Advancing Pharmaceutical Intermediate Production with Efficient Rhodium-Catalyzed Furan-2(5H)-one Synthesis
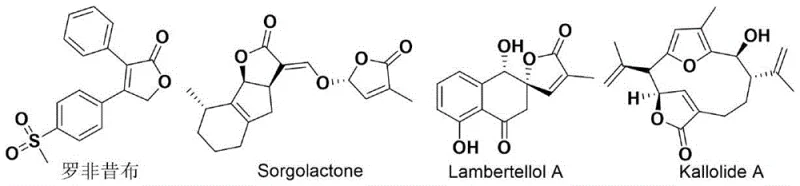
The pharmaceutical and fine chemical industries are constantly seeking more efficient pathways to access complex heterocyclic scaffolds that serve as the backbone for bioactive molecules. A significant breakthrough in this domain is documented in Chinese Patent CN114044771B, which discloses a novel preparation method for compounds possessing a 5-hydroxyfuran-2(5H)-one skeleton. This structural motif is prevalent in numerous pharmacologically active agents, including selective COX-2 inhibitors like Rofecoxib and various natural products such as Sorgolactone and Lambertellol A, which exhibit antibacterial and anti-inflammatory properties. The patent introduces a highly efficient rhodium-catalyzed tandem reaction strategy that streamlines the construction of these valuable cores from readily available indole and 4-hydroxy-2-alkynoate precursors. By leveraging a C–H olefination, directing group migration, lactonization, and C–H hydroxylation cascade, this technology offers a transformative approach to synthesizing high-purity pharmaceutical intermediates with exceptional atom economy.
Traditional synthetic routes to furan-2(5H)-one derivatives often suffer from significant limitations that hinder their application in large-scale commercial manufacturing. Conventional methods typically require multi-step sequences involving harsh reaction conditions, expensive stoichiometric oxidants, and sensitive protecting group manipulations. These legacy processes frequently necessitate strict inert atmospheres and elevated temperatures, leading to increased energy consumption and safety risks associated with thermal runaways. Furthermore, the poor regioselectivity and stereoselectivity observed in older methodologies often result in complex impurity profiles, necessitating costly and time-consuming purification steps that drastically reduce overall yield. The reliance on precious metal catalysts that are difficult to recover or remove also poses challenges for meeting stringent residual metal specifications required for active pharmaceutical ingredients (APIs).
In stark contrast, the novel approach detailed in the patent utilizes a sophisticated rhodium(III) catalytic system that operates under remarkably mild and practical conditions. The reaction proceeds efficiently at room temperature (25°C) in an open air atmosphere, eliminating the need for expensive inert gas lines and specialized pressure equipment. By employing [Cp*RhCl2]2 as the catalyst and sodium acetate as a benign additive in acetone solvent, the process achieves high yields and excellent functional group tolerance. This one-pot tandem transformation not only simplifies the operational workflow but also significantly enhances the sustainability profile of the synthesis by reducing waste generation and energy usage. The ability to directly construct the complex 5-hydroxyfuran-2(5H)-one framework from simple starting materials represents a paradigm shift in how these critical intermediates can be produced for the global market.
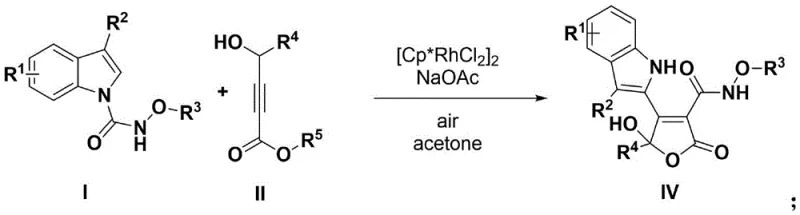
The mechanistic intricacies of this rhodium-catalyzed C–H activation cascade provide deep insights into its high efficiency and selectivity. The catalytic cycle is initiated by the coordination of the rhodium species to the directing group on the indole substrate, facilitating a concerted metalation-deprotonation (CMD) process that activates the proximal C–H bond. Subsequent migratory insertion of the 4-hydroxy-2-alkynoate into the Rh–C bond sets the stage for the formation of the new carbon-carbon bond. Crucially, the presence of the hydroxyl group on the alkynoate enables an intramolecular nucleophilic attack, triggering lactonization to form the furanone ring. The final C–H hydroxylation step, likely mediated by the oxygen in the air acting as the terminal oxidant, installs the essential hydroxyl functionality at the 5-position with high stereocontrol. This elegant sequence avoids the formation of unstable intermediates and minimizes side reactions, ensuring a clean conversion to the desired product.
