Advanced Synthesis of 9-Fluorene-Anthracene Compounds for High-Performance Blue OLED Displays
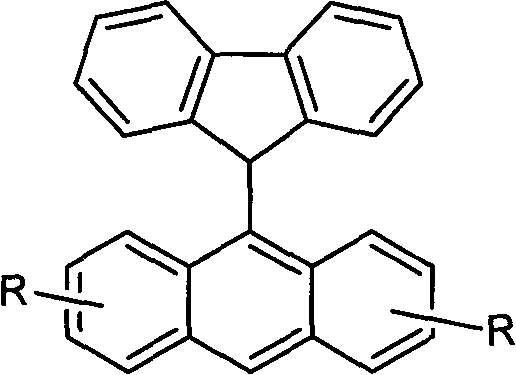
The rapid evolution of flat-panel display technology has placed unprecedented demands on the performance and stability of organic electroluminescent materials, particularly in the realm of blue light emission. As detailed in patent CN101817719B, a significant breakthrough has been achieved in the development of 9-fluorene-anthracene compounds, which serve as critical blue light emitting materials for next-generation OLED devices. This patent outlines a robust synthetic methodology that connects the 9-position of a fluorene ring directly to the 9-position of an anthracene ring, creating a conjugated system with superior thermal stability and luminous efficiency. Unlike traditional approaches that often struggle with poor stability or low brightness in the blue spectrum, this novel architecture leverages the inherent rigidity of the fluorene unit combined with the high quantum efficiency of anthracene. The technical disclosure provides a comprehensive pathway for producing these high-value electronic chemicals, addressing the long-standing industry challenge of finding blue emitters that do not degrade rapidly under operational stress. For R&D directors and procurement specialists alike, this represents a pivotal shift towards more accessible and chemically stable precursors for advanced display manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of complex polycyclic aromatic hydrocarbons for OLED applications has relied heavily on transition-metal catalyzed cross-coupling reactions, such as Suzuki or Heck couplings, which necessitate the use of expensive palladium catalysts and strictly anhydrous conditions. These conventional pathways often suffer from significant drawbacks, including the high cost of noble metal catalysts, the generation of toxic heavy metal waste that requires rigorous and costly purification protocols, and sensitivity to oxygen and moisture which complicates reactor operations. Furthermore, achieving high regioselectivity at the 9-position of anthracene can be challenging without protecting groups, leading to lower overall yields and difficult separation processes that inflate the final cost of the electronic chemical. The reliance on sophisticated catalytic systems also introduces supply chain vulnerabilities, as the availability and price volatility of palladium can directly impact the manufacturing economics of the final luminescent material.
The Novel Approach
In stark contrast, the methodology described in CN101817719B introduces a streamlined metal-mediated coupling strategy that bypasses the need for precious metal catalysts entirely. By utilizing abundant and inexpensive metal powders such as iron, aluminum, or zinc in a carbon disulfide solvent system, the process achieves direct coupling between 9-bromofluorene and anthracene derivatives under simple reflux conditions. This approach not only drastically simplifies the reaction setup by eliminating the need for inert atmosphere gloveboxes typically required for sensitive organometallic chemistry but also facilitates easier workup procedures where the metal residues can be removed via simple filtration. The ability to operate at reflux temperatures for 12 to 24 hours ensures complete conversion while maintaining a operational simplicity that is highly attractive for industrial scale-up. This shift from homogeneous noble metal catalysis to heterogeneous base metal mediation represents a fundamental optimization in the manufacturing logic of OLED intermediates.
Mechanistic Insights into Metal-Mediated Coupling at the 9-Position
The core chemical transformation involves the activation of the carbon-bromine bond at the 9-position of the fluorene ring by the surface of the metal powder, likely proceeding through a radical or organometallic intermediate mechanism that facilitates nucleophilic attack on the anthracene core. The reaction conditions promote the formation of a new carbon-carbon bond specifically at the 9-position of the anthracene ring, which is the most reactive site due to its high electron density and steric accessibility. This regioselectivity is crucial for maintaining the planarity and conjugation length of the final molecule, which directly correlates with its electronic properties and blue emission wavelength. The use of carbon disulfide as a solvent is particularly strategic, as it effectively dissolves the aromatic reactants while remaining stable under the reflux conditions required to activate the metal surface. Understanding this mechanism allows process chemists to fine-tune the particle size of the metal powder and the agitation speed to maximize the surface area available for the reaction, thereby optimizing the reaction kinetics without altering the fundamental reagent stoichiometry.
Furthermore, the patent highlights the structural modification of the anthracene ring through the introduction of tert-butyl groups, which plays a vital role in the physical properties of the final material. These bulky alkyl groups act as steric spacers that prevent the planar aromatic cores from stacking too closely together, a phenomenon known as pi-pi stacking that often leads to fluorescence quenching in the solid state. By suppressing these intermolecular forces, the tert-butyl substituents significantly enhance the solubility of the 9-fluorene-anthracene compound in common organic processing solvents, facilitating solution-based deposition techniques like spin-coating or inkjet printing. This structural engineering ensures that the material not only emits efficient blue light with a peak wavelength around 415 to 435 nanometers but also possesses the thermal stability and morphological integrity required for the long operational lifetimes demanded by commercial display panels.
