Advanced Synthetic Route for 3,4-Disubstituted Isoxazoles Enabling Scalable Pharmaceutical Manufacturing
Introduction to Patent CN102659705A and Isoxazole Significance
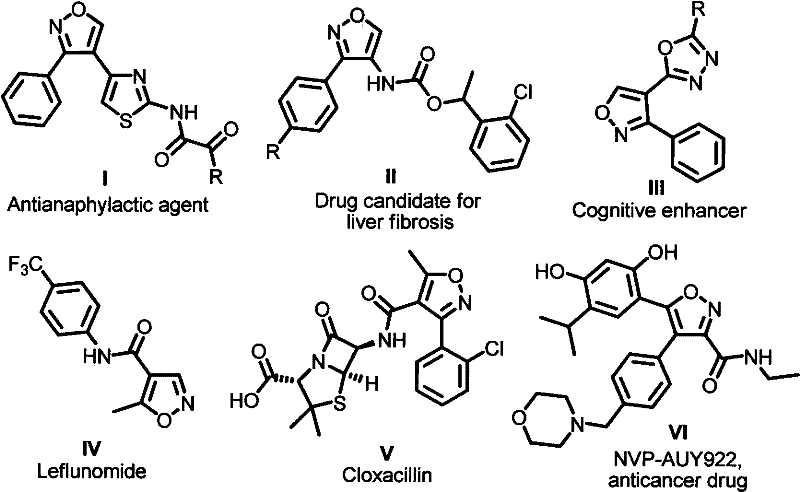
The pharmaceutical industry constantly seeks robust and scalable synthetic methodologies for constructing privileged heterocyclic scaffolds, among which the 3,4-disubstituted isoxazole core stands out due to its pervasive presence in bioactive molecules. Patent CN102659705A discloses a groundbreaking synthetic method for these compounds, utilizing N-hydroxychloroimide compounds, aldehydes, and secondary amines as primary building blocks. This innovation addresses critical bottlenecks in medicinal chemistry by providing a route that is not only operationally simple but also highly efficient, yielding products with significant pharmacological potential including anticancer, antiallergic, and antitumor activities. The versatility of this scaffold is evidenced by its occurrence in diverse therapeutic agents ranging from antianaphylactic agents to cognitive enhancers and anticancer drugs like NVP-AUY922. For R&D directors and procurement specialists, understanding the nuances of this patent is essential for securing a reliable pharmaceutical intermediate supplier capable of delivering high-purity materials for drug development pipelines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of 3,4-disubstituted isoxazole rings has been plagued by synthetic inefficiencies that hinder large-scale manufacturing. Traditional approaches often rely on multi-step reaction sequences that accumulate impurities and reduce overall throughput. Furthermore, many established protocols necessitate the use of transition metal catalysts, which introduce significant challenges regarding residual metal removal—a critical quality attribute for active pharmaceutical ingredients (APIs). These metal-catalyzed processes frequently require stringent anhydrous conditions, specialized equipment, and expensive ligands, driving up the cost of goods sold (COGS). Additionally, conventional methods often suffer from low atom economy and poor regioselectivity, leading to difficult purification scenarios that delay project timelines. For supply chain heads, these complexities translate into longer lead times and higher risks of batch failure, making the search for alternative, metal-free methodologies a strategic priority for cost reduction in pharmaceutical intermediate manufacturing.
The Novel Approach
The methodology described in CN102659705A represents a paradigm shift by employing a base-mediated cascade reaction that bypasses the need for precious metal catalysts. This novel approach utilizes inexpensive and commercially available starting materials—N-hydroxychloroimides, aldehydes, and secondary amines—which are stable and do not require special storage conditions, thereby enhancing supply chain reliability. The reaction proceeds under mild conditions, typically initiating at 0°C and warming to room temperature, which minimizes energy consumption and safety hazards associated with exothermic events. By streamlining the synthesis into a concise sequence that forms the 4,5-dihydroisoxazole intermediate followed by a straightforward oxidation step, this method drastically simplifies the operational workflow. The high yields reported across various substrates demonstrate the robustness of this chemistry, offering a practical solution for the commercial scale-up of complex pharmaceutical intermediates while maintaining stringent purity specifications.