From an impurity control perspective, the high regioselectivity of the rhodium catalyst is paramount for ensuring product quality. The directing group strategy ensures that functionalization occurs exclusively at the desired position on the indole ring, preventing the formation of regioisomers that are notoriously difficult to separate. Moreover, the mild reaction conditions prevent the decomposition of sensitive functional groups such as esters, nitriles, and halides, which might otherwise degrade under harsher acidic or basic conditions. The use of acetone as a solvent further aids in maintaining a homogeneous reaction mixture, promoting consistent heat transfer and mass transport. These factors collectively contribute to a robust process capable of delivering high-purity intermediates that meet the rigorous standards demanded by regulatory bodies for drug substance manufacturing.
How to Synthesize 5-Hydroxyfuran-2(5H)-one Derivatives Efficiently
Implementing this advanced synthetic methodology requires careful attention to reaction parameters to maximize yield and reproducibility. The process begins with the precise weighing of the indole substrate and the rhodium catalyst, followed by the addition of the sodium acetate additive to activate the catalytic species. The reaction mixture is then treated with a solution of the 4-hydroxy-2-alkynoate in acetone, ensuring thorough mixing to facilitate the initial coordination steps. Maintaining the reaction at ambient temperature while exposed to air is critical, as oxygen plays a vital role in the oxidative hydroxylation step of the catalytic cycle. Following the stipulated reaction time, standard workup procedures involving solvent removal and silica gel chromatography allow for the isolation of the target compound in high purity. For detailed operational protocols and safety guidelines, please refer to the standardized synthesis steps outlined below.
- Combine indole substrate, [Cp*RhCl2]2 catalyst (5 mol%), and NaOAc additive in a reaction vessel.
- Add acetone solvent containing the 4-hydroxy-2-alkynoate coupling partner under air atmosphere.
- Stir the mixture at 25°C for 5-24 hours, then purify via flash chromatography to isolate the target product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this rhodium-catalyzed technology offers substantial strategic benefits beyond mere chemical efficiency. The elimination of inert atmosphere requirements and high-temperature heating translates directly into reduced operational expenditures (OPEX) by lowering energy costs and simplifying reactor infrastructure needs. The use of commodity solvents like acetone and inexpensive additives like sodium acetate further drives down the raw material costs compared to processes relying on exotic reagents or cryogenic conditions. Additionally, the high functional group tolerance means that a diverse library of intermediates can be produced using a unified platform technology, reducing the need for multiple dedicated production lines and minimizing inventory complexity. This flexibility allows manufacturers to respond rapidly to changing market demands for different API precursors without significant capital investment.
- Cost Reduction in Manufacturing: The streamlined one-pot nature of this tandem reaction significantly reduces the number of unit operations required, thereby lowering labor costs and equipment occupancy time. By avoiding the use of stoichiometric oxidants and harsh reagents, the process generates less hazardous waste, leading to substantial savings in waste disposal and environmental compliance costs. The high isolated yields reported across a broad substrate scope ensure that raw material utilization is maximized, minimizing the cost per kilogram of the final intermediate. Furthermore, the ease of catalyst handling and the potential for catalyst recovery strategies can further optimize the economic viability of large-scale production runs.
