Advanced Synthesis of Fluorinated Piperazine Intermediates for Commercial API Manufacturing
Advanced Synthesis of Fluorinated Piperazine Intermediates for Commercial API Manufacturing
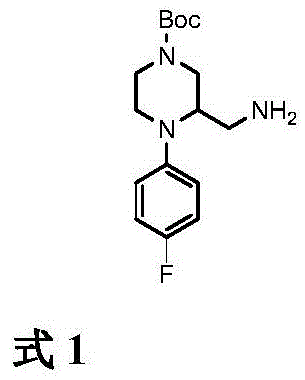
The pharmaceutical industry constantly demands novel intermediates that offer superior purity profiles and scalable synthetic routes for next-generation drug candidates. A significant breakthrough in this domain is documented in Chinese patent CN111892544A, which discloses a robust method for synthesizing tert-butyl 3-(aminomethyl)-4-(4-fluorophenyl)piperazine-1-carboxylate. This compound serves as a critical building block in the construction of complex heterocyclic APIs, particularly those targeting central nervous system disorders and oncology pathways. The disclosed methodology represents a strategic advancement over traditional approaches by utilizing a linear eight-step sequence that maximizes atom economy while minimizing hazardous waste generation. For R&D directors and procurement specialists, understanding the nuances of this synthesis is vital for securing a reliable supply chain for high-value fluorinated scaffolds.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of substituted piperazines has been plagued by challenges related to regioselectivity and the formation of difficult-to-remove impurities. Conventional routes often rely on the direct alkylation of unsubstituted piperazine, which frequently results in over-alkylation and the generation of symmetrical byproducts that require extensive chromatographic purification. Furthermore, traditional methods may employ harsh reaction conditions or expensive transition metal catalysts that leave behind toxic residues, complicating the regulatory approval process for the final active pharmaceutical ingredient. The lack of control over stereochemistry and the introduction of the aminomethyl side chain often necessitates additional protection and deprotection steps, thereby inflating the cost of goods sold and extending the overall lead time for material delivery.
The Novel Approach
In contrast, the methodology outlined in patent CN111892544A introduces a highly controlled, stepwise construction of the piperazine ring system starting from readily available benzylamine. This novel approach strategically builds the carbon-nitrogen backbone before closing the ring, allowing for precise installation of the 4-fluorophenyl group and the aminomethyl functionality. By employing a sequence of substitution, reduction, and cyclization reactions, the process achieves remarkable yields, with key steps such as the lithium aluminum hydride reduction reaching efficiencies as high as 98.4%. This logical disconnection of the target molecule not only simplifies the purification protocols but also ensures a consistent impurity profile, making it an ideal candidate for reliable pharmaceutical intermediate supplier partnerships focused on quality and consistency.
Mechanistic Insights into Stepwise Piperazine Construction
The core of this synthetic strategy lies in the meticulous orchestration of nucleophilic substitutions and reductive cyclizations. The process initiates with the acylation of benzylamine using chloroacetyl chloride at low temperatures (0-3°C) to prevent poly-acylation, forming N-benzyl-2-chloroacetamide. This intermediate then undergoes a crucial nucleophilic substitution with 4-fluorobenzylamine under elevated temperatures (90-110°C) to install the fluorinated aromatic ring. The subsequent reduction of the amide bond using lithium aluminum hydride (LiAlH4) is a pivotal transformation, converting the carbonyl group into a methylene bridge with exceptional efficiency. This reduction step is critical for establishing the flexible ethylamine linker required for the subsequent ring-closing reaction, demonstrating the power of classical reducing agents in modern complex molecule synthesis.
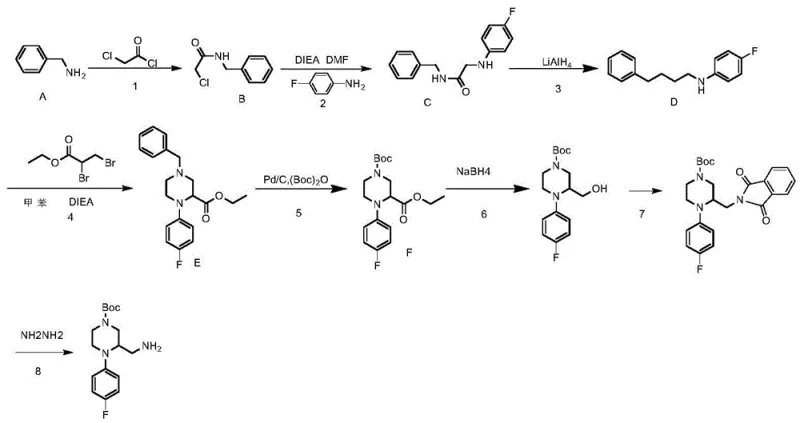
Following the formation of the linear diamine precursor, the cyclization step utilizes ethyl 2,3-dibromopropionate to close the piperazine ring, creating the core heterocyclic structure. The introduction of the tert-butoxycarbonyl (Boc) protecting group via catalytic hydrogenation using Pd(OH)2/C serves a dual purpose: it protects the secondary amine and simultaneously removes the benzyl protecting group in a single pot operation. The final stages involve the conversion of the ester side chain to an aminomethyl group via reduction, Mitsunobu reaction with phthalimide, and subsequent hydrazinolysis. This sequence ensures that the primary amine is introduced late in the synthesis, preventing unwanted side reactions during the earlier high-temperature cyclization steps, thereby enhancing the overall purity of the final high-purity pharmaceutical intermediate.
