Advanced One-Step Synthesis of Multi-Substituted Indenol Derivatives for Commercial Scale-Up
The pharmaceutical and fine chemical industries are constantly seeking efficient pathways to access complex molecular scaffolds that serve as critical building blocks for drug discovery. Patent CN102887808A introduces a groundbreaking preparation method for multi-substituted indenol derivatives, a structural motif found in significant natural products like Coleophomones D and adrenergic blockers such as Atipamezole. This technology leverages a sophisticated three-component tandem reaction involving diazo compounds, alcohols, and o-formylchalcones, catalyzed by metal complexes to achieve high diastereoselectivity in a single operational step. The ability to construct these intricate frameworks directly from simple precursors represents a significant leap forward in synthetic efficiency, offering a robust solution for generating diverse compound libraries essential for new drug screening.
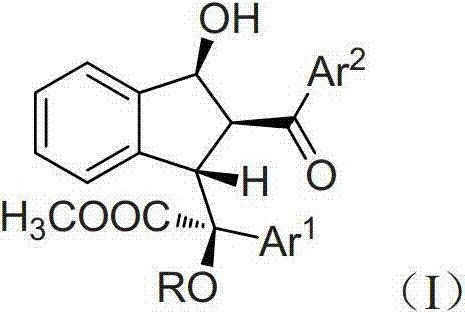
Furthermore, the versatility of this synthetic approach allows for extensive structural modification through the variation of aryl groups and alcohol substituents, enabling the rapid exploration of chemical space. The method employs rhodium carboxylates or other transition metal catalysts to facilitate the transformation under mild conditions, typically at room temperature, which drastically reduces energy consumption and operational risks associated with high-temperature processes. For procurement managers and supply chain directors, this translates to a reliable pharmaceutical intermediate supplier capability that can adapt to varying substrate requirements without compromising on yield or purity, ensuring a steady flow of high-quality materials for downstream applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of multi-substituted indenol derivatives has been plagued by significant inefficiencies that hinder large-scale production and rapid development cycles. Traditional routes often necessitate the prior construction of complex precursor compounds, such as multi-substituted indanones, followed by a selective reduction of the carbonyl group to yield the desired alcohol functionality. These multi-step sequences are not only operationally cumbersome but also suffer from narrow substrate applicability, limiting the diversity of the final product library. Moreover, the harsh reaction conditions frequently required for these transformations can lead to safety hazards and increased waste generation, creating substantial bottlenecks in cost reduction in pharmaceutical intermediate manufacturing.
The Novel Approach
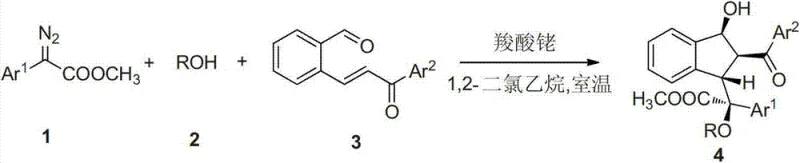
In stark contrast, the methodology disclosed in CN102887808A revolutionizes this landscape by enabling the direct assembly of the target indenol skeleton through a one-pot cascade reaction. By utilizing readily available starting materials like diazo compounds and o-formylchalcones, this novel approach eliminates the need for isolating unstable intermediates, thereby streamlining the entire workflow. The reaction proceeds with exceptional atom economy and high selectivity, driven by the precise control of the metal catalyst, which ensures that the complex molecular architecture is built with minimal byproduct formation. This shift from linear, multi-step syntheses to a convergent, one-step strategy significantly enhances the commercial scale-up of complex pharmaceutical intermediates, providing a clear pathway for industrial adoption.
Mechanistic Insights into Rhodium-Catalyzed Tandem Cyclization
The core of this technological breakthrough lies in the intricate mechanistic pathway orchestrated by the rhodium catalyst, which facilitates a series of concerted transformations. Initially, the metal catalyst promotes the decomposition of the diazo compound to generate a reactive metal carbene species. This electrophilic intermediate then interacts with the alcohol component to form an oxygen ylide, a crucial transient species that dictates the subsequent stereochemical outcome. The ylide is subsequently captured by the carbon-carbon double bond of the chalcone substrate, initiating a cyclization event that sets the stage for the final ring closure. This sequence exemplifies the power of modern organometallic catalysis in controlling reactivity and selectivity simultaneously.
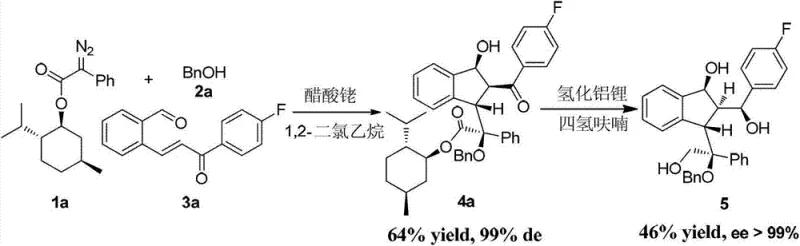
Following the initial cyclization, the resulting enol intermediate undergoes an intramolecular aldol-like addition with the pendant aldehyde group, effectively locking in the multi-substituted indenol structure in a single operation. This mechanism not only explains the high diastereoselectivity observed (often with d.r. values > 20:1) but also highlights the robustness of the process against varying electronic properties of the substrates. When chiral diazo compounds, such as phenyldiazo menthyl esters, are employed, the steric environment provided by the chiral auxiliary further enhances enantioselectivity, yielding optically pure products with ee values exceeding 99%. Such precision is vital for R&D teams focused on developing high-purity active pharmaceutical ingredients where impurity profiles must be strictly controlled.
