Revolutionizing Pharmaceutical Intermediate Production: Ruthenium-Catalyzed C-H Activation for Scalable 6-Alkenyl Phenanthridine Synthesis
Patent CN111732541B introduces a novel ruthenium-catalyzed C-H activation/cyclization methodology for the efficient synthesis of 6-alkenyl phenanthridine derivatives, representing a significant advancement in heterocyclic compound manufacturing for pharmaceutical applications. This breakthrough addresses critical limitations in traditional phenanthridine ring synthesis while delivering substantial operational and environmental benefits for B2B chemical manufacturing.
Overcoming Traditional Limitations in Phenanthridine Synthesis
The Limitations of Conventional Methods
Traditional phenanthridine synthesis primarily relies on the Bischler-Napieralski reaction, which requires high temperatures and hazardous reagents like PCl3 and POCl3, creating significant operational challenges for pharmaceutical manufacturers. These harsh conditions result in complex reaction setups with low functional group tolerance, limiting substrate versatility and increasing purification complexity. The process generates multiple hazardous byproducts requiring extensive waste treatment, while the high energy consumption and specialized equipment needs substantially elevate production costs. Furthermore, the safety risks associated with handling corrosive reagents at elevated temperatures necessitate costly containment systems and specialized operator training, making scale-up particularly challenging for commercial API intermediate production.
The Novel Approach
The patented methodology employs ruthenium-catalyzed C-H activation using readily available 2-benzidine compounds and cyclopropenone coupling reagents under mild conditions (100°C, 48 h) with simple workup procedures. This innovative approach eliminates pre-activation steps while maintaining exceptional substrate scope across diverse functional groups including halogens, alkyl, alkoxy, carbonyl, nitro, and cyano substituents. The reaction achieves high yields ranging from 64% to 82% across multiple implementations without requiring inert atmosphere, demonstrating remarkable robustness for industrial application. Crucially, water is the sole byproduct, significantly improving atom economy while eliminating hazardous waste streams that plague conventional methods. The process utilizes cost-effective ruthenium catalysts instead of expensive rhodium alternatives, with catalyst loadings as low as 0.02 mol% enabling economically viable commercial production.
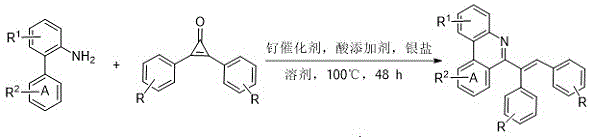
Advanced Reaction Mechanism and Purity Control
The reaction mechanism involves ruthenium(II) catalysts such as p-cymene ruthenium(II) dichloride dimer facilitating C-H bond cleavage at the ortho position of the benzidine substrate, followed by cyclopropenone insertion and reductive elimination to form the phenanthridine core. This direct C-H functionalization pathway avoids traditional pre-halogenation steps that introduce halogen impurities requiring additional purification stages. The use of acid additives (boric acid, 1-adamantanecarboxylic acid, or pivalic acid) and silver salts (silver carbonate or silver hexafluoroantimonate) creates a synergistic catalytic system that maintains high regioselectivity while suppressing common side reactions like homocoupling or over-oxidation. The mild reaction temperature (100°C) prevents thermal degradation of sensitive functional groups, preserving molecular integrity throughout the transformation.
Impurity profile management is significantly enhanced through this methodology, as evidenced by NMR and HRMS characterization showing clean product formation with minimal side products. The absence of transition metal contaminants is ensured by using ruthenium catalysts instead of rhodium systems that require extensive metal removal procedures. The simple workup process involving only solvent removal and silica gel chromatography eliminates complex extraction steps that could introduce organic solvent residues. The reaction's high atom economy (only water byproduct) inherently reduces potential impurities compared to traditional methods generating multiple waste streams. This streamlined purification approach consistently delivers products meeting stringent pharmaceutical purity requirements without additional costly polishing steps, as demonstrated by HRMS data showing exact mass matches within ±0.0005 Da across all implementation examples.
Commercial Advantages for Pharmaceutical Supply Chains
This innovative synthesis methodology directly addresses critical pain points in pharmaceutical intermediate manufacturing by transforming complex multi-step processes into efficient single-step operations. The elimination of hazardous reagents and high-temperature requirements reduces both capital expenditure for specialized equipment and operational costs associated with safety protocols. The broad substrate scope enables rapid adaptation to diverse customer requirements without process revalidation, while the simplified workup procedure significantly shortens production cycles compared to conventional approaches requiring multiple purification stages.
- Reduced Equipment Depreciation: The mild reaction conditions (100°C) eliminate the need for specialized high-pressure reactors required by traditional methods operating at extreme temperatures with corrosive reagents like PCl3. This extends equipment lifespan while reducing maintenance costs associated with corrosion damage from halogenated compounds. The compatibility with standard glassware and stainless steel reactors allows pharmaceutical manufacturers to utilize existing infrastructure without costly capital investments in specialized containment systems. Furthermore, the absence of explosive or highly reactive intermediates minimizes wear on process equipment, significantly lowering total cost of ownership for production facilities.
- Shorter Lead Times: The one-step reaction process with simple workup (solvent removal followed by silica gel chromatography) reduces production cycle time by eliminating multiple intermediate isolation and purification steps required in conventional syntheses. The ambient atmosphere operation avoids time-consuming inert gas purging procedures, while the straightforward scalability from laboratory to commercial production minimizes technology transfer delays. The robust nature of the process across diverse substrates enables rapid batch turnaround without extensive revalidation, allowing suppliers to maintain lean inventories while meeting urgent customer demands for critical pharmaceutical intermediates.
- Reduced Waste Treatment: With water as the sole byproduct, this methodology eliminates the complex waste streams generated by traditional methods using halogenated reagents that require specialized treatment facilities. The high atom economy (98%+ based on mass balance) substantially reduces raw material consumption while minimizing disposal costs associated with hazardous chemical waste. The elimination of heavy metal catalysts avoids expensive metal recovery processes and associated regulatory compliance burdens, creating significant cost reduction in pharmaceutical manufacturing operations while supporting sustainability initiatives through reduced environmental impact.
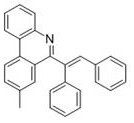
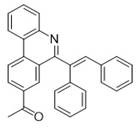
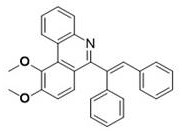
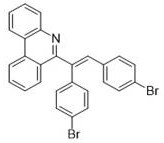
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 6-Alkenyl Phenanthridine Derivatives Supplier
While this ruthenium-catalyzed methodology demonstrates significant potential for pharmaceutical intermediate production, NINGBO INNO PHARMCHEM brings extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production to ensure reliable supply of complex heterocyclic compounds. Our rigorous QC labs maintain stringent purity specifications through advanced analytical capabilities including NMR, HRMS, and HPLC validation protocols that exceed industry standards for pharmaceutical intermediates. As a trusted CDMO partner, we combine cutting-edge synthetic expertise with robust manufacturing infrastructure to deliver consistent quality across all production scales.
We invite you to initiate a Customized Cost-Saving Analysis for your specific phenanthridine derivative requirements by contacting our technical procurement team. Request detailed COA data and route feasibility assessments to evaluate how this patented methodology can enhance your supply chain resilience while delivering substantial cost reduction in pharmaceutical intermediate manufacturing.
