conditions but susceptible to cleavage in extreme pH environments, which informs the design of the workup procedure. The process utilizes a straightforward extraction protocol involving organic solvents like methyl tert-butyl ether followed by washing with sodium sulfite to quench residual oxidants, ensuring a clean isolation of the target molecule. This mechanistic understanding allows chemists to tailor the reaction parameters for various substrates, ranging from simple aliphatic chains to complex heteroaryl systems, as evidenced by the successful synthesis of diverse derivatives including propyl and benzyl variants with consistently high yields and purity levels across different scales.
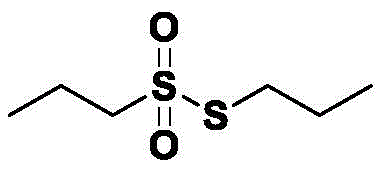
How to Synthesize Sulfonyl Thioester Efficiently
The implementation of this synthesis route requires careful attention to the stoichiometric ratios and thermal management to ensure reproducibility and safety. The general protocol involves charging a reaction vessel with the thiol substrate, a polar solvent such as acetone or acetonitrile, water, and the specific halide additive, followed by heating to the designated temperature range. Once the system is equilibrated, hydrogen peroxide is added dropwise to manage the exotherm and control the rate of oxidation, with the reaction progress monitored via gas chromatography until the starting material is fully consumed. For a detailed breakdown of the specific reagent quantities, temperature ramps, and workup procedures validated in the patent examples, please refer to the standardized synthesis guide below.
- Charge a reaction vessel with thiol substrate, a compatible solvent (e.g., acetone or acetonitrile), water, and an alkali metal halide additive such as potassium bromide.
- Heat the mixture to a mild temperature range of 30-55°C and slowly add hydrogen peroxide solution while maintaining strict pH control between 6 and 8.
- Upon reaction completion, perform liquid-liquid extraction using organic solvents like methyl tert-butyl ether, wash with sodium sulfite to remove excess oxidant, and concentrate under reduced pressure.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, this patented technology offers transformative benefits that directly impact the bottom line and operational resilience of chemical manufacturing operations. By replacing expensive and hazardous reagents like acyl chlorides and disulfides with commodity chemicals such as thiols and hydrogen peroxide, the raw material costs are significantly reduced, creating a more competitive pricing structure for the final intermediate. The elimination of transition metal catalysts, which were shown in comparative examples to yield poor results and complicate purification, further streamlines the production process by removing the need for costly metal scavenging steps and reducing the risk of heavy metal contamination in the final API. This simplification of the synthetic route translates into shorter cycle times and lower utility consumption, enhancing the overall economic efficiency of the manufacturing campaign.
- Cost Reduction in Manufacturing: The substitution of high-cost oxidants and metal catalysts with inexpensive halide salts and hydrogen peroxide drives a substantial decrease in direct material expenses. Additionally, the high selectivity of the reaction minimizes the formation of byproducts, which reduces the load on purification units and lowers the consumption of solvents and energy required for distillation or recrystallization. This lean manufacturing approach ensures that the cost of goods sold is optimized, allowing for better margin management in volatile market conditions without compromising on the quality specifications required by regulatory bodies.
- Enhanced Supply Chain Reliability: Utilizing widely available commercial starting materials like n-propyl mercaptan and benzyl mercaptan mitigates the risk of supply disruptions associated with specialized or custom-synthesized reagents. The robustness of the reaction conditions, which tolerate a range of solvents and operate at mild temperatures, ensures consistent batch-to-batch performance, thereby guaranteeing a steady flow of high-quality intermediates to downstream customers. This reliability is crucial for maintaining continuous production schedules in pharmaceutical manufacturing, where delays in intermediate supply can have cascading effects on the entire drug development timeline.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated in multi-liter setups with straightforward workup procedures that do not require exotic equipment. The use of hydrogen peroxide as a green oxidant aligns with increasingly stringent environmental regulations by avoiding the generation of heavy metal waste and corrosive byproducts. This eco-friendly profile not only simplifies waste disposal and reduces treatment costs but also enhances the sustainability credentials of the supply chain, appealing to global partners who prioritize green chemistry principles in their vendor selection criteria.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and advantages of this sulfonyl thioester synthesis method, derived directly from the experimental data and comparative analysis provided in the patent documentation. These insights are intended to clarify the operational benefits and chemical rationale behind the halide-promoted oxidation strategy for R&D and technical procurement teams evaluating this technology for adoption. Understanding these nuances is essential for assessing the feasibility of integrating this route into existing manufacturing frameworks.
Q: Why is the halide additive critical in this sulfonyl thioester synthesis?
A: The alkali or alkaline earth metal halide acts as a crucial promoter that facilitates the oxidation of the thiol by hydrogen peroxide. Without this additive, the reaction efficiency drops significantly, and the use of expensive transition metal catalysts becomes necessary, which complicates purification and increases costs.
Q: What is the typical purity achievable with this new method?
A: This patented process consistently delivers products with gas phase purity exceeding 99%, which is notably higher than many commercially available imported materials. This high purity reduces the need for complex rectification steps often required in older synthetic routes.
Q: Is this process suitable for large-scale industrial manufacturing?
A: Yes, the process is designed for industrial scalability. It utilizes inexpensive, commercially available starting materials like thiols and hydrogen peroxide, operates at mild temperatures (30-55°C), and avoids hazardous reagents like acyl chlorides, making it safe and cost-effective for tonnage production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Sulfonyl Thioester Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic technologies to maintain a competitive edge in the global fine chemical market. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. We are committed to delivering high-purity sulfonyl thioesters that meet stringent purity specifications, leveraging our rigorous QC labs to verify every batch against the highest industry standards. Our infrastructure is designed to handle complex oxidative chemistries safely, providing our partners with a secure and reliable source of critical intermediates for their pharmaceutical and agrochemical programs.
We invite you to collaborate with us to explore how this innovative synthesis route can be tailored to your specific project needs, offering a pathway to significant cost savings and supply chain optimization. Please contact our technical procurement team to request a Customized Cost-Saving Analysis, where we can evaluate your current sourcing strategy against the efficiencies offered by this new method. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our capabilities align with your quality and volume requirements, ensuring a partnership that drives mutual success in the development of next-generation therapeutic agents.
