Advanced Rhodium-Catalyzed Synthesis of High-Purity Tetrahydropyrrole Derivatives for Pharmaceutical Applications
The pharmaceutical and fine chemical industries are constantly seeking more efficient pathways to construct complex nitrogen-containing heterocyclic scaffolds, which serve as the backbone for numerous bioactive molecules. Patent CN109988090B introduces a groundbreaking synthetic methodology for producing tetrahydropyrrole derivatives, a critical structural motif found in alkaloids such as Kainic Acid and Cocaine. This innovation addresses the longstanding challenges associated with traditional ring-closure strategies by employing a rhodium-catalyzed coupling of diazo compounds and aryl ketenes. As a reliable pharmaceutical intermediates supplier, understanding such technological shifts is vital for maintaining a competitive edge in drug development pipelines. The disclosed method not only streamlines the synthetic route into a single operational step but also ensures exceptional stereochemical control, yielding products with high diastereoselectivity under remarkably mild conditions.
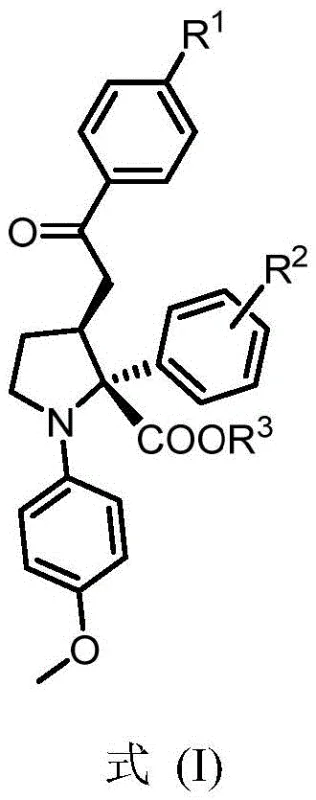
This novel approach represents a significant leap forward in the construction of polysubstituted tetrahydropyrrole rings, which are essential fragments in the synthesis of active natural products and drugs. The versatility of the method allows for various substituents at the R1, R2, and R3 positions, including alkyl, alkoxy, and halogen groups, providing a broad scope for medicinal chemistry optimization. By leveraging this technology, manufacturers can achieve cost reduction in pharmaceutical intermediates manufacturing through reduced material consumption and simplified processing. The ability to access these complex scaffolds efficiently opens new avenues for the discovery of neurodegenerative disease therapeutics, aligning with the growing demand for high-purity OLED material and specialty chemical precursors in adjacent high-tech sectors.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the tetrahydropyrrole ring skeleton has relied heavily on intramolecular ring-closure reactions. While conceptually straightforward, these traditional methods are plagued by significant practical drawbacks that hinder their efficiency in large-scale production. The primary issue lies in the propensity for numerous side reactions, which inevitably lead to complex impurity profiles that are difficult and costly to remove. Furthermore, the yields obtained from these conventional cyclization processes are often merely average, necessitating larger starting material inputs to achieve target output quantities. This inefficiency translates directly into higher production costs and increased waste generation, posing environmental and economic burdens on the supply chain. Additionally, many traditional routes require harsh reaction conditions or multiple protection-deprotection steps, further complicating the manufacturing process and extending lead times for high-purity pharmaceutical intermediates.
The Novel Approach
In stark contrast to the cumbersome legacy methods, the technology disclosed in CN109988090B utilizes a direct, intermolecular reaction between diazo compounds and aryl ketenes catalyzed by rhodium acetate. This strategy bypasses the need for pre-functionalized cyclization precursors, effectively condensing the synthetic sequence into a single, robust step. The reaction proceeds under mild temperatures ranging from 0°C to 40°C, significantly reducing energy consumption and safety risks associated with high-temperature operations. The use of 4A molecular sieves as a water absorbent ensures the stability of the catalytic system, while the choice of common organic solvents like dichloromethane facilitates easy handling and workup. This streamlined protocol not only enhances atom economy but also delivers products with superior diastereoselectivity, typically exceeding a dr value of 95:5, thereby minimizing the need for extensive downstream purification.