How to Synthesize tert-Butyl 3-(aminomethyl)-4-(4-fluorophenyl)piperazine-1-carboxylate Efficiently
Executing this synthesis requires strict adherence to the reaction parameters defined in the patent to ensure optimal yield and safety. The process involves handling reactive reagents such as lithium aluminum hydride and diethyl azodicarboxylate (DEAD), necessitating rigorous temperature control and inert atmosphere conditions. Operators must monitor each step via thin-layer chromatography (TLC) to confirm complete consumption of starting materials before proceeding to workup. The detailed standardized synthesis steps below outline the specific stoichiometry, solvent choices, and isolation techniques required to replicate the high yields reported in the intellectual property documentation.
- Substitution of benzylamine with chloroacetyl chloride to form N-benzyl-2-chloroacetamide.
- Nucleophilic substitution with 4-fluorobenzylamine followed by LiAlH4 reduction to form the amine backbone.
- Cyclization using ethyl 2,3-dibromopropionate to form the piperazine ring, followed by Boc protection and functional group manipulation.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers tangible benefits regarding cost stability and material availability. The reliance on commodity chemicals such as benzylamine, 4-fluorobenzylamine, and common solvents like tetrahydrofuran and toluene mitigates the risk of raw material shortages that often plague specialty chemical manufacturing. By avoiding proprietary or scarce catalysts, the production process becomes more resilient to market fluctuations, ensuring a continuous flow of materials for downstream API production. This stability is crucial for maintaining uninterrupted manufacturing schedules in the fast-paced pharmaceutical sector.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts in favor of more economical reagents like LiAlH4 and Pd/C significantly lowers the direct material costs associated with production. Furthermore, the high yields observed in critical steps, particularly the reduction and cyclization phases, reduce the amount of raw material required per kilogram of finished product. This efficiency translates into substantial cost savings in pharmaceutical intermediate manufacturing, allowing for more competitive pricing models without compromising on the stringent quality standards required by regulatory bodies.
- Enhanced Supply Chain Reliability: The use of robust, well-understood chemical transformations ensures that the synthesis can be reliably transferred between different manufacturing sites if necessary. The simplicity of the workup procedures, which primarily involve standard extractions and crystallizations, reduces the dependency on specialized equipment or highly skilled labor for purification. This operational flexibility enhances supply chain reliability, reducing lead time for high-purity intermediates and ensuring that just-in-time delivery commitments to global clients can be consistently met.
- Scalability and Environmental Compliance: The synthetic route is designed with scalability in mind, utilizing reaction conditions that are easily adaptable from laboratory glassware to large-scale industrial reactors. The waste streams generated are primarily organic solvents and salts that can be managed through standard recovery and treatment protocols, aligning with modern environmental compliance standards. This ease of commercial scale-up of complex heterocycles ensures that production volumes can be rapidly increased from 100 kgs to 100 MT annual commercial production levels to meet surging market demand.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this fluorinated piperazine derivative. These insights are derived directly from the technical specifications and experimental data provided in the patent literature, offering clarity on process capabilities and quality assurance measures. Understanding these details is essential for partners evaluating the feasibility of integrating this intermediate into their own drug development pipelines.
Q: What are the critical purity specifications for this piperazine intermediate?
A: High-purity standards are essential, typically requiring HPLC purity above 98% with严格控制 control over residual solvents and heavy metals, especially given the use of Pd/C and LiAlH4 in the synthesis.
Q: Is the 8-step synthesis route scalable for industrial production?
A: Yes, the patent describes robust conditions using common solvents like THF and toluene, with yields ranging from 50% to over 98% in key steps, indicating strong potential for commercial scale-up.
Q: How does this method improve upon conventional piperazine synthesis?
A: This route avoids harsh conditions often associated with direct piperazine alkylation, utilizing a stepwise construction that allows for better impurity profiling and higher overall yield.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable tert-Butyl 3-(aminomethyl)-4-(4-fluorophenyl)piperazine-1-carboxylate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of your drug development programs. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements regardless of the project phase. We operate with stringent purity specifications and utilize rigorous QC labs to verify that every batch of tert-butyl 3-(aminomethyl)-4-(4-fluorophenyl)piperazine-1-carboxylate meets the highest industry standards for identity, assay, and impurity content.
We invite you to collaborate with us to optimize your supply chain for this valuable building block. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume needs. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our manufacturing capabilities can support your long-term commercial goals.
