Advanced Synthesis of Donepezil Hydrochloride: A Breakthrough in Selectivity and Yield for Commercial API Manufacturing
Introduction to Novel Donepezil Hydrochloride Synthesis
The pharmaceutical landscape for neurological agents demands increasingly efficient and pure synthetic routes, particularly for high-volume drugs like Donepezil Hydrochloride, a pivotal acetylcholinesterase inhibitor used in treating Alzheimer's disease. Patent CN111100062A introduces a transformative methodology that addresses long-standing challenges in selectivity and yield associated with conventional manufacturing processes. This innovation leverages a strategic combination of condensation, catalytic transfer hydrogenation, and selective oxidation to deliver a product with exceptional purity profiles suitable for stringent regulatory environments. By shifting away from high-pressure hydrogenation and complex protection-deprotection sequences, this route offers a streamlined pathway that aligns perfectly with modern green chemistry principles and cost-effective production goals. For global procurement teams and R&D directors, understanding the nuances of this patent is critical for securing a reliable pharmaceutical intermediates supplier capable of meeting future demand without compromising on quality or delivery timelines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Donepezil Hydrochloride has been plagued by significant inefficiencies that hinder large-scale commercial viability. Traditional routes, such as those disclosed in earlier patents like EP0711756A1, often rely on direct catalytic hydrogenation of pyridine-containing intermediates. As illustrated in the reaction schemes of prior art, these methods frequently encounter severe selectivity problems during the hydrogenation step, where the reduction of the carbon-carbon double bond is accompanied by unwanted side reactions. This lack of selectivity results in crude products containing substantial impurity levels, often necessitating multiple, yield-eroding purification steps to achieve pharmaceutical grade standards. Furthermore, the reliance on high-pressure hydrogen gas introduces safety hazards and requires specialized, capital-intensive equipment, creating bottlenecks in the supply chain for many manufacturers. The cumulative effect of these drawbacks is a total process yield that can fall drastically, sometimes remaining below 20% in older methodologies, rendering them economically unfeasible for competitive markets.
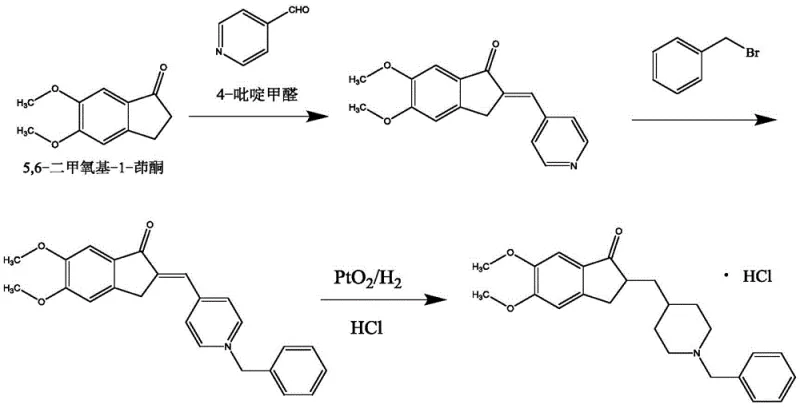
The Novel Approach
In stark contrast, the methodology outlined in CN111100062A presents a robust solution by decoupling the reduction and oxidation steps through a clever transfer hydrogenation strategy. Instead of subjecting the sensitive enone intermediate to harsh direct hydrogenation conditions, the process utilizes formic acid as a hydrogen source in the presence of a palladium catalyst. This approach not only mitigates the safety risks associated with high-pressure hydrogen but also provides superior control over the reduction trajectory. The subsequent use of Jones reagent allows for the precise restoration of the ketone functionality, effectively correcting any over-reduction that might occur during the hydrogen transfer phase. This two-step redox maneuver ensures that the final ketone product is obtained with minimal structural degradation or isomeric impurities. By eliminating the need for complex protecting groups and simplifying the workup procedures to basic filtration and crystallization, this novel approach drastically reduces the operational complexity and environmental footprint of the synthesis.
