Advanced Manganese-Catalyzed Synthesis of Chromone-3-Phosphonate Derivatives for Commercial Scale-Up
Advanced Manganese-Catalyzed Synthesis of Chromone-3-Phosphonate Derivatives for Commercial Scale-Up
The pharmaceutical and agrochemical industries continuously demand efficient, scalable, and environmentally benign synthetic routes for complex heterocyclic intermediates. Patent CN101497629B introduces a transformative methodology for the preparation of chromone-3-phosphonate derivatives, utilizing manganese(III) acetate as a key mediator. This innovation addresses critical bottlenecks in traditional synthesis by enabling reactions under significantly milder thermal conditions, ranging from 20°C to 100°C, compared to the harsh 190°C required by legacy protocols. For R&D directors and procurement specialists, this represents a pivotal shift towards safer, more cost-effective manufacturing of high-purity pharmaceutical intermediates. The technology leverages widely available chromone and flavone scaffolds, expanding the accessible chemical space for drug discovery while ensuring robust supply chain continuity through simplified operational parameters.
The Limitations of Conventional Methods vs. The Novel Approach
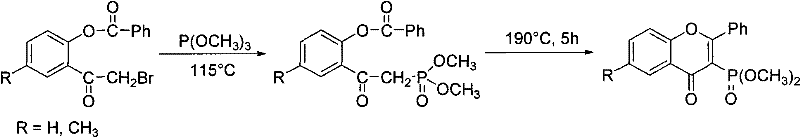
The Limitations of Conventional Methods
Prior art methodologies for synthesizing flavone-3-phosphonates, such as those reported by Kostka and Budzisz, rely on extremely vigorous reaction conditions that pose significant safety and efficiency challenges. As illustrated in the historical context of the field, these conventional routes often necessitate heating reactants to temperatures as high as 190°C to drive the phosphorylation forward. Such extreme thermal stress frequently leads to the decomposition of sensitive organic substrates, resulting in poor atom economy and inconsistent product quality. Furthermore, the starting materials required for these older methods are often difficult to source commercially, creating supply chain vulnerabilities and driving up raw material costs. The combination of low yields, typically ranging from 18% to 47%, and the generation of complex impurity profiles due to thermal degradation makes these legacy processes unsuitable for modern GMP-compliant manufacturing environments.
The Novel Approach
The disclosed invention fundamentally reengineers the synthetic landscape by employing manganese(III) acetate as an oxidative promoter in conjunction with phosphite esters. This novel approach allows the reaction to proceed efficiently at temperatures between 20°C and 100°C, drastically reducing energy consumption and eliminating the risk of thermal decomposition associated with previous techniques. By utilizing chromone derivatives as initiators, the method accommodates a vast array of substituents, including alkyl, alkoxy, halogen, and nitro groups, thereby offering unparalleled versatility for medicinal chemists. The use of green solvents such as ethanol and acetic acid not only aligns with stringent environmental regulations but also simplifies downstream processing and waste management. This strategic shift from harsh thermal conditions to mild catalytic mediation ensures higher reproducibility and facilitates the transition from laboratory benchtop to pilot plant operations without compromising yield or purity.
Mechanistic Insights into Manganese(III) Acetate Mediated Radical Phosphorylation
The core of this technological advancement lies in the unique reactivity of manganese(III) acetate, which acts as a single-electron oxidant to generate carbon-centered radicals on the chromone scaffold. In this mechanism, the manganese species facilitates the homolytic cleavage or activation necessary to functionalize the electron-deficient double bond at the 3-position of the chromone ring. The reaction proceeds through a radical addition pathway where the phosphite ester adds across the activated olefinic bond, followed by oxidation and elimination steps to restore aromaticity or conjugation within the heterocyclic system. This radical-mediated process is highly regioselective, ensuring that the phosphonate group is installed specifically at the C-3 position, which is crucial for the biological activity of the resulting flavonoid derivatives. Understanding this mechanistic nuance allows process chemists to fine-tune stoichiometry, typically maintaining a chromone to manganese acetate molar ratio of 1:3, to maximize conversion efficiency.
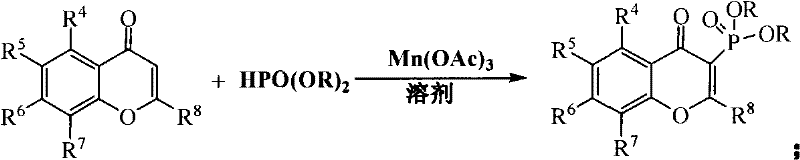
Impurity control in this system is inherently superior due to the mild reaction environment which suppresses side reactions such as polymerization or ring-opening that are common at elevated temperatures. The compatibility of the method with diverse substrates, including 2-phenylchromones and 2-heteroaromatic chromones, demonstrates the robustness of the catalytic cycle against electronic variations in the substrate. For instance, electron-withdrawing groups like nitro or halogen substituents on the phenyl ring do not inhibit the reaction, allowing for the synthesis of a broad library of analogs essential for SAR studies. The use of solvents like ethanol further aids in stabilizing intermediate species and facilitating heat transfer, ensuring a homogeneous reaction mixture that minimizes localized hot spots and potential degradation pathways. This level of mechanistic control translates directly to a cleaner crude product profile, reducing the burden on purification teams and increasing overall throughput.