How to Synthesize Multi-Substituted Indenol Derivatives Efficiently
Implementing this synthesis requires careful attention to reaction parameters to maximize yield and selectivity while maintaining safety standards. The process involves dissolving the diazo compound in a dry organic solvent and adding it slowly to a mixture containing the chalcone, alcohol, catalyst, and molecular sieves. The use of molecular sieves is critical as they act as water scavengers, preventing the hydrolysis of sensitive intermediates and driving the equilibrium towards the desired product. Detailed standard operating procedures regarding stoichiometry, addition rates, and purification protocols are essential for reproducibility and successful technology transfer.
- Prepare the reaction mixture by combining o-formylchalcone, alcohol, rhodium acetate catalyst, and molecular sieves in an organic solvent such as 1,2-dichloroethane.
- Dissolve the diazo compound in the organic solvent and add it dropwise to the reaction mixture at room temperature over a period of 2 hours using a peristaltic pump.
- Stir the reaction for an additional hour, filter the mixture, remove the solvent via rotary evaporation, and purify the crude product using column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented methodology offers profound advantages that directly address the pain points of modern chemical supply chains. By consolidating multiple synthetic steps into a single operation, the process inherently reduces the consumption of solvents, reagents, and energy, leading to substantial cost savings in manufacturing. The reliance on commercially available and inexpensive raw materials, such as o-phthalaldehyde derivatives and simple alcohols, ensures that the supply chain remains resilient against market fluctuations. Furthermore, the mild reaction conditions eliminate the need for specialized high-pressure or high-temperature equipment, lowering the barrier to entry for contract manufacturing organizations and enhancing overall supply continuity.
- Cost Reduction in Manufacturing: The elimination of intermediate isolation and purification steps significantly lowers the operational expenditure associated with labor and material handling. Since the reaction achieves high conversion and selectivity, the burden on downstream purification is reduced, minimizing solvent usage and waste disposal costs. This streamlined approach allows for a more competitive pricing structure for the final intermediates, providing a distinct economic advantage in the marketplace without compromising on quality standards.
- Enhanced Supply Chain Reliability: The simplicity of the raw material sourcing strategy mitigates the risk of supply disruptions. Because the precursors are commodity chemicals rather than bespoke synthetic intermediates, procurement teams can secure multiple supply sources, ensuring consistent availability. Additionally, the robustness of the reaction conditions means that production schedules are less likely to be impacted by equipment failures or safety incidents, guaranteeing reliable delivery timelines for downstream pharmaceutical clients.
- Scalability and Environmental Compliance: The process aligns well with green chemistry principles due to its high atom economy and reduced waste generation. The ability to scale this reaction from laboratory benchtop to pilot plant and eventually to commercial tonnage is facilitated by the straightforward workup procedures involving filtration and standard chromatography. This scalability ensures that the technology can meet the growing demand for these intermediates while adhering to increasingly stringent environmental regulations regarding solvent emissions and chemical waste.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis is crucial for stakeholders evaluating its potential for integration into their existing portfolios. The following questions address common inquiries regarding the scope, selectivity, and practical implementation of the technology. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for decision-making.
Q: What are the key advantages of this one-step synthesis method compared to traditional routes?
A: Traditional methods often require multi-step synthesis involving the construction of complex precursors and selective reduction of indanones, which leads to cumbersome operations and harsh reaction conditions. In contrast, this novel method utilizes a one-pot three-component reaction that operates under mild conditions (room temperature) with readily available raw materials, significantly simplifying the process and improving operational safety.
Q: What level of stereoselectivity can be achieved with this catalytic system?
A: The method demonstrates excellent stereocontrol, achieving high diastereoselectivity with d.r. values typically greater than 20:1. When using chiral auxiliaries such as L-menthyl esters, the process can yield optically pure products with ee values exceeding 99%, making it highly suitable for the synthesis of chiral pharmaceutical intermediates.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process is designed for scalability. It uses cheap and easily obtainable raw materials, simple operation procedures, and avoids hazardous conditions. The use of molecular sieves as water scavengers and standard purification techniques like column chromatography ensures that the method meets the requirements for green chemistry and industrial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Multi-Substituted Indenol Derivatives Supplier
As the demand for complex pharmaceutical intermediates continues to rise, partnering with an experienced CDMO is essential for translating innovative synthetic methods into commercial reality. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project moves seamlessly from development to market. Our rigorous QC labs and commitment to stringent purity specifications guarantee that every batch of multi-substituted indenol derivatives meets the highest industry standards, supporting your regulatory filings and clinical trial needs with confidence.
We invite you to collaborate with our technical team to explore how this advanced synthesis can optimize your supply chain and reduce overall project costs. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits specific to your volume requirements. Please contact our technical procurement team today to索取 specific COA data and route feasibility assessments, and let us help you secure a competitive edge in the global pharmaceutical market.