Mechanistic Insights into Transfer Hydrogenation and Selective Oxidation
The core chemical innovation lies in the mechanistic pathway of the catalytic transfer hydrogenation followed by Jones oxidation. In the first stage, the enone intermediate (Compound III) undergoes reduction where formic acid serves as the hydride donor. The palladium on carbon catalyst facilitates the transfer of hydrogen atoms to the carbon-carbon double bond and potentially the carbonyl group, forming a saturated alcohol intermediate (Compound IV). This step is crucial because formic acid acts as a milder reducing agent compared to molecular hydrogen, allowing the reaction to proceed at moderate temperatures between 40°C and 60°C. The mechanism avoids the aggressive conditions that typically lead to the cleavage of benzyl groups or the formation of fully reduced cycloalkane byproducts. Following this, the introduction of the Jones reagent (chromium trioxide in sulfuric acid) selectively targets the secondary alcohol generated in the previous step. The chromium(VI) species efficiently oxidizes the alcohol back to the desired ketone without affecting the methoxy groups on the indanone ring or the benzyl moiety on the piperidine ring. This precise chemoselectivity is the key to achieving the reported purity levels exceeding 99%.
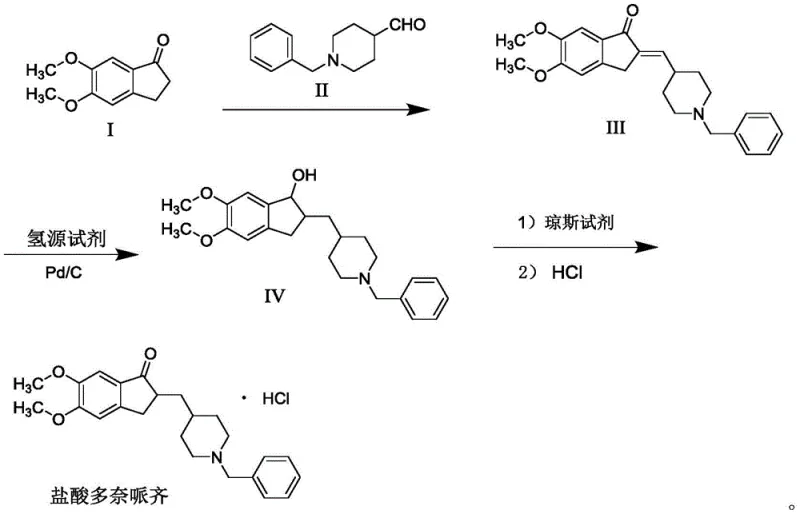
Furthermore, the impurity control mechanism is inherently built into this redox cycle. In conventional direct hydrogenation, partial reduction or isomerization can create difficult-to-remove impurities that co-elute with the product. However, in this novel route, any alcohol formed is quantitatively converted back to the ketone, effectively 'cleaning up' the intermediate stream. The use of a mixed catalyst system in the initial condensation step—comprising a base like potassium carbonate or calcium chloride alongside a phase transfer catalyst like potassium iodide—further enhances purity by driving the aldol-type condensation to completion while minimizing side reactions. The solid nature of these catalysts allows for their complete removal via simple filtration before the concentration step, preventing metal contamination in the final API. This rigorous control over the reaction environment ensures that the impurity profile remains exceptionally low, reducing the burden on downstream purification units and increasing the overall throughput of the manufacturing facility.
How to Synthesize Donepezil Hydrochloride Efficiently
The practical execution of this synthesis involves a straightforward three-stage protocol that is highly amenable to scale-up in standard reactor setups. The process begins with the condensation of 5,6-dimethoxy-1-indanone and 1-benzylpiperidine-4-formaldehyde in a toluene-water biphasic system, promoted by the mixed catalyst system to yield the enone intermediate. Following isolation, the enone is subjected to transfer hydrogenation using formic acid and Pd/C in an ethanol-acetone mixture, a step that requires careful temperature monitoring to ensure complete reduction without degradation. Finally, the resulting alcohol is oxidized using freshly prepared Jones reagent, followed by pH adjustment with hydrochloric acid to precipitate the final hydrochloride salt. This sequence eliminates the need for cryogenic conditions or exotic reagents, making it accessible for a wide range of chemical manufacturers. For detailed operational parameters and safety guidelines, please refer to the standardized synthesis steps provided below.
- Condense 5,6-dimethoxy-1-indanone with 1-benzylpiperidine-4-formaldehyde using a mixed catalyst system in toluene to form the enone intermediate.