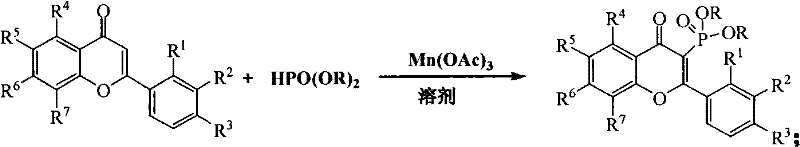
How to Synthesize Chromone-3-Phosphonate Efficiently
Implementing this synthesis protocol requires careful attention to reagent quality and reaction monitoring to ensure optimal outcomes. The process begins with the dissolution of the selected chromone derivative, such as 2-phenyl-4H-benzopyran-4-one, and the phosphite ester, such as diethyl phosphite, in a solvent like absolute ethanol. Manganese(III) acetate is then added to the mixture, serving as the critical oxidant that drives the transformation. The reaction vessel is heated, typically to around 90°C, and maintained under these conditions while monitoring progress via thin-layer chromatography (TLC). Once the starting material is consumed, usually within 2 hours, the reaction is quenched, and the product is isolated through standard workup procedures involving column chromatography. Detailed standardized synthesis steps are provided in the guide below to assist technical teams in replicating these results accurately.
- Dissolve the chromone or flavone derivative, phosphite ester, and manganese(III) acetate in a suitable solvent such as ethanol or acetic acid.
- Heat the reaction mixture to a temperature between 20°C and 100°C, preferably around 90°C, and maintain stirring.
- Monitor the reaction progress via TLC until completion, typically within 2 hours, then isolate the product via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented methodology offers substantial benefits that directly impact the bottom line and operational resilience of chemical manufacturing enterprises. The shift to mild reaction conditions and the use of commodity-grade solvents like ethanol and acetic acid significantly lowers the barrier to entry for production, reducing both capital expenditure on specialized high-temperature equipment and operational costs related to energy consumption. The availability of diverse chromone starting materials ensures that supply chains remain robust, mitigating the risk of production stoppages due to raw material shortages. Furthermore, the simplified post-treatment process, which avoids complex extraction or neutralization steps often required by harsher chemistries, streamlines the manufacturing workflow and reduces labor hours per batch. These factors collectively contribute to a more agile and cost-efficient production model capable of meeting the dynamic demands of the global pharmaceutical market.
- Cost Reduction in Manufacturing: The elimination of extreme thermal requirements and the adoption of inexpensive, green solvents drastically reduce utility costs and solvent procurement expenses. By avoiding the need for specialized high-pressure or high-temperature reactors, facilities can utilize existing standard glass-lined or stainless-steel equipment, deferring capital investment. The higher yields achieved through this method mean less raw material is wasted per kilogram of final product, directly improving the cost of goods sold (COGS). Additionally, the reduced formation of thermal degradation byproducts minimizes the loss of valuable intermediates during purification, further enhancing overall process economics and profitability margins for large-scale production runs.
- Enhanced Supply Chain Reliability: The reliance on widely available chromone derivatives and common phosphite esters ensures a stable and diversified supply base, reducing dependency on niche suppliers. The flexibility to use various solvents, including ethanol and acetic acid, provides procurement teams with options to switch suppliers based on regional availability and pricing fluctuations without affecting reaction performance. This adaptability is crucial for maintaining continuous production schedules in the face of global logistics disruptions. Moreover, the mild conditions extend the lifespan of reactor vessels and ancillary equipment by reducing corrosion and thermal stress, leading to lower maintenance downtime and more consistent output volumes over the long term.
- Scalability and Environmental Compliance: The use of green solvents aligns perfectly with increasingly stringent environmental regulations, simplifying the permitting process for new manufacturing lines and reducing waste disposal costs. The straightforward workup procedure involving column chromatography is easily adaptable to preparative HPLC or crystallization methods on a larger scale, ensuring that purity specifications can be met consistently. The absence of toxic heavy metal catalysts or hazardous reagents simplifies effluent treatment and reduces the environmental footprint of the facility. This compliance advantage not only mitigates regulatory risk but also enhances the corporate sustainability profile, which is becoming a key criterion for partnerships with major multinational pharmaceutical companies.
Frequently Asked Questions (FAQ)
The following questions address common technical and operational inquiries regarding the implementation of this manganese-catalyzed phosphorylation technology. These insights are derived directly from the experimental data and scope defined in the patent documentation, providing clarity for process development teams. Understanding these nuances is essential for successful technology transfer and scale-up activities. The answers reflect the specific advantages of this route over traditional methods, focusing on yield optimization and substrate compatibility.
Q: What are the advantages of using manganese acetate over conventional high-temperature methods?
A: The manganese acetate mediated method operates at significantly milder temperatures (20-100°C) compared to conventional methods requiring 190°C, preventing substrate decomposition and improving overall yield and safety.
Q: Which solvents are compatible with this phosphorylation reaction?
A: The process supports a wide range of green and common organic solvents including ethanol, acetic acid, acetonitrile, and toluene, with ethanol often providing optimal results for solubility and environmental compliance.
Q: Is this method scalable for industrial production of pharmaceutical intermediates?
A: Yes, the use of readily available raw materials, simple workup procedures involving standard column chromatography, and mild reaction conditions makes this route highly suitable for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chromone-3-Phosphonate Supplier
NINGBO INNO PHARMCHEM stands at the forefront of custom synthesis, leveraging advanced catalytic technologies like the manganese-mediated phosphorylation described in CN101497629B to deliver high-value intermediates. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from gram-scale optimization to full industrial manufacturing. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of chromone-3-phosphonate derivative meets the exacting standards required for API synthesis. Our commitment to quality and consistency makes us a trusted partner for complex organic synthesis projects globally.
We invite you to engage with our technical procurement team to discuss how this innovative synthetic route can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of switching to this greener, more efficient methodology. We encourage potential partners to contact us for specific COA data and route feasibility assessments to validate the suitability of these intermediates for your pipeline. Let us collaborate to accelerate your drug development timeline with reliable, high-quality chemical solutions.
