Advanced Synthesis of 1H-Pyrrolo[3,2-c]pyridine-6-ol for High-Purity Pharmaceutical Intermediates
Advanced Synthesis of 1H-Pyrrolo[3,2-c]pyridine-6-ol for High-Purity Pharmaceutical Intermediates
The pharmaceutical industry continuously demands robust and scalable synthetic routes for complex heterocyclic scaffolds, particularly those serving as key building blocks for cardiovascular therapeutics. Patent CN111978320B introduces a groundbreaking methodology for the synthesis of 1H-pyrrolo[3,2-c]pyridine-6-ol, a critical intermediate in the development of renin inhibitors used to manage hypertension and reduce cardiovascular risks. This technical disclosure addresses a long-standing challenge in medicinal chemistry where traditional hydrolysis methods fail to convert specific halogenated precursors into their corresponding hydroxyl derivatives. By leveraging a strategic benzyl protection group approach followed by selective hydrogenolysis, this innovation offers a viable pathway for producing high-purity renin inhibitor intermediates with exceptional efficiency. For R&D directors and procurement specialists, understanding this novel route is essential for securing reliable supply chains and optimizing the cost reduction in API manufacturing for next-generation antihypertensive drugs.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
In the realm of heterocyclic chemistry, the conversion of halogen atoms to hydroxyl groups on pyridine rings is typically achieved through standard acidic or basic hydrolysis protocols. However, when applied to the specific substrate of 6-chloro-5-azaindole, these conventional wisdom approaches collapse completely. As illustrated in the general reaction scheme below, standard treatment with strong bases like sodium hydroxide or acids like hydrochloric acid yields no target product, likely due to the unique electron density distribution and stability issues inherent to the azaindole core. This failure represents a significant bottleneck for process chemists attempting to access this valuable scaffold, often forcing them to explore longer, more expensive multi-step sequences or abandon the route entirely. The inability to directly hydrolyze the chlorine atom necessitates a fundamental rethinking of the synthetic strategy to bypass these thermodynamic or kinetic barriers.
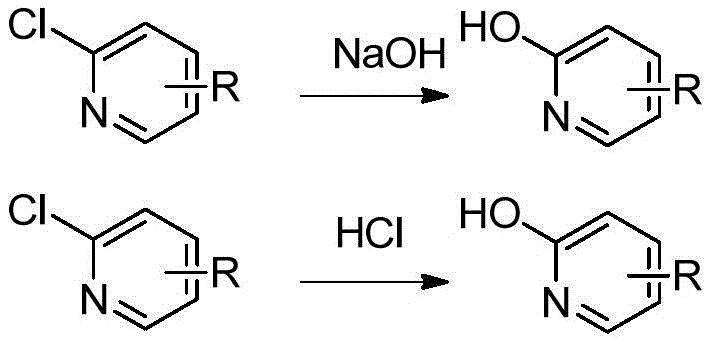
The Novel Approach
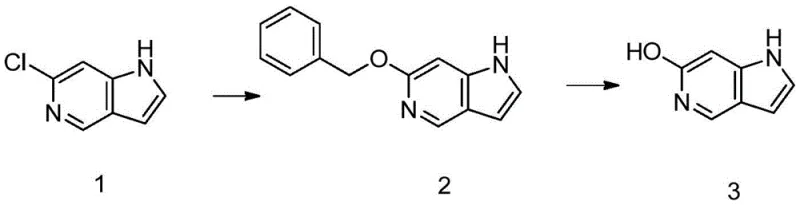
To overcome the inertness of the chloro-azaindole towards direct hydrolysis, the patented method employs a clever detour via a benzyl ether intermediate. Instead of attacking the chlorine directly with hydroxide ions, the process first installs a benzyloxy group through a nucleophilic substitution reaction using benzyl alcohol and a strong base. This transformation successfully converts the recalcitrant chloro-group into a stable ether linkage, which can subsequently be cleaved under mild reducing conditions. This two-step sequence—protection followed by deprotection—effectively solves the problem of converting chlorine on 6-chloro-5-azaindole into hydroxyl, a feat previously unreported in literature. The result is a short, high-yielding route that transforms a difficult chemical problem into a manageable industrial process, ensuring the successful preparation of the 1H-pyrrolo[3,2-c]pyridine-6-ol product with minimal impurity generation.
Mechanistic Insights into Benzyl Protection and Hydrogenolysis
The success of this synthesis relies on the precise orchestration of two distinct chemical mechanisms: nucleophilic aromatic substitution facilitated by strong bases, and catalytic hydrogenolysis controlled by specific additives. In the first step, potassium tert-butoxide acts as a potent base to deprotonate benzyl alcohol, generating a reactive benzyloxide species that attacks the electron-deficient carbon at the 6-position of the azaindole ring. This reaction proceeds efficiently in solvents like dioxane at elevated temperatures (around 100°C), driving the equilibrium towards the formation of the 6-(benzyloxy)-1H-pyrrolo[3,2-c]pyridine intermediate. The choice of base and solvent is critical here, as it must be strong enough to facilitate the substitution without degrading the sensitive heterocyclic core, achieving yields exceeding 86% under optimized conditions.
The second step involves the removal of the benzyl protecting group via catalytic hydrogenation, a process that requires careful tuning to prevent side reactions. The use of palladium on carbon (Pd/C) as a catalyst in a hydrogen atmosphere allows for the cleavage of the benzyl-oxygen bond, regenerating the free hydroxyl group. Crucially, the addition of a secondary base, specifically triethylenediamine (TEDA), plays a pivotal role in maintaining the integrity of the molecule. Without this additive, the reducing conditions can lead to the over-reduction of the pyrrole double bond, forming the unwanted 2,3-dihydro by-product. The presence of TEDA effectively inhibits this over-reduction, ensuring that the reaction stops precisely at the desired alcohol stage with yields reaching nearly 92%. This mechanistic nuance highlights the sophistication of the process design.
