Scalable Synthesis of Oxazepine Derivatives for Advanced Hepatitis B Therapeutics
Scalable Synthesis of Oxazepine Derivatives for Advanced Hepatitis B Therapeutics
The global pharmaceutical landscape is constantly evolving to address chronic viral infections, with Hepatitis B remaining a critical public health challenge affecting hundreds of millions worldwide. In response to the urgent need for effective small molecule oral inhibitors capable of reducing HBsAg levels, recent intellectual property developments have introduced robust synthetic methodologies for key therapeutic scaffolds. Specifically, patent CN112390816B discloses a novel preparation method for oxazepine compounds of Formula (I), which serve as potent surface antigen inhibitors. This technology represents a significant leap forward in medicinal chemistry, addressing the limitations of previous nucleotide drugs that failed to directly reduce HBsAg levels effectively. By focusing on the structural integrity and scalable production of these complex heterocycles, the disclosed process offers a viable pathway for developing next-generation antiviral agents that target the functional cure of chronic Hepatitis B through the elimination of surface antigens.
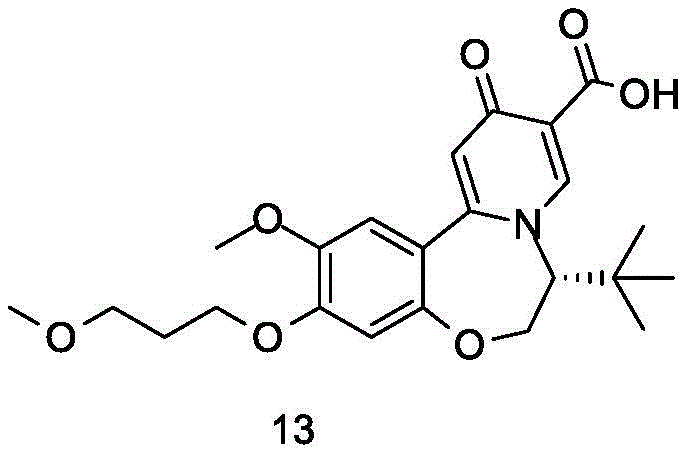
The development of reliable oxazepine intermediate suppliers is contingent upon mastering the intricate balance between chemical complexity and manufacturing feasibility. The compounds described in this patent feature a fused ring system incorporating nitrogen and oxygen atoms, substituted with various alkyl and alkoxy groups to optimize biological activity. For R&D directors evaluating potential candidates, the purity profile and impurity spectrum of these intermediates are paramount. The patent details a comprehensive approach to constructing the oxazepine core, ensuring that stereocenters are controlled and functional groups are protected appropriately throughout the synthesis. This level of control is essential for meeting the stringent regulatory requirements of the pharmaceutical industry, where even trace impurities can derail clinical trials. Consequently, understanding the mechanistic underpinnings of this synthesis is crucial for any organization aiming to secure a stable supply chain for these high-value antiviral precursors.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art methods, such as those disclosed in patent application WO2018214875, relied heavily on specific reagents like Compound B-1 to effect the critical ring-closing step. However, industrial experience has shown that the synthesis condition of Compound B-1 is exceptionally harsh, leading to significant difficulties in purification and quality control. The instability of this reagent often results in poor reaction repeatability and notoriously low yields, reported to be only about 7% in the pharmaceutical synthesis process. Such inefficiencies are unacceptable in a commercial setting, as they lead to excessive waste, high production costs, and unpredictable supply timelines. Furthermore, the inability to control the purity of Compound B-1 propagates errors into subsequent steps, compromising the overall quality of the final active pharmaceutical ingredient. These technical bottlenecks highlight the critical need for a more robust and economical synthetic strategy that can withstand the rigors of large-scale manufacturing.
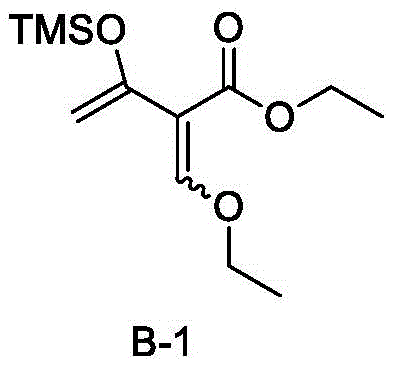
The Novel Approach
The innovative methodology presented in CN112390816B circumvents these historical challenges by introducing a superior reagent, designated as Compound B (specifically Compound 6 in the examples), which is both economical and environmentally friendly. Unlike its predecessor, this new reagent is easy to prepare and exhibits remarkable stability, allowing for controllable quality throughout the production batch. The impact on process efficiency is profound, with the yield of the critical coupling reaction improving drastically from single digits to approximately 80%. This substantial increase in yield not only enhances the economic viability of the process but also ensures reaction stability, making it suitable for large-scale production. By replacing the problematic reagent with a more accessible alternative, the new route simplifies the workflow and reduces the burden on downstream purification units, thereby streamlining the entire manufacturing campaign for cost reduction in API manufacturing.
