Advanced Synthesis of Fluorinated 1,3-Diketones for Commercial Scale-up
Advanced Synthesis of Fluorinated 1,3-Diketones for Commercial Scale-up
The integration of fluorine atoms into organic scaffolds represents a cornerstone strategy in modern medicinal chemistry, significantly enhancing the metabolic stability, lipophilicity, and bioavailability of drug candidates. A pivotal development in this domain is documented in patent CN108689825B, which discloses a highly efficient methodology for synthesizing 2-(trifluoroethylene)-1,3-diketone and 2-(2,2-difluoroethyl)-1,3-diketone compounds. This technology addresses the critical demand for reliable fluorine material suppliers capable of delivering complex intermediates with consistent quality. By leveraging inexpensive reagents such as trifluoroacetic anhydride and difluoroacetic anhydride, the process circumvents the need for exotic fluorinating agents, thereby streamlining the supply chain for high-value pharmaceutical intermediates. The robustness of this synthetic route ensures that manufacturers can meet the stringent purity specifications required for downstream API production while maintaining cost-effectiveness.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the introduction of fluorinated motifs onto 1,3-diketone backbones has been fraught with synthetic challenges that hinder large-scale adoption. Conventional routes often rely on harsh fluorinating reagents that are not only hazardous to handle but also generate significant amounts of toxic waste, complicating environmental compliance. Furthermore, many existing methods suffer from poor regioselectivity, leading to difficult-to-separate impurity profiles that necessitate costly purification steps like preparative HPLC. The use of transition metal catalysts in some alternative pathways introduces the risk of heavy metal contamination, requiring additional scavenging processes that erode overall process efficiency. These factors collectively contribute to extended lead times and inflated production costs, creating bottlenecks for procurement teams seeking reliable sources of fluorinated building blocks.
The Novel Approach
In stark contrast, the methodology outlined in CN108689825B offers a streamlined, one-pot solution that dramatically simplifies the manufacturing landscape. By employing trifluoroacetic anhydride or difluoroacetic anhydride as the fluorine source in the presence of triethylamine, the reaction proceeds smoothly under thermal conditions without the need for specialized catalysts. This approach not only enhances operational safety by eliminating pyrophoric reagents but also improves the atom economy of the transformation. The versatility of the method is evidenced by its compatibility with a wide array of substrates, ranging from simple phenyl rings to complex heterocycles, ensuring broad applicability across different therapeutic areas. This technological leap positions the process as a preferred choice for cost reduction in pharmaceutical intermediates manufacturing, enabling producers to deliver high-quality materials with greater speed and reliability.
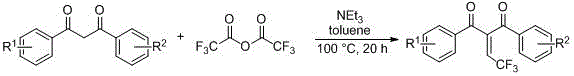
Mechanistic Insights into Anhydride-Mediated Fluorination
The core of this synthetic innovation lies in the activation of the 1,3-diketone methylene position by a mild organic base, typically triethylamine, to form a reactive enolate species. Upon addition of the fluorinated anhydride, nucleophilic attack occurs at the carbonyl carbon, establishing the new carbon-carbon bond essential for the fluorinated motif. In the case of trifluoroacetic anhydride, the subsequent elimination of trifluoroacetic acid drives the formation of the exocyclic double bond, yielding the 2-(trifluoroethylene) derivative. This elimination step is thermodynamically favorable under the specified heating conditions of 80-130°C, ensuring high conversion rates. For the difluoroethyl analogues, the reaction pathway involves acylation followed by a transformation that retains the saturated ethyl chain, showcasing the tunability of the system based on the anhydride reagent selected. Understanding these mechanistic nuances allows process chemists to fine-tune reaction parameters for optimal yield and purity.
Impurity control is inherently built into this mechanism due to the specificity of the acylation-elimination sequence. Unlike radical fluorination methods that can lead to poly-fluorinated byproducts or degradation of the aromatic rings, this ionic pathway preserves the integrity of the substrate's functional groups. The use of triethylamine as a non-nucleophilic base minimizes side reactions such as O-acylation, directing the chemistry predominantly towards C-acylation at the active methylene site. Post-reaction workup involving standard aqueous extraction and silica gel chromatography effectively removes residual amines and acid byproducts, resulting in a final product with a clean impurity profile. This level of control is crucial for R&D directors who require materials that meet rigorous regulatory standards for clinical trial applications.