- Enhanced Supply Chain Reliability: The robustness of this reaction under air and at room temperature makes it highly suitable for deployment in diverse manufacturing facilities, including those in emerging markets where specialized infrastructure may be limited. The wide availability of the starting indole and alkynoate building blocks ensures a stable supply of raw materials, mitigating the risk of shortages that often plague supply chains dependent on niche reagents. The scalability of the process, as evidenced by successful gram-scale experiments, provides confidence that production can be ramped up quickly to meet surges in demand for downstream pharmaceutical products. This reliability is crucial for maintaining continuous production schedules and meeting delivery commitments to global partners.
- Scalability and Environmental Compliance: Operating at ambient temperature and pressure inherently reduces the safety risks associated with high-energy chemical processes, facilitating easier regulatory approval for plant modifications or new installations. The use of green solvents and the generation of minimal byproducts align with modern principles of green chemistry, helping companies meet increasingly stringent environmental, social, and governance (ESG) targets. The simplified purification process reduces the consumption of silica and eluents, further decreasing the environmental footprint of the manufacturing operation. These factors collectively enhance the long-term sustainability of the supply chain, making it more resilient to regulatory changes and resource constraints.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis is essential for stakeholders evaluating its integration into existing production portfolios. The following questions address common inquiries regarding the practical implementation, scope, and limitations of this rhodium-catalyzed methodology. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation, ensuring accuracy and relevance for technical decision-makers. By clarifying these aspects, we aim to facilitate a smoother transition from laboratory discovery to commercial application.
Q: What are the key advantages of this Rh-catalyzed method over traditional synthesis?
A: This method utilizes a tandem C-H olefination/lactonization sequence under mild conditions (25°C, air), eliminating the need for harsh reagents, inert atmospheres, and multi-step protection/deprotection strategies common in older routes.
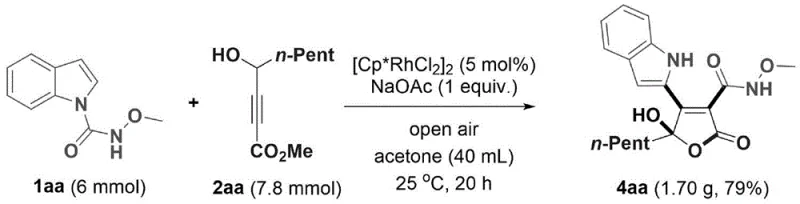
Q: Is this process scalable for commercial production?
A: Yes, the patent demonstrates successful gram-scale amplification (up to 1.70g with 79% yield), indicating robust potential for kilogram-to-ton scale manufacturing with appropriate engineering controls.
Q: What is the substrate scope for this reaction?
A: The reaction tolerates a wide range of functional groups including halogens (F, Cl, Br, I), alkyl, alkoxy, cyano, and ester groups on the indole ring, as well as various alkyl and aryl substituents on the alkynoate.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Hydroxyfuran-2(5H)-one Supplier
As the global demand for complex pharmaceutical intermediates continues to rise, partnering with an experienced CDMO like NINGBO INNO PHARMCHEM ensures access to cutting-edge synthetic technologies and reliable supply. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, leveraging our state-of-the-art facilities to deliver compounds with stringent purity specifications. Our rigorous QC labs employ advanced analytical techniques to verify the identity and quality of every batch, ensuring that our products meet the highest international standards for drug development and manufacturing. By integrating innovative methods such as the rhodium-catalyzed tandem reaction described in CN114044771B, we offer our clients a competitive edge through superior process efficiency and cost-effectiveness.
We invite you to collaborate with our technical procurement team to explore how this advanced synthesis route can optimize your supply chain and reduce your overall manufacturing costs. Contact us today to request a Customized Cost-Saving Analysis tailored to your specific project needs. Our experts are ready to provide specific COA data and comprehensive route feasibility assessments to support your R&D and commercialization goals. Let NINGBO INNO PHARMCHEM be your trusted partner in bringing next-generation therapeutics to market faster and more efficiently.