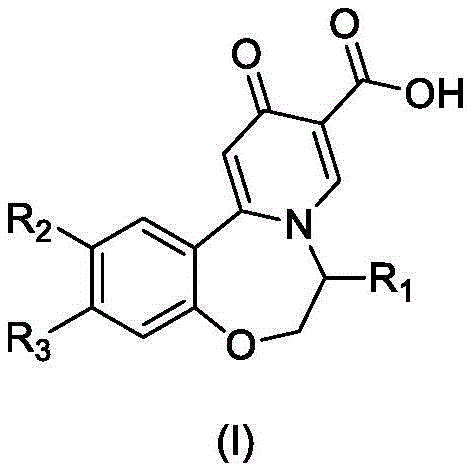
Mechanistic Insights into Oxazepine Ring Formation and Oxidation
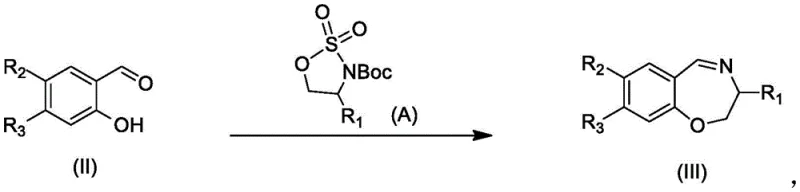
The core of this synthetic achievement lies in the strategic construction of the seven-membered oxazepine ring via a condensation reaction between an aldehyde intermediate (Compound II) and a chiral amine derivative (Compound A). This step forms the imine intermediate (Compound III), which is subsequently subjected to a cyclization reaction with the beta-keto ester derivative (Compound B). The choice of reagents here is critical; the use of mild acidic conditions facilitates the formation of the tetrahydro-1H-pyrano[3,2-c]pyridine skeleton without degrading sensitive functional groups. Following the ring closure, the dihydro-pyridone intermediate (Compound IV) undergoes an oxidation step to establish the fully aromatic pyridone system found in the final Formula (I). This oxidation is typically achieved using iodine and dimethyl sulfoxide, a combination that provides the necessary driving force for aromatization while maintaining compatibility with the diverse substituents present on the benzene ring. Understanding this mechanistic sequence allows chemists to fine-tune reaction parameters for optimal conversion and minimal byproduct formation.
Furthermore, the synthesis of the chiral amine precursor (Compound A) demonstrates a keen awareness of safety and scalability in reagent selection. The process employs sodium borohydride for the reduction of carboxylic acids to alcohols, a distinct improvement over the traditional use of lithium aluminum hydride. While lithium aluminum hydride is a powerful reducing agent, its active chemical nature poses severe safety hazards, including the risk of spontaneous ignition upon contact with water or moist air. In contrast, sodium borohydride is significantly cheaper, safer to handle, and can be added at controlled temperatures around 0°C. This substitution eliminates the need for specialized anhydrous conditions required by pyrophoric reagents, thereby reducing energy consumption and enhancing operational safety. The resulting active ester intermediate is stable, ensuring that the reduction proceeds smoothly to generate the required hydroxyl group for subsequent cyclization, thus enabling the stable and safe production of Compound A-3 on a large scale.
How to Synthesize Oxazepine Compound 13 Efficiently
The practical implementation of this technology is best illustrated through the detailed preparation of Compound 13, a specific embodiment of the general Formula (I). This multi-step sequence begins with the protection of amino acids and proceeds through alkylation, condensation, and final oxidation. Each step has been optimized for high yield and purity, utilizing common solvents like ethanol, ethyl acetate, and dichloromethane to facilitate easy workup and solvent recovery. The process demonstrates how complex heterocyclic systems can be assembled from readily available starting materials through a logical progression of functional group transformations. For technical teams looking to replicate or adapt this chemistry, the key lies in strict temperature control during the exothermic reduction and oxidation phases, as well as efficient purification strategies such as crystallization and slurry washing. The detailed standardized synthesis steps for producing this high-purity oxazepine intermediate are outlined below.
- Prepare intermediate Compound 4 via Boc-protection, reduction with sodium borohydride, and oxidative cyclization.
- Synthesize Compound 6 by hydrolyzing the enol ether precursor in water under mild heating conditions.