How to Synthesize 9-Fluorene-Anthracene Efficiently
The synthesis protocol outlined in the patent provides a clear and reproducible framework for generating these high-purity OLED intermediates, starting with the precise weighing of 9-bromofluorene and the chosen anthracene derivative. The process begins by dissolving these key starting materials in carbon disulfide within a round-bottom flask equipped with a reflux condenser, ensuring that the solvent vapors are condensed and returned to the reaction vessel to maintain a constant volume and concentration. Following the addition of the metal powder catalyst, the mixture is subjected to vigorous stirring under reflux for a period ranging from 12 to 24 hours, allowing sufficient time for the coupling reaction to reach completion. Detailed standardized operating procedures for temperature control, filtration techniques, and chromatographic purification are essential to ensure batch-to-batch consistency and high purity levels required for electronic applications.
- Dissolve 9-bromofluorene and anthracene derivatives in carbon disulfide solvent within a reflux apparatus.
- Add metal powder catalyst such as iron, aluminum, or zinc and maintain reflux stirring for 12 to 24 hours.
- Cool the reaction mixture, filter off metal residues, concentrate the filtrate, and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthesis route offers profound benefits for procurement managers and supply chain directors seeking to optimize the cost structure of OLED material manufacturing. The elimination of expensive palladium catalysts and the associated ligands removes a significant cost driver from the bill of materials, while simultaneously simplifying the supply chain by relying on commodity chemicals like iron powder and carbon disulfide that are globally available in bulk quantities. This reduction in dependency on precious metals mitigates the risk of price volatility and supply disruptions, ensuring a more predictable and stable cost of goods sold over the long term. Additionally, the simplified purification process, which avoids complex heavy metal scavenging steps, reduces the consumption of specialized silica gels and solvents, further contributing to substantial cost savings in the downstream processing stages.
- Cost Reduction in Manufacturing: The substitution of noble metal catalysts with base metal powders such as iron or zinc results in a drastic reduction in raw material expenses, as these metals are orders of magnitude cheaper than palladium or platinum equivalents. Moreover, the heterogeneous nature of the catalyst allows for easy removal via filtration, eliminating the need for costly and time-consuming chromatographic steps specifically designed to reduce metal residuals to ppm levels. This streamlined workflow reduces labor hours and utility consumption per kilogram of product, driving down the overall manufacturing overhead. The high atom economy of the direct coupling reaction also minimizes waste generation, aligning with green chemistry principles that can lead to lower waste disposal fees.
- Enhanced Supply Chain Reliability: The raw materials required for this synthesis, including 9-bromofluorene, anthracene, and zinc powder, are commodity chemicals produced by numerous suppliers worldwide, ensuring a robust and diversified supply base. This abundance prevents single-source bottlenecks and allows procurement teams to negotiate favorable terms based on market competition rather than scarcity. The stability of the intermediates and the final product also simplifies logistics, as the materials do not require cryogenic shipping or extreme storage conditions, reducing the risk of degradation during transit. Consequently, lead times for high-purity electronic chemicals can be significantly shortened, enabling faster response to market demands for new display technologies.
- Scalability and Environmental Compliance: The reaction conditions described, involving simple reflux in a standard solvent, are inherently scalable from gram-scale laboratory experiments to multi-ton industrial reactors without requiring specialized high-pressure or cryogenic equipment. This ease of scale-up accelerates the time-to-market for new OLED formulations, allowing manufacturers to ramp up production quickly to meet consumer electronics cycles. Furthermore, the use of recyclable metal powders and the potential for solvent recovery systems enhances the environmental profile of the process, helping companies meet increasingly stringent regulatory standards for volatile organic compound emissions and hazardous waste generation.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of 9-fluorene-anthracene derivatives, based on the specific embodiments and data provided in the patent literature. These insights are intended to clarify the feasibility of implementing this technology within existing manufacturing frameworks and to highlight the specific performance metrics achieved during development. Understanding these details is critical for technical teams evaluating the integration of this material into their device architectures.
Q: What are the primary advantages of this synthesis method over traditional palladium-catalyzed routes?
A: This method utilizes inexpensive metal powders like iron or zinc instead of costly palladium catalysts, significantly reducing raw material costs and eliminating the need for complex heavy metal removal steps.
Q: How does the introduction of tert-butyl groups affect the material properties?
A: The addition of tert-butyl groups at the 2 and 6 positions of the anthracene ring suppresses intermolecular forces, thereby improving the solubility of the material in organic solvents and enhancing processability for device fabrication.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process involves simple reflux conditions and readily available raw materials, making it highly amenable to scale-up from laboratory grams to multi-ton commercial production without complex equipment requirements.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 9-Fluorene-Anthracene Supplier
As the demand for high-efficiency blue OLED materials continues to surge, partnering with an experienced chemical manufacturer is essential for securing a competitive advantage in the display market. NINGBO INNO PHARMCHEM leverages extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production to deliver consistent quality and supply continuity for complex electronic intermediates. Our state-of-the-art facilities are equipped with rigorous QC labs and advanced analytical instrumentation to ensure that every batch meets stringent purity specifications, free from the metal contaminants that can degrade device performance. We understand the critical nature of supply chain resilience and are committed to providing a stable source of high-performance luminescent materials that empower your innovation.
We invite you to collaborate with our technical team to explore how this optimized synthesis route can be tailored to your specific production needs. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits of switching to this metal-mediated coupling process. Please contact our technical procurement team today to obtain specific COA data and route feasibility assessments, and let us support your journey toward next-generation display excellence with reliable, high-quality chemical solutions.