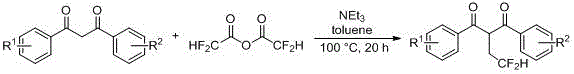
How to Synthesize 2-(Trifluoroethylene)-1,3-diketone Efficiently
Executing this synthesis requires careful attention to reaction stoichiometry and thermal management to maximize yield. The patent specifies a molar ratio range where the anhydride is used in slight excess relative to the diketone substrate, typically between 0.05 to 2.00 equivalents, to drive the equilibrium forward. Solvent selection plays a pivotal role, with toluene, tetrahydrofuran, and acetonitrile proving effective in dissolving both organic substrates and ionic intermediates. The reaction is conducted under an inert nitrogen atmosphere to prevent moisture ingress, which could hydrolyze the sensitive anhydride reagent. Following the 20-hour heating period, the crude mixture is subjected to a standard extractive workup using ethyl acetate and brine, followed by drying over anhydrous magnesium sulfate. Detailed standardized synthesis steps are provided in the guide below.
- Charge a reaction vessel with 1,3-diketone derivative, trifluoroacetic anhydride, triethylamine, and a solvent such as toluene under nitrogen atmosphere.
- Heat the mixture to 80-130°C and stir for approximately 20 hours to ensure complete conversion.
- Perform workup by extracting with ethyl acetate, washing with brine, drying over magnesium sulfate, and purifying via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this synthetic route offers profound benefits that resonate deeply with procurement managers and supply chain heads focused on resilience and efficiency. The reliance on commodity chemicals like trifluoroacetic anhydride and triethylamine decouples production from the volatility associated with specialized fluorinating agents, ensuring a stable and predictable supply chain. The simplicity of the operation, requiring only standard heating and stirring equipment, lowers the barrier to entry for contract manufacturing organizations, fostering a competitive supplier landscape that drives down costs. Moreover, the high yields reported in key examples, such as the 85% yield for the diphenyl derivative, translate directly into reduced raw material consumption per kilogram of output, enhancing overall process economics.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and cryogenic cooling requirements significantly lowers the operational expenditure (OPEX) associated with production. By utilizing a thermal process that operates at atmospheric pressure, facilities can avoid the capital investment needed for high-pressure reactors or specialized containment systems. The use of recyclable solvents like toluene further contributes to cost savings by minimizing waste disposal fees and solvent purchase costs. These cumulative efficiencies allow suppliers to offer more competitive pricing structures without compromising on the quality of the fluorinated intermediates.
- Enhanced Supply Chain Reliability: The availability of raw materials is a critical factor in maintaining uninterrupted production schedules. Since trifluoroacetic anhydride and 1,3-diketone derivatives are produced on a multi-ton scale globally, the risk of supply shortages is markedly reduced compared to niche fluorine sources. The robustness of the reaction conditions also means that the process is less susceptible to minor fluctuations in utility supplies, such as cooling water temperature variations. This inherent stability ensures that delivery timelines remain consistent, allowing pharmaceutical companies to plan their clinical and commercial manufacturing campaigns with greater confidence.
- Scalability and Environmental Compliance: Scaling this process from gram to kilogram quantities involves linear adjustments rather than fundamental changes in chemistry, reducing the technical risk associated with technology transfer. The absence of heavy metals simplifies the environmental permitting process and reduces the burden of wastewater treatment, aligning with increasingly strict global environmental regulations. The generation of trifluoroacetic acid as a byproduct can be managed through standard neutralization protocols, ensuring that the facility maintains a low environmental footprint while achieving substantial production volumes.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these fluorinated compounds. They are derived from the specific experimental data and beneficial effects described in the patent documentation, providing clarity for potential partners evaluating this technology for their supply chains.
Q: What are the primary advantages of using trifluoroacetic anhydride in this synthesis?
A: Using trifluoroacetic anhydride allows for a direct, one-step introduction of the trifluoroethylene moiety onto the 1,3-diketone scaffold. This method avoids complex multi-step sequences often required for fluorination, utilizing cheap and readily available reagents to achieve high yields, such as the 85% yield observed in benchmark examples.
Q: Can this method be applied to substrates with electron-withdrawing groups?
A: Yes, the patent demonstrates excellent substrate tolerance. Examples include substrates with cyano, fluoro, chloro, and bromo substituents. While yields may vary depending on steric and electronic effects, the protocol remains effective across a broad range of aromatic substitutions, making it suitable for diverse API intermediate libraries.
Q: Is the process scalable for industrial production?
A: The process utilizes standard organic solvents like toluene and common bases like triethylamine, operating at moderate temperatures (80-130°C). The absence of sensitive transition metal catalysts or cryogenic conditions simplifies the engineering requirements, facilitating straightforward scale-up from laboratory to commercial manufacturing volumes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-(Trifluoroethylene)-1,3-diketone Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of fluorinated intermediates in the development of next-generation therapeutics and agrochemicals. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can support your needs from early-stage discovery through to full-scale commercialization. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 2-(trifluoroethylene)-1,3-diketone meets the highest industry standards. Our commitment to quality and consistency makes us a trusted partner for multinational corporations seeking to secure their supply of critical building blocks.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis method can be integrated into your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of switching to this efficient route. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your unique molecular targets, ensuring a seamless path from concept to commercial reality.