How to Synthesize 1H-Pyrrolo[3,2-c]pyridine-6-ol Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires strict adherence to the optimized parameters defined in the patent to ensure maximum yield and purity. The process begins with the preparation of the benzyl ether intermediate, where stoichiometry and temperature control are paramount to driving the substitution to completion. Following isolation, the intermediate undergoes hydrogenolysis, where the concentration of the amine additive must be carefully managed to suppress side reactions while maintaining catalytic activity. The detailed standardized synthetic steps, including specific molar ratios, solvent choices, and workup procedures, are outlined in the guide below for technical teams ready to adopt this superior methodology.
- React 6-chloro-5-azaindole with benzyl alcohol and potassium tert-butoxide in dioxane at 100°C for 16 hours to form the benzyl ether intermediate.
- Perform catalytic hydrogenolysis on the intermediate using 10% Pd/C and triethylenediamine in methanol at room temperature to obtain the final alcohol product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this patented synthetic route offers profound logistical and economic benefits compared to traditional or hypothetical alternatives. The primary advantage lies in the utilization of commercially available raw material reagents, specifically 6-chloro-5-azaindole and benzyl alcohol, which are readily accessible in the global chemical market. This availability eliminates the risk of supply bottlenecks associated with exotic or custom-synthesized starting materials, thereby enhancing supply chain reliability and ensuring consistent production schedules. Furthermore, the avoidance of dangerous and toxic chemical reagents simplifies the regulatory compliance landscape, reducing the administrative burden and safety costs associated with handling hazardous substances in a manufacturing environment.
- Cost Reduction in Manufacturing: The streamlined two-step nature of this synthesis significantly lowers the operational expenditure required to produce the target intermediate. By eliminating the need for complex multi-step sequences or harsh hydrolysis conditions that often require specialized corrosion-resistant equipment, the overall capital and operational costs are substantially reduced. The high yields observed in both steps (over 86% and 91% respectively) mean that less raw material is wasted, directly improving the atom economy and lowering the cost per kilogram of the final active pharmaceutical ingredient. Additionally, the mild reaction conditions reduce energy consumption, contributing to further financial efficiencies in large-scale production runs.
- Enhanced Supply Chain Reliability: The robustness of this chemical process translates directly into a more resilient supply chain for downstream drug manufacturers. Because the reaction conditions are mild—operating at reflux temperatures for the first step and room temperature for the second—the risk of batch failure due to thermal runaway or uncontrollable exotherms is minimized. This predictability allows for tighter delivery windows and more accurate forecasting, which is critical for just-in-time manufacturing models in the pharmaceutical sector. The use of common solvents like dioxane and methanol further ensures that solvent supply disruptions are unlikely to halt production, securing the continuity of supply for critical cardiovascular medications.
- Scalability and Environmental Compliance: From an environmental and scaling perspective, this method is exceptionally well-suited for commercial scale-up of complex heterocycles. The absence of heavy metal catalysts other than the recoverable palladium on carbon, and the lack of toxic reagents, simplifies waste stream management and effluent treatment. This aligns with modern green chemistry principles and increasingly stringent environmental regulations, reducing the liability and cost associated with waste disposal. The simplicity of the operation, involving standard filtration and spin-drying techniques, means that the process can be easily transferred from gram-scale laboratory synthesis to ton-scale industrial reactors without requiring extensive re-engineering of the production line.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this synthesis route, derived directly from the experimental data and beneficial effects described in the patent documentation. These insights are intended to clarify the specific advantages of this method over prior art and to assist technical teams in evaluating its feasibility for their specific production needs. Understanding these nuances is key to leveraging the full potential of this technology for high-value pharmaceutical applications.
Q: Why does direct hydrolysis fail for 6-chloro-5-azaindole?
A: Direct acid or base hydrolysis of the chlorine atom at the 6-position of the azaindole ring fails to produce the target hydroxyl compound due to the specific electronic environment of the heterocycle, often resulting in no reaction or decomposition rather than substitution.
Q: What is the critical role of triethylenediamine (TEDA) in the second step?
A: Triethylenediamine acts as a crucial additive during the hydrogenolysis step; its presence effectively inhibits the over-reduction of the pyrrole ring, preventing the formation of the unwanted by-product 2,3-dihydro-1H-pyrrolo[3,2-c]pyridin-6-ol and ensuring high selectivity.
Q: Is this synthesis method suitable for industrial scale-up?
A: Yes, the method utilizes commercially available raw materials, avoids dangerous toxic reagents, and operates under mild conditions (reflux and room temperature hydrogenation), making it highly suitable for safe and efficient commercial scale-up of complex heterocycles.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1H-Pyrrolo[3,2-c]pyridine-6-ol Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the development of life-saving cardiovascular therapies. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to market availability is seamless and efficient. We are committed to delivering products that meet stringent purity specifications, supported by our rigorous QC labs that utilize advanced analytical techniques to verify every batch. By adopting the advanced synthetic strategies outlined in recent patents like CN111978320B, we continue to push the boundaries of what is possible in fine chemical manufacturing, offering our partners a competitive edge through superior quality and reliability.
We invite pharmaceutical companies and research institutions to collaborate with us to optimize their supply chains for renin inhibitor intermediates. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality standards. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise in complex heterocycle synthesis can support your next breakthrough in drug development.