- Perform catalytic transfer hydrogenation on the enone intermediate using formic acid and Pd/C to reduce the double bond and form the alcohol intermediate.
- Oxidize the alcohol intermediate using Jones reagent to restore the ketone functionality, followed by salt formation with hydrochloric acid.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this synthesis route offers profound advantages that directly address the pain points of procurement managers and supply chain heads in the pharmaceutical industry. The shift from high-pressure hydrogenation to atmospheric transfer hydrogenation significantly lowers the barrier to entry for manufacturing, as it removes the requirement for specialized high-pressure autoclaves and the associated safety infrastructure. This reduction in capital expenditure translates directly into lower fixed costs per kilogram of product, allowing for more competitive pricing structures in the global market. Additionally, the simplified workup procedures, which rely on filtration and crystallization rather than complex chromatographic separations, drastically reduce solvent consumption and waste generation. This efficiency gain not only lowers the cost of goods sold but also aligns with increasingly strict environmental regulations, ensuring long-term supply continuity without the risk of regulatory shutdowns due to compliance issues.
- Cost Reduction in Manufacturing: The elimination of expensive high-pressure equipment and the use of readily available reagents like formic acid and common solvents leads to substantial operational savings. The high yield of the process means that less raw material is wasted per unit of output, further driving down the variable costs associated with production. Moreover, the ability to recover and reuse catalysts or the simplicity of their disposal reduces the financial burden of waste management. These factors combine to create a highly cost-efficient manufacturing model that can withstand market fluctuations in raw material pricing.
- Enhanced Supply Chain Reliability: The mild reaction conditions and the use of stable, non-hazardous reagents enhance the robustness of the supply chain. Unlike processes requiring cryogenic temperatures or unstable intermediates, this route can be executed reliably in standard facilities with minimal risk of batch failure due to equipment malfunction. The high purity of the crude product reduces the dependency on third-party purification services, shortening the overall lead time from synthesis to final release. This reliability is crucial for maintaining consistent inventory levels and meeting the just-in-time delivery requirements of major pharmaceutical clients.
- Scalability and Environmental Compliance: The process is inherently scalable, as demonstrated by the successful transition from gram-scale experiments to multi-kilogram batches without loss of efficiency. The reduction in solvent usage and the generation of less hazardous waste streams simplify the environmental permitting process and reduce the ecological footprint of the manufacturing site. This scalability ensures that suppliers can rapidly ramp up production to meet surges in demand, such as those driven by new drug approvals or generic market expansions, without compromising on quality or sustainability standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis method. These insights are derived directly from the patent data and are intended to clarify the feasibility and benefits of adopting this route for commercial production. Understanding these details is essential for stakeholders evaluating the potential for technology transfer or long-term supply partnerships.
Q: Why is transfer hydrogenation preferred over direct hydrogenation for Donepezil synthesis?
A: Direct hydrogenation often suffers from selectivity issues, leading to over-reduction or impurity formation. Transfer hydrogenation using formic acid offers milder conditions and better control, which is subsequently corrected by selective Jones oxidation to ensure high purity.
Q: What are the key advantages of the mixed catalyst system in the condensation step?
A: The use of a mixed catalyst (e.g., potassium carbonate/calcium chloride with potassium iodide) enhances the reaction rate and allows for easy removal of solid catalysts by simple filtration, significantly simplifying post-treatment and reducing waste.
Q: How does this method impact the overall yield compared to traditional routes?
A: Traditional routes often report yields below 20% or around 70-75% with significant impurities. This novel method achieves yields exceeding 90% with purity greater than 99%, making it highly suitable for industrial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Donepezil Hydrochloride Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic routes to maintain competitiveness in the global pharmaceutical market. Our team of expert chemists has extensively evaluated the methodology described in CN111100062A and possesses the technical capability to implement this high-yield, high-purity process at an industrial scale. We boast extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent and reliable supply of Donepezil Hydrochloride intermediates. Our state-of-the-art facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch meets the highest international standards for safety and efficacy.
We invite procurement leaders and R&D directors to collaborate with us to leverage this cutting-edge technology for your supply chain. By partnering with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and logistical needs. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us demonstrate how our commitment to innovation can drive value and efficiency in your Donepezil Hydrochloride sourcing strategy.
