Advancing Pharmaceutical Intermediates: Catalyst-Free Synthesis of 7-Methylpyrazolopyrimidines
The pharmaceutical industry continuously seeks robust and scalable methodologies for constructing nitrogen-containing heterocycles, particularly pyrazolopyrimidine derivatives which serve as critical scaffolds in modern drug discovery. As detailed in patent CN114835713A, a groundbreaking approach has been established for the synthesis of 7-methylpyrazolopyrimidine compounds, addressing long-standing challenges in regioselectivity and operational complexity. These derivatives exhibit profound biological activities, functioning as benzodiazepine receptor modulators, COX-2 inhibitors, and 5-HT6 receptor antagonists, making them indispensable for developing antitumor, antiviral, and anxiolytic agents. The disclosed technology leverages a one-pot tandem reaction strategy that directly couples aromatic aldehydes with 3-aminopyrazoles in the presence of di-tert-butyl peroxide (DTBP). This innovation represents a significant leap forward for any reliable pharmaceutical intermediates supplier aiming to streamline the production of high-value bioactive cores without compromising on purity or yield.
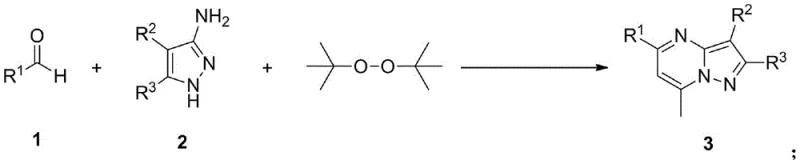
Historically, the construction of the pyrazolopyrimidine core has been fraught with synthetic inefficiencies that hinder large-scale manufacturing and increase overall production costs. Conventional methodologies predominantly rely on the condensation of 3-aminopyrazoles with pre-prepared methyl-containing 1,3-dielectrophilic reagents, a multi-step sequence that necessitates the isolation and purification of unstable intermediates. These traditional routes often suffer from harsh reaction conditions that degrade sensitive functional groups, leading to lower overall throughput and increased waste generation. Furthermore, a critical limitation of prior art is the poor regioselectivity during the ring-closing step, which frequently results in the formation of the undesired 5-methylpyrazolopyrimidine isomer alongside the target 7-methyl product. Separating these structural isomers is notoriously difficult and resource-intensive, requiring extensive chromatographic purification that drastically reduces the final mass balance and economic viability of the process for commercial scale-up of complex heterocyclic compounds.
In stark contrast, the novel approach outlined in the patent data introduces a streamlined, catalyst-free protocol that circumvents the need for pre-functionalized electrophiles entirely. By utilizing simple, commercially available aromatic aldehydes and 3-aminopyrazoles as starting materials, the process achieves direct cyclization through a radical-mediated mechanism initiated by DTBP. This one-pot transformation proceeds under relatively mild thermal conditions, typically between 120-140°C, in 1,2-dichloroethane, eliminating the requirement for exotic catalysts or additives. The operational simplicity is further enhanced by the high regioselectivity inherent to this new pathway, which effectively suppresses the formation of the 5-methyl isomer, thereby ensuring a cleaner reaction profile. For procurement teams, this translates to cost reduction in pharmaceutical intermediates manufacturing by minimizing raw material complexity and reducing the number of unit operations required to reach the final active pharmaceutical ingredient precursor.
Mechanistic Insights into DTBP-Mediated Radical Cyclization
Understanding the mechanistic underpinnings of this transformation is crucial for R&D directors evaluating the robustness and reproducibility of the synthesis for scale-up purposes. The reaction is driven by the thermal decomposition of di-tert-butyl peroxide (DTBP), which generates tert-butoxy radicals that abstract hydrogen atoms from the methyl group of the intermediate species or facilitate the oxidative coupling process. This radical pathway promotes the formation of a key C-C bond between the aldehyde carbon and the pyrazole ring, followed by intramolecular cyclization and subsequent aromatization to yield the stable pyrazolopyrimidine system. The absence of transition metals ensures that the reaction mechanism is not susceptible to catalyst poisoning by trace impurities often found in industrial-grade solvents or reagents, thereby enhancing the reliability of the process across different batches. Moreover, the specific electronic interactions between the substituents on the aromatic aldehyde and the pyrazole nitrogen atoms guide the regiochemical outcome, favoring the thermodynamically stable 7-methyl configuration over the kinetic 5-methyl byproduct.
