Advanced Metal-Free Manufacturing of N-Substituted Benzothiazin-4-one Pharmaceutical Intermediates
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable, and environmentally benign synthetic routes for heterocyclic scaffolds that serve as critical building blocks for drug discovery. A significant breakthrough in this domain is documented in Chinese Patent CN114181169B, which discloses a highly efficient, metal-free method for preparing N-(substituted)benzothiazin-4-one derivatives. These compounds are renowned for their diverse biological activities, including potent anti-tumor, anti-malarial, and myocardial protective properties, making them invaluable assets in the pipeline of modern medicinal chemistry. The core innovation lies in the strategic utilization of a chloromethyl transfer agent combined with an iodide-mediated activation system, which facilitates the simultaneous construction of C-S and C-N bonds without the need for transition metal catalysts. This technological leap addresses long-standing challenges regarding heavy metal residues and complex purification protocols that have historically plagued the synthesis of these privileged structures. For global procurement and R&D teams, this patent represents a pivotal shift towards greener manufacturing paradigms that align with stringent regulatory standards while maintaining high operational efficiency.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of N-substituted benzothiazin-4-one compounds has been fraught with significant technical and economic hurdles that impede large-scale production. The first generation of synthetic strategies relied heavily on N-substituted-2-methylsulfinylbenzamide precursors activated by tert-butyldimethyl(1-methoxyvinyloxy)silane and zinc iodide. This approach is fundamentally flawed due to the reliance on expensive, moisture-sensitive, and toxic silicon reagents, which complicate handling and waste disposal. Furthermore, the prerequisite preparation of the sulfoxide starting material from corresponding thioethers adds unnecessary synthetic steps, drastically reducing overall atom economy and throughput. A second conventional pathway involves the condensation of o-carboxybenzenethiol with formaldehyde and amines in refluxing toluene. While conceptually simple, this method suffers from abysmal yields, typically hovering between 40% and 50%, and utilizes volatile, pungent, and hazardous raw materials like formaldehyde and thiols that pose severe occupational health risks. A third reported method employs silver oxide as a promoter alongside Selectfluor derivatives; however, the mandatory use of stoichiometric silver salts introduces prohibitive costs and creates a massive burden for removing trace heavy metals to meet pharmaceutical purity specifications.
The Novel Approach
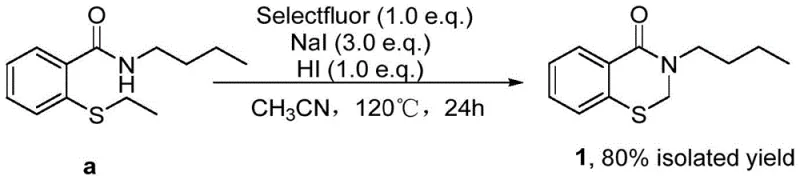
In stark contrast to these legacy technologies, the methodology outlined in CN114181169B offers a streamlined, one-pot solution that elegantly bypasses the need for precious metals or toxic silanes. The novel process utilizes readily available N-(substituted)-2-sulfanylbenzamides as starting materials, which undergo a transformative cyclization in the presence of 1-chloromethyl-4-fluoro-1,4-diazabicyclo[2.2.2]octane di(tetrafluoroborate) salt, sodium iodide, and hydroiodic acid. By operating in acetonitrile at 120°C, this system achieves remarkable conversion rates, exemplified by the synthesis of N-(n-butyl)benzothiazin-4-one in an impressive 80% isolated yield. The reaction mechanism is driven by the in situ generation of reactive iodine species that facilitate C-S bond cleavage followed by the transfer of a chloromethyl group from the additive to the substrate. This dual-activation strategy not only accelerates the reaction kinetics but also ensures high selectivity, minimizing the formation of side products that often complicate downstream processing. The elimination of silver and silicon reagents translates directly into a cleaner reaction profile and a significantly simplified workup procedure.