- Condense Compound 11 with Compound 6 using glacial acetic acid, followed by iodine-mediated oxidation to yield Compound 13.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition from the legacy synthesis route to this novel methodology offers tangible benefits that extend beyond mere chemical yield. The primary advantage lies in the drastic simplification of the raw material portfolio. By eliminating the need for hazardous, hard-to-source reagents like lithium aluminum hydride and the unstable Compound B-1, the supply chain becomes more resilient and less prone to disruptions caused by regulatory restrictions on dangerous goods. The reliance on commodity chemicals such as sodium borohydride and simple beta-keto esters ensures a steady flow of materials from multiple vendors, reducing dependency on single-source suppliers. This diversification of the supply base is critical for maintaining continuity in the production of life-saving antiviral medications, especially in a global market where logistics can be unpredictable. Moreover, the improved process robustness translates directly into more reliable delivery schedules for downstream API manufacturers.
- Cost Reduction in Manufacturing: The economic impact of this new process is driven by the elimination of expensive and dangerous reagents, which lowers the direct material cost per kilogram of product. The switch to sodium borohydride not only reduces the purchase price of the reducing agent but also minimizes the costs associated with special handling, storage, and waste disposal of pyrophoric materials. Additionally, the significant improvement in reaction yield means that less starting material is wasted, maximizing the output from each batch and reducing the overall cost of goods sold. The simplified purification steps further contribute to cost savings by reducing solvent consumption and processing time, making the entire manufacturing operation leaner and more profitable without compromising on quality standards.
- Enhanced Supply Chain Reliability: The stability of the intermediates generated in this route, particularly Compound 6 and Compound 4, allows for the potential creation of strategic stockpiles, buffering the production line against upstream fluctuations. Since the reagents are non-hazardous and stable at ambient conditions, transportation and storage logistics are significantly streamlined, removing the need for specialized cold chain or hazard-class shipping. This ease of handling ensures that raw materials can be sourced globally with fewer regulatory hurdles, shortening lead times for high-purity oxazepine intermediates. The reproducibility of the reaction, evidenced by the consistent 80% yield in the key coupling step, guarantees that production targets can be met reliably, fostering trust between the chemical supplier and the pharmaceutical client.
- Scalability and Environmental Compliance: From an environmental and safety perspective, this process aligns perfectly with modern green chemistry principles and strict industrial safety regulations. The avoidance of pyrophoric reagents reduces the risk of workplace accidents, lowering insurance premiums and liability exposure for the manufacturing facility. The use of aqueous workups and standard organic solvents facilitates efficient waste treatment and solvent recycling, minimizing the environmental footprint of the production process. As regulatory bodies increasingly scrutinize the environmental impact of pharmaceutical manufacturing, adopting a route that generates less hazardous waste and consumes less energy provides a competitive advantage. This compliance readiness ensures that the commercial scale-up of complex oxazepine derivatives can proceed without regulatory delays, securing long-term market access.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of these oxazepine compounds. They are derived from the specific experimental data and process descriptions found in the patent literature, providing clarity on the feasibility and advantages of this synthetic route. Understanding these details helps stakeholders make informed decisions about integrating this technology into their existing manufacturing portfolios. The answers reflect the consensus on safety, efficiency, and scalability as demonstrated in the provided examples.
Q: Why is sodium borohydride preferred over lithium aluminum hydride in this process?
A: Sodium borohydride is significantly safer and cheaper than lithium aluminum hydride. It avoids the risk of spontaneous combustion upon contact with moisture, making it ideal for large-scale industrial production while maintaining high reduction efficiency.
Q: What is the primary advantage of the new cyclization reagent compared to prior art?
A: The new reagent (Compound B/6) eliminates the harsh conditions and poor repeatability associated with the prior art reagent B-1. It increases the reaction yield from approximately 7% to around 80%, ensuring consistent quality and supply stability.
Q: Is this synthesis route suitable for commercial scale-up?
A: Yes, the process utilizes stable intermediates and mild reaction conditions, such as aqueous workups and standard solvents like ethanol and ethyl acetate. The avoidance of pyrophoric reagents facilitates safe operation in 50L reactors and beyond.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Oxazepine Compound Supplier
The technical breakthroughs detailed in patent CN112390816B underscore the immense potential of these oxazepine derivatives in the fight against chronic Hepatitis B. At NINGBO INNO PHARMCHEM, we recognize the complexity involved in translating such sophisticated laboratory chemistry into commercial reality. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project moves seamlessly from gram-scale optimization to full-scale manufacturing. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of intermediate meets the exacting standards required for clinical and commercial API synthesis. We are committed to being a reliable oxazepine compound supplier who understands the critical nature of antiviral drug development.
We invite pharmaceutical partners to collaborate with us to leverage this advanced synthetic technology for your pipeline. Our team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how our optimized process can reduce your overall production expenses. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments for your project. By partnering with us, you gain access to a secure, scalable, and cost-effective supply chain solution that supports your mission to deliver life-saving therapies to patients worldwide.