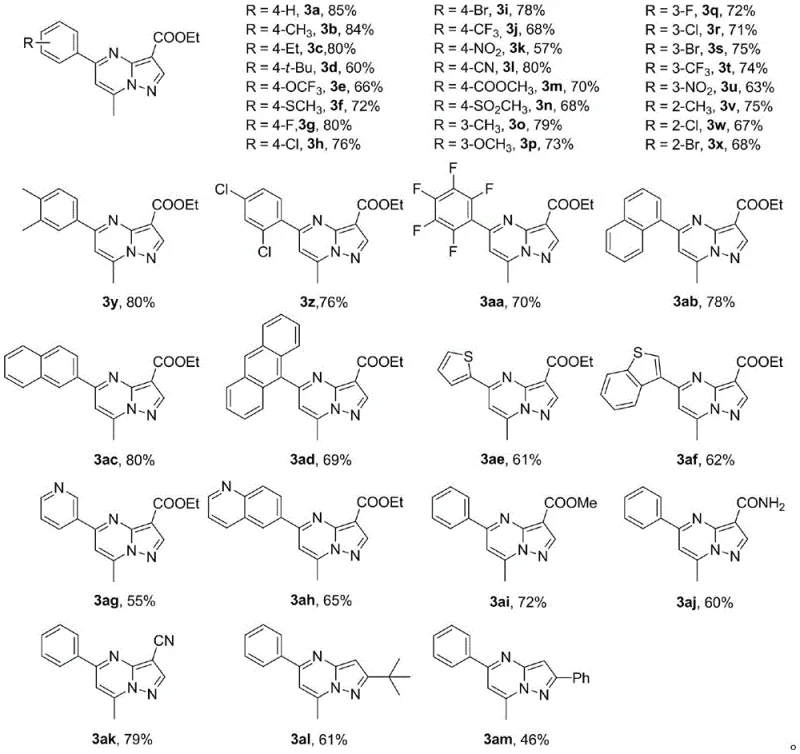
Impurity control is a paramount concern in the synthesis of pharmaceutical intermediates, and this method offers distinct advantages in managing the impurity profile compared to traditional condensation reactions. The high selectivity of the radical-mediated cyclization minimizes the generation of regioisomeric impurities, which are often the most challenging contaminants to remove during crystallization or distillation. Additionally, the use of DTBP as a stoichiometric oxidant results in tert-butanol as a benign byproduct, which is easily removed during the aqueous workup phase, unlike heavy metal salts that require specialized scavenging resins. The broad substrate tolerance demonstrated in the patent data indicates that various electron-withdrawing and electron-donating groups on the aromatic ring do not significantly perturb the reaction efficiency, suggesting a robust mechanism that can accommodate diverse structural modifications. This consistency is vital for maintaining stringent purity specifications required by regulatory bodies, ensuring that the final high-purity 7-methylpyrazolopyrimidines meet the rigorous quality standards of global healthcare markets.
How to Synthesize 7-Methylpyrazolopyrimidine Efficiently
Implementing this synthesis route in a laboratory or pilot plant setting requires adherence to specific operational parameters to maximize yield and safety while maintaining the integrity of the chemical transformation. The process begins with the precise weighing and dissolution of the aromatic aldehyde and 3-aminopyrazole derivatives in 1,2-dichloroethane, followed by the addition of di-tert-butyl peroxide in a controlled manner to manage the exothermic potential of radical initiation. The reaction mixture is then heated in a sealed vessel to maintain the necessary pressure and temperature profile, typically around 130°C, for a duration sufficient to drive the conversion to completion as monitored by TLC or HPLC. Detailed standardized synthesis steps are provided in the guide below to ensure reproducibility and safety compliance during the handling of peroxides and organic solvents.
- Dissolve aromatic aldehyde, 3-aminopyrazole, and di-tert-butyl peroxide (DTBP) in 1,2-dichloroethane solvent with a molar ratio of 1: 1:3.
- Heat the reaction mixture in a sealed tube at a temperature range of 120-140°C for approximately 10 hours to facilitate the cyclization.