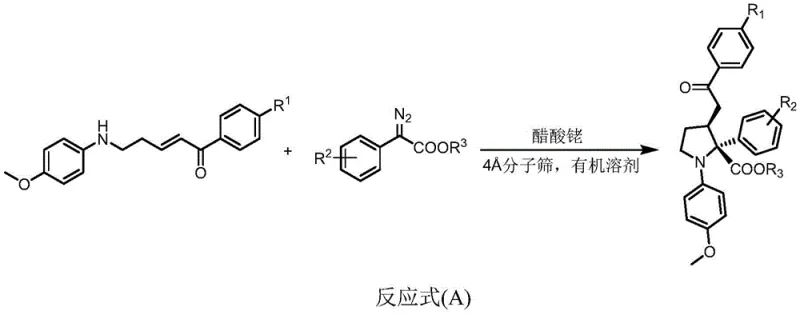
Mechanistic Insights into Rhodium-Catalyzed Cyclization
The success of this synthetic methodology hinges on the precise mechanistic pathway facilitated by the rhodium catalyst. The reaction initiates with the interaction between the diazo compound and the rhodium acetate catalyst to form a reactive rhodium metal carbene intermediate. This electrophilic species is then attacked nucleophilically by the secondary amine present in the aryl ketene substrate, leading to the formation of a crucial ammonium ylide intermediate. This ylide subsequently undergoes an intramolecular attack on the α,β-unsaturated olefin moiety, triggering the ring-closure event that forms the tetrahydropyrrole core. The spatial arrangement of the transition state, influenced by steric hindrance as depicted in the mechanism diagrams, dictates the stereochemical outcome, favoring the formation of the cis-product with high fidelity. Understanding this catalytic cycle is essential for R&D directors aiming to optimize reaction parameters for specific substrate classes.
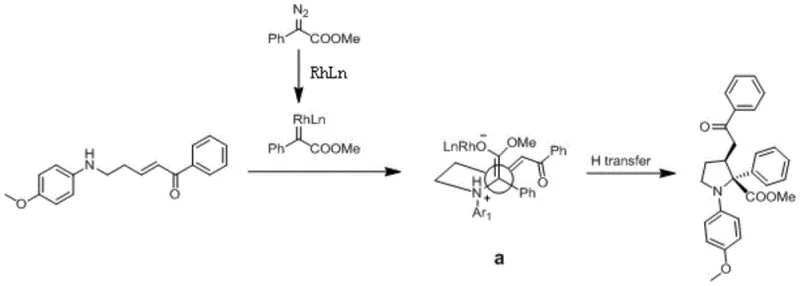
Beyond the primary transformation, the mechanism offers inherent advantages regarding impurity control, a critical concern for regulatory compliance in pharmaceutical manufacturing. The high selectivity of the rhodium carbene formation and the subsequent nucleophilic attack minimizes the generation of regioisomers and byproducts that typically complicate traditional syntheses. The rapid consumption of the diazo compound, often completed within 1 to 2 hours, reduces the window for decomposition reactions that could generate hazardous or difficult-to-remove impurities. Furthermore, the mild reaction conditions prevent thermal degradation of sensitive functional groups on the aromatic rings, preserving the integrity of the final molecule. This level of control over the reaction pathway ensures a cleaner crude product profile, which simplifies the purification process and enhances the overall yield of the target tetrahydropyrrole derivative, making it an attractive option for commercial scale-up of complex polymer additives or fine chemicals.
How to Synthesize Tetrahydropyrrole Derivatives Efficiently
Implementing this synthesis requires careful attention to reagent stoichiometry and addition rates to maximize the efficiency of the rhodium catalyst. The process begins by preparing a homogeneous mixture of the aryl ketene substrate, the rhodium acetate catalyst, and 4A molecular sieves in a suitable organic solvent such as dichloromethane. Simultaneously, the diazo compound is dissolved in the same solvent to create a feed solution. The reaction is typically conducted at a controlled temperature of 40°C, where the diazo solution is added slowly via a syringe pump to manage the exotherm and concentration of the reactive carbene species. Detailed standardized synthesis steps follow below to guide process engineers in replicating this high-yielding transformation.
- Prepare a mixed solution of aryl ketene, rhodium acetate catalyst, and 4A molecular sieves in an organic solvent such as dichloromethane.
- Dissolve the diazo compound in the same organic solvent to create a separate diazo solution.