Mechanistic Insights into Base-Mediated Cyclization and Oxidation
The core of this synthetic strategy involves a sophisticated yet elegant mechanism where the interplay between the nucleophilic secondary amine, the electrophilic aldehyde, and the N-hydroxychloroimide dictates the formation of the heterocyclic ring. Initially, the base facilitates the generation of reactive species from the N-hydroxychloroimide, which then engages in a cycloaddition-like process with the aldehyde and amine components. This concerted interaction leads to the formation of the 4,5-dihydroisoxazole intermediate (Compound 4 in the patent schema), establishing the fundamental carbon-nitrogen-oxygen framework. The choice of base is critical, with organic bases like triethylamine proving highly effective in promoting this transformation without inducing side reactions. The mechanistic pathway ensures high regioselectivity, minimizing the formation of isomeric byproducts that often complicate downstream processing. This level of control is paramount for R&D teams focused on impurity profiling and ensuring that the final API meets regulatory standards for chemical identity and purity.
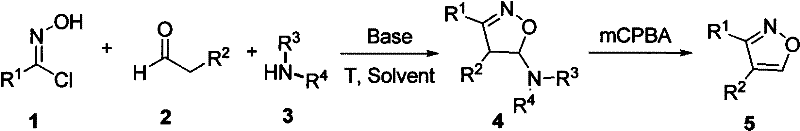
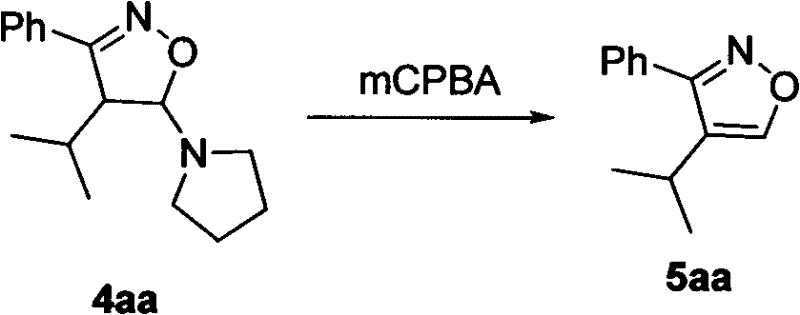
Following the cyclization, the subsequent oxidation step is pivotal for aromatizing the ring system to achieve the final bioactive conformation. The patent specifies the use of mCPBA (meta-chloroperoxybenzoic acid) as the oxidant, which efficiently dehydrogenates the 4,5-dihydroisoxazole intermediate to yield the fully aromatic 3,4-disubstituted isoxazole. This oxidation is conducted at room temperature, further underscoring the mild nature of the overall process. The mechanism likely involves the formation of an N-oxide or similar transient species that eliminates to restore aromaticity. Understanding this oxidative transformation is crucial for process chemists aiming to optimize reaction times and stoichiometry. The ability to perform this oxidation with high conversion rates, as evidenced by the disappearance of starting materials monitored by TLC, ensures that the process is not only chemically sound but also practically viable for industrial applications where consistency and reproducibility are non-negotiable requirements.
How to Synthesize 3,4-Disubstituted Isoxazole Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires careful attention to reaction parameters to maximize yield and purity. The process begins with the dissolution of the secondary amine and base in a suitable solvent such as dichloromethane (DCM) or toluene, followed by cooling to 0°C to control the initial exotherm upon addition of the aldehyde. The N-hydroxychloroimide is then introduced portion-wise to maintain reaction homogeneity and prevent localized concentration spikes. After the formation of the dihydro-intermediate, the mixture is warmed to room temperature to complete the cyclization. The subsequent oxidation step involves the addition of 1.5 equivalents of mCPBA, allowing the reaction to proceed until TLC analysis confirms the complete consumption of the intermediate. Detailed standardized synthetic steps for this protocol are provided in the guide below, ensuring that technical teams can replicate the high yields reported in the patent data.
- React N-hydroxychloroimide, aldehyde, and secondary amine in a solvent with a base at 0°C to room temperature to form 4,5-dihydroisoxazole.
- Purify the intermediate 4,5-dihydroisoxazole compound via flash column chromatography.