Mechanistic Insights into Metal-Free C-S and C-N Bond Construction
The mechanistic elegance of this transformation lies in its ability to orchestrate complex bond formations through a purely organic and halogen-mediated pathway. The reaction initiates with the activation of the sulfanyl group on the benzamide substrate, likely involving an oxidative addition or halogen exchange process mediated by the iodide/acid system. This activation weakens the C-S bond of the alkylthio group, rendering it susceptible to cleavage. Concurrently, the 1-chloromethyl-4-fluoro-1,4-diazabicyclo[2.2.2]octane di(tetrafluoroborate) salt serves as a crucial electrophilic methylene source. Unlike traditional methylation agents that might lead to over-alkylation or polymerization, this rigid bicyclic structure delivers the chloromethyl unit with high precision. The subsequent intramolecular nucleophilic attack by the amide nitrogen onto the activated methylene carbon closes the six-membered ring, establishing the stable benzothiazin-4-one core. This concerted mechanism avoids the formation of unstable radical intermediates often seen in metal-catalyzed cycles, thereby enhancing the reproducibility and safety of the process on a kilogram scale.
From an impurity control perspective, the absence of transition metals is a game-changer for pharmaceutical quality assurance. Traditional silver-catalyzed routes often leave behind trace amounts of silver ions that are notoriously difficult to remove and can catalyze degradation pathways in the final drug substance. By employing a metal-free protocol, the risk of heavy metal contamination is effectively nullified, simplifying the analytical validation required for regulatory filings. Furthermore, the use of hydroiodic acid and sodium iodide generates soluble inorganic salts that are easily removed during the aqueous workup or column chromatography stages. The substrate scope studies indicate that the electronic nature of the N-substituent has a moderate influence on the reaction efficiency, with alkyl groups like n-butyl performing slightly better than bulky cyclohexyl or aromatic benzyl groups. This suggests that steric hindrance around the nitrogen center is the primary factor governing reaction rates, providing valuable guidance for process optimization when scaling up diverse analogues.
How to Synthesize N-Substituted Benzothiazin-4-one Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires strict adherence to the optimized parameters defined in the patent to maximize yield and purity. The process is designed to be operationally simple, utilizing standard glassware and heating equipment without the need for specialized inert atmosphere techniques beyond standard sealing. The key to success lies in the precise molar ratios of the activators; specifically, maintaining a 1:3:1 ratio of substrate to sodium iodide to hydroiodic acid is critical for driving the equilibrium towards the cyclized product. Deviation from these stoichiometric balances can lead to incomplete conversion or the accumulation of uncyclized intermediates. The reaction temperature of 120°C in acetonitrile provides the necessary thermal energy to overcome the activation barrier for C-S bond cleavage while keeping the solvent in a manageable liquid phase under pressure. For detailed operational procedures, safety data sheets, and specific troubleshooting guides regarding quenching and isolation, please refer to the standardized synthesis protocol below.
- Charge a sealed reaction vessel with acetonitrile solvent, N-(substituted)-2-sulfanylbenzamide substrate, 1-chloromethyl-4-fluoro-1,4-diazabicyclo[2.2.2]octane di(tetrafluoroborate) salt, sodium iodide, and hydroiodic acid.
- Heat the reaction mixture to 120°C under vigorous stirring conditions and maintain this temperature for approximately 24 hours to ensure complete conversion.
- Upon completion, concentrate the reaction solution under reduced pressure and purify the crude residue via column chromatography to isolate the target N-(substituted)benzothiazin-4-one.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this metal-free technology offers substantial strategic advantages that extend far beyond simple yield improvements. The primary value proposition is the drastic reduction in raw material costs associated with eliminating precious metal catalysts. Silver oxide, used in previous generations of this chemistry, is a high-cost commodity with volatile pricing dynamics; replacing it with commodity chemicals like sodium iodide and hydroiodic acid stabilizes the cost of goods sold (COGS) and insulates the supply chain from geopolitical fluctuations in the precious metals market. Additionally, the removal of toxic silane reagents reduces the complexity of hazardous material handling and storage, lowering insurance premiums and compliance overheads. The simplified purification process, which avoids the need for specialized scavenger resins to remove heavy metals, further accelerates the production cycle time, allowing for faster turnover of manufacturing suites and increased overall capacity utilization.