- Quench the reaction with water, extract with ethyl acetate, wash with brine, dry over sodium sulfate, and purify via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, the adoption of this metal-free synthesis protocol offers substantial benefits that extend beyond mere chemical efficiency to impact the overall supply chain resilience and cost structure. The elimination of transition metal catalysts removes a significant cost center associated with both the purchase of precious metals and the downstream processing required to reduce metal residues to ppm levels. Furthermore, the reliance on commodity chemicals such as aromatic aldehydes and simple pyrazoles ensures a stable supply base that is less vulnerable to the geopolitical and logistical fluctuations often seen with specialized catalytic reagents. This stability is critical for reducing lead time for high-purity pharmaceutical intermediates, allowing manufacturers to respond more agilely to market demands without being bottlenecked by complex reagent procurement cycles. The simplified workup procedure also reduces solvent consumption and waste disposal costs, aligning with increasingly stringent environmental regulations and corporate sustainability goals.
- Cost Reduction in Manufacturing: The absence of expensive transition metal catalysts fundamentally alters the cost equation by removing the need for costly metal scavengers and extensive purification steps typically required to meet residual metal specifications. This streamlined process significantly lowers the variable cost per kilogram of the produced intermediate, allowing for more competitive pricing structures in the final drug product. Additionally, the one-pot nature of the reaction reduces labor hours and equipment occupancy time, further driving down the overhead allocation for each production batch. These cumulative efficiencies result in substantial cost savings that can be reinvested into further R&D or passed on to improve market competitiveness without compromising margin.
- Enhanced Supply Chain Reliability: By utilizing widely available starting materials like substituted benzaldehydes and aminopyrazoles, the manufacturing process becomes decoupled from the supply constraints often associated with specialized organometallic reagents. This diversification of the raw material base mitigates the risk of production stoppages due to single-source supplier failures or logistics disruptions in the global chemical trade. The robustness of the reaction conditions also means that the process can be transferred between different manufacturing sites with minimal re-validation, ensuring continuity of supply even in the face of regional operational challenges. Such flexibility is invaluable for maintaining consistent inventory levels and meeting just-in-time delivery commitments to downstream pharmaceutical partners.
- Scalability and Environmental Compliance: The reaction operates under solvent conditions and temperatures that are readily manageable in standard stainless steel reactors, facilitating a smooth transition from gram-scale laboratory synthesis to multi-ton commercial production. The generation of benign byproducts like tert-butanol simplifies waste stream management and reduces the environmental footprint associated with hazardous waste disposal. This alignment with green chemistry principles not only lowers compliance costs but also enhances the brand reputation of the manufacturer as a responsible partner in the sustainable development of new medicines. The high atom economy and reduced E-factor of this process make it an ideal candidate for long-term industrial adoption.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this novel synthesis technology for 7-methylpyrazolopyrimidine derivatives. These answers are derived directly from the experimental data and technical specifications outlined in the patent documentation to provide accurate guidance for potential partners. Understanding these details is essential for evaluating the feasibility of integrating this route into existing manufacturing pipelines or new drug development projects. We encourage stakeholders to review these insights to fully appreciate the strategic value this methodology brings to the production of complex heterocyclic intermediates.
Q: How does this method improve regioselectivity compared to conventional routes?
A: Conventional methods often produce unwanted 5-methylpyrazolopyrimidine isomers due to poor control during ring formation. This novel protocol utilizes a specific radical-mediated pathway that inherently favors the 7-methyl configuration, significantly reducing isomeric impurities and simplifying downstream purification.
Q: Are transition metal catalysts required for this synthesis?
A: No, this process is entirely metal-free. It relies on di-tert-butyl peroxide (DTBP) as an oxidant to drive the reaction, eliminating the need for expensive transition metal catalysts and the subsequent costly steps required to remove heavy metal residues from the final product.
Q: What is the substrate scope for this reaction?
A: The method demonstrates broad applicability across various aromatic aldehydes, including those with electron-donating and electron-withdrawing groups, as well as heterocyclic aldehydes. This versatility allows for the efficient production of diverse derivatives suitable for different pharmaceutical applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 7-Methylpyrazolopyrimidine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting innovative synthetic routes that balance scientific rigor with commercial practicality to deliver superior pharmaceutical intermediates. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that promising laboratory technologies like this catalyst-free synthesis are successfully translated into robust industrial processes. We are committed to maintaining stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of 7-methylpyrazolopyrimidine meets the exacting standards required for clinical and commercial applications. Our infrastructure is designed to handle complex chemistries safely and efficiently, providing a secure foundation for your supply chain.
We invite you to collaborate with us to leverage this advanced technology for your specific drug development needs, ensuring a competitive edge in time-to-market and cost efficiency. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your project volume and quality requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our capabilities align with your strategic goals. Let us partner together to bring high-quality, cost-effective therapeutic solutions to patients worldwide.