- Oxidize the purified intermediate using mCPBA (1.5 equivalents) at room temperature to obtain the final 3,4-disubstituted isoxazole product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthetic route offers substantial strategic benefits for procurement managers and supply chain leaders looking to optimize their vendor networks. The primary advantage lies in the significant cost reduction in manufacturing driven by the elimination of expensive transition metal catalysts and ligands. By relying on commodity chemicals like triethylamine and common aldehydes, the raw material costs are drastically lowered, and the supply risk is minimized since these reagents are sourced from a broad global market. Furthermore, the simplicity of the workup procedure, which typically involves standard extraction and column chromatography, reduces the demand for specialized purification resins or complex distillation setups. This operational simplicity translates directly into lower capital expenditure (CAPEX) for manufacturing facilities and reduced operating expenses (OPEX), making the final isoxazole intermediates more price-competitive in the global marketplace.
- Cost Reduction in Manufacturing: The exclusion of precious metals removes the necessity for costly metal scavenging steps and rigorous testing for residual metals, which are major cost drivers in API production. Additionally, the high yields achieved across a wide substrate scope mean less raw material waste and higher throughput per batch. The use of ambient temperature conditions for significant portions of the reaction also lowers energy costs associated with heating or cryogenic cooling, contributing to a leaner and more economical production model that enhances profit margins for downstream drug manufacturers.
- Enhanced Supply Chain Reliability: Since the starting materials are industrial commodities with wide sources, the risk of supply disruption due to single-source dependency is virtually eliminated. The stability of the reagents allows for bulk purchasing and long-term storage without degradation, enabling manufacturers to maintain healthy inventory levels and respond quickly to fluctuating market demands. This robustness ensures reducing lead time for high-purity pharmaceutical intermediates, allowing drug developers to accelerate their clinical trial timelines without being bottlenecked by material shortages or complex logistics associated with hazardous or unstable reagents.
- Scalability and Environmental Compliance: The process is inherently designed for scalability, with reaction conditions that are easily transferable from gram-scale laboratory synthesis to ton-scale industrial production. The mild reaction environment reduces the generation of hazardous waste streams, aligning with modern green chemistry principles and stringent environmental regulations. This compliance reduces the burden on waste treatment facilities and minimizes the environmental footprint of the manufacturing process, a key consideration for companies aiming to meet sustainability goals while ensuring the continuous supply of critical drug substances.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and claims within patent CN102659705A, providing a factual basis for decision-making. Understanding these details helps stakeholders evaluate the feasibility of integrating this method into their existing production workflows. The answers cover aspects ranging from reaction optimization to the specific advantages of the oxidation step, ensuring a comprehensive understanding of the technology's value proposition.
Q: What are the key advantages of this synthesis method over traditional metal-catalyzed routes?
A: This method eliminates the need for expensive transition metal catalysts and complex multi-step sequences. It utilizes readily available industrial commodities as raw materials, operates under mild conditions (0°C to room temperature), and achieves high yields, significantly simplifying the purification process and reducing environmental waste.
Q: Can this process be scaled for industrial production of API intermediates?
A: Yes, the patent explicitly states the method is suitable for industrial scale production. The use of stable, cheap raw materials, simple operation procedures, and the absence of special storage conditions or harsh reaction environments make it highly adaptable for large-scale manufacturing of pharmaceutical intermediates.
Q: What is the role of mCPBA in this synthetic pathway?
A: mCPBA (meta-Chloroperoxybenzoic acid) acts as the oxidizing agent in the second stage of the reaction. It converts the initially formed 4,5-dihydroisoxazole intermediate into the aromatic 3,4-disubstituted isoxazole ring system, which is crucial for the biological activity of the final drug molecule.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3,4-Disubstituted Isoxazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of new drug development. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to market supply is seamless. We are committed to delivering 3,4-disubstituted isoxazole compounds with stringent purity specifications, supported by our rigorous QC labs that employ advanced analytical techniques to verify identity and potency. Our capability to implement the efficient, metal-free synthesis described in CN102659705A allows us to offer a superior value proposition, combining technical excellence with commercial viability to support your long-term pharmaceutical projects.
We invite you to engage with our technical procurement team to discuss how our manufacturing capabilities can align with your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how our optimized processes can reduce your overall production costs. We encourage potential partners to contact us for specific COA data and route feasibility assessments, ensuring that you have all the necessary information to make informed sourcing decisions. Let us be your trusted partner in navigating the complexities of pharmaceutical supply chains and delivering the high-performance materials your innovations demand.