- Cost Reduction in Manufacturing: The economic impact of switching to this metal-free protocol is profound, primarily driven by the substitution of expensive stoichiometric oxidants with inexpensive inorganic salts. By eliminating the need for silver oxide and complex silane reagents, the direct material costs are significantly lowered, creating immediate margin expansion opportunities for high-volume production. Furthermore, the simplified workup procedure reduces the consumption of silica gel and solvents during purification, contributing to additional savings in waste disposal and utility costs. The high isolated yields reported, such as 80% for the n-butyl derivative, ensure that raw material utilization is maximized, minimizing the financial loss associated with unreacted starting materials and byproducts.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the reliance on globally available, commodity-grade reagents rather than specialized catalysts that may have single-source suppliers. Sodium iodide, hydroiodic acid, and acetonitrile are produced by multiple manufacturers worldwide, ensuring a continuous and competitive supply stream that mitigates the risk of production stoppages due to vendor shortages. The robustness of the reaction conditions also means that the process is less sensitive to minor variations in reagent quality, allowing for greater flexibility in sourcing strategies. This reliability is crucial for maintaining consistent delivery schedules to downstream pharmaceutical customers who depend on just-in-time inventory models for their clinical and commercial programs.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this process aligns perfectly with modern green chemistry principles, facilitating easier regulatory approval for commercial scale-up. The absence of heavy metals simplifies the environmental impact assessment and reduces the burden on wastewater treatment facilities, as effluent streams do not require complex metal precipitation steps. The use of acetonitrile, a common industrial solvent with well-established recovery and recycling infrastructure, further enhances the sustainability profile of the manufacturing process. These factors collectively reduce the time and capital expenditure required to build or retrofit production facilities, enabling rapid deployment of commercial-scale manufacturing lines for these high-value intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthetic route. These insights are derived directly from the experimental data and comparative analysis presented in the patent documentation, providing a clear understanding of the technology's capabilities and limitations. Understanding these nuances is essential for R&D teams evaluating the feasibility of integrating this chemistry into their existing pipelines and for procurement specialists assessing the long-term viability of the supply source.
Q: What are the critical reagents required for this metal-free cyclization?
A: The process relies on a specific combination of 1-chloromethyl-4-fluoro-1,4-diazabicyclo[2.2.2]octane di(tetrafluoroborate) salt as the methylene source, sodium iodide (3.0 equivalents), and hydroiodic acid (1.0 equivalent) in acetonitrile solvent.
Q: How does this method improve upon traditional silver-catalyzed routes?
A: Unlike prior art methods requiring expensive and toxic silver oxide or cumbersome silane reagents, this novel approach operates under metal-free conditions, significantly simplifying purification and reducing heavy metal contamination risks in the final API intermediate.
Q: What is the substrate scope for the N-substituent group?
A: The methodology demonstrates excellent versatility, successfully accommodating various N-substituents including n-butyl, methyl, benzyl, and cyclohexyl groups with isolated yields ranging from 58% to 80%.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-Substituted Benzothiazin-4-one Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the metal-free synthesis described in CN114181169B and have integrated this advanced methodology into our state-of-the-art development portfolio. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from bench-scale discovery to industrial manufacturing is seamless and efficient. Our rigorous QC labs and stringent purity specifications guarantee that every batch of N-substituted benzothiazin-4-one intermediates meets the exacting standards required for global pharmaceutical applications. We are committed to delivering high-purity benzothiazin-4-one derivatives that empower your drug discovery programs with reliable, high-quality building blocks.
We invite you to collaborate with us to leverage this cutting-edge technology for your specific project needs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating exactly how this metal-free route can optimize your budget. Please contact our technical procurement team today to request specific COA data for our current inventory and to discuss comprehensive route feasibility assessments for your target molecules. Let us be your partner in accelerating the development of next-generation therapeutics through superior chemical innovation.