- Add the diazo solution to the mixed solution at 40°C using a syringe pump, stir until completion, and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this rhodium-catalyzed protocol offers substantial strategic benefits beyond mere technical performance. The shift from multi-step, low-yield cyclizations to a direct one-step coupling dramatically simplifies the logistics of raw material sourcing and inventory management. By utilizing cheap and readily available compounds such as substituted phenyl diazo compounds and aryl ketenes as starting materials, the process mitigates the risk of supply chain disruptions associated with exotic or hard-to-source reagents. The reduction in reaction steps inherently lowers the cumulative cost of goods sold, as fewer unit operations mean less labor, lower utility consumption, and reduced equipment occupancy time. This efficiency translates into a more resilient supply chain capable of responding rapidly to market demands for critical pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The economic impact of this technology is driven primarily by the elimination of expensive and time-consuming purification stages associated with traditional methods. Since the reaction exhibits high diastereoselectivity and generates fewer side products, the burden on downstream processing is significantly alleviated, leading to substantial cost savings in solvent usage and chromatography media. Furthermore, the catalyst loading is minimal, typically around 0.01 molar equivalents, which optimizes the utilization of the precious rhodium metal. The overall atom economy of the reaction is high, meaning a greater proportion of the raw material mass is incorporated into the final product, reducing waste disposal costs and enhancing the sustainability profile of the manufacturing process.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route contributes directly to supply chain stability by reducing the variability often seen in complex organic syntheses. The mild reaction conditions (0°C-40°C) and short reaction times (1-2 hours) allow for flexible scheduling and faster turnaround times between batches. This agility is crucial for maintaining continuous production flows and meeting tight delivery deadlines for global clients. Additionally, the use of standard organic solvents and common laboratory equipment means that the process can be easily transferred between different manufacturing sites without requiring specialized infrastructure, thereby diversifying supply sources and reducing geopolitical risks.
- Scalability and Environmental Compliance: Scaling this process from laboratory to commercial production is facilitated by its straightforward operational parameters and lack of hazardous high-pressure or high-temperature requirements. The generation of waste is minimized due to the high selectivity and atom economy, aligning with increasingly stringent environmental regulations and corporate sustainability goals. The simplified workup procedure, often involving standard column chromatography or crystallization, reduces the volume of chemical waste requiring treatment. This environmental compatibility not only lowers compliance costs but also enhances the brand reputation of the manufacturer as a responsible producer of fine chemicals and pharmaceutical ingredients.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this tetrahydropyrrole synthesis technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity for stakeholders evaluating this method for their own production needs. Understanding these details is key to making informed decisions about process adoption and partnership opportunities.
Q: What are the key advantages of this Rh-catalyzed method over traditional cyclization?
A: Unlike traditional intramolecular ring closures which often suffer from side reactions and average yields, this method utilizes a direct intermolecular reaction between diazo compounds and aryl ketenes. It operates under mild conditions (0°C-40°C) and achieves high diastereoselectivity (dr > 95:5) in a single step.
Q: What is the role of 4A molecular sieves in this synthesis?
A: The 4A molecular sieves act as a water absorbent within the reaction system. By removing trace moisture, they help maintain the activity of the rhodium catalyst and prevent the hydrolysis of sensitive intermediates, thereby ensuring high reaction efficiency and product purity.
Q: Is this process suitable for large-scale pharmaceutical manufacturing?
A: Yes, the process is highly scalable due to its operational simplicity and mild temperature requirements. The use of readily available raw materials and the elimination of complex multi-step sequences significantly reduce production lead times and facilitate commercial scale-up for high-purity pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tetrahydropyrrole Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic methodologies like the one described in CN109988090B for the future of pharmaceutical manufacturing. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory discoveries are successfully translated into robust industrial processes. We are committed to delivering high-purity tetrahydropyrrole derivatives that meet stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical capabilities. Our expertise in rhodium-catalyzed transformations allows us to optimize these reactions for maximum yield and selectivity, providing our partners with a consistent and reliable source of critical intermediates.
We invite you to collaborate with us to leverage this cutting-edge technology for your drug development projects. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our optimized synthesis can enhance your supply chain efficiency and reduce your overall manufacturing costs. Let us be your partner in navigating the complexities of modern chemical synthesis.
