Industrial Synthesis of 2-Methyl-1,2,4-Triazole-3-Amine: A Scalable Route for Pharmaceutical Intermediates
The development of efficient synthetic routes for nitrogen-containing heterocycles remains a cornerstone of modern medicinal chemistry, particularly for scaffolds that serve as critical building blocks in drug discovery. Patent CN111620829A introduces a robust and industrially viable methodology for the synthesis of 2-methyl-1,2,4-triazole-3-amine, a compound of significant interest due to its potential utility in various bioactive molecules. This technology addresses a longstanding gap in the market where suitable industrial synthesis methods were previously scarce or overly complex. By leveraging a concise three-step sequence, the patent outlines a pathway that transforms easily accessible raw materials into high-value intermediates with remarkable efficiency. For R&D directors and process chemists, this represents a pivotal advancement, offering a reproducible protocol that balances yield optimization with operational simplicity. The strategic design of this route not only enhances the accessibility of this specific triazole derivative but also sets a new benchmark for the commercial scale-up of complex heterocycles, ensuring a steady supply for downstream pharmaceutical applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of substituted triazoles has often been plagued by challenges related to regioselectivity and the requirement for harsh reaction conditions. Traditional methods frequently rely on direct alkylation strategies that suffer from poor control over N-substitution patterns, leading to difficult-to-separate isomeric mixtures that compromise overall purity. Furthermore, many existing protocols necessitate the use of expensive or hazardous reagents, such as strong bases at cryogenic temperatures or toxic alkylating agents, which pose significant safety risks and environmental burdens in a manufacturing setting. The lack of a standardized, high-yielding industrial process has historically constrained the availability of 2-methyl-1,2,4-triazole-3-amine, forcing procurement teams to rely on small-batch custom synthesis with long lead times. These conventional approaches often result in substantial waste generation and inflated production costs, making them economically unfeasible for large-scale API manufacturing where cost reduction in API manufacturing is a primary objective.
The Novel Approach
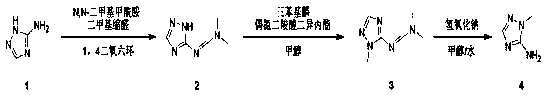
In stark contrast to these legacy methods, the novel approach detailed in the patent employs a sophisticated yet practical three-step strategy that circumvents traditional pitfalls. The process initiates with the formation of a formamidine intermediate using N,N-dimethylformamide dimethyl acetal, a reagent known for its mild reactivity and ease of handling. This is followed by a highly selective N-methylation step utilizing a Mitsunobu-type reaction condition with triphenylphosphine and diisopropyl azodicarboxylate, which ensures precise substitution at the desired nitrogen atom without affecting other sensitive functionalities. The final step involves a straightforward hydrolysis under basic conditions to reveal the target amine. This sequence not only streamlines the synthetic pathway but also significantly improves the overall yield, reportedly reaching up to 40.5%, which is a substantial improvement over fragmented literature methods. By utilizing common solvents like 1,4-dioxane and methanol, the process aligns perfectly with the needs of a reliable pharmaceutical intermediate supplier seeking to optimize throughput.
Mechanistic Insights into Triazole N-Methylation and Hydrolysis
The core of this synthetic innovation lies in the mechanistic elegance of the second step, which effectively achieves N-methylation through a modified Mitsunobu protocol. In this transformation, triphenylphosphine reacts with diisopropyl azodicarboxylate to generate a reactive phosphonium intermediate, which subsequently activates the methanol solvent as a nucleophile source. This activated species then attacks the nitrogen center of the triazole ring, facilitated by the electron-withdrawing nature of the adjacent formamidine group formed in the first step. This specific activation mode is crucial for overcoming the inherent low nucleophilicity of the triazole nitrogen, ensuring that the methylation occurs selectively at the N1 position rather than forming unwanted N2 or N4 isomers. The reaction proceeds smoothly at ambient temperature (25°C), which minimizes thermal degradation of sensitive intermediates and reduces energy consumption. Understanding this mechanism allows process chemists to fine-tune stoichiometry and addition rates to maximize conversion while minimizing the formation of triphenylphosphine oxide byproducts, which can be easily removed during subsequent purification stages.
Following the methylation, the final hydrolysis step is equally critical for ensuring the integrity of the final product's impurity profile. The treatment of the methylated intermediate with sodium hydroxide in a methanol-water mixture facilitates the cleavage of the formamidine protecting group, regenerating the primary amine functionality at the 3-position of the triazole ring. This hydrolysis is conducted at a moderate temperature of 70°C, which is sufficient to drive the reaction to completion overnight without inducing decomposition of the triazole core. The choice of a biphasic or mixed solvent system here is strategic, as it ensures adequate solubility of both the organic intermediate and the inorganic base, promoting homogeneous reaction kinetics. From a quality control perspective, this step is vital for removing residual reagents and ensuring that the final 2-methyl-1,2,4-triazole-3-amine meets the rigorous purity standards required for high-purity triazole derivatives used in drug substance synthesis. The ability to control these mechanistic variables precisely is what distinguishes this patent as a superior method for commercial production.
How to Synthesize 2-Methyl-1,2,4-Triazole-3-Amine Efficiently
The execution of this synthesis requires careful attention to reaction parameters to ensure optimal yields and reproducibility across different scales. The process begins with the reflux of 3-amino-1,2,4-triazole with N,N-dimethylformamide dimethyl acetal in anhydrous 1,4-dioxane, a step that must be monitored to ensure complete consumption of the starting material before proceeding. The subsequent methylation step demands the controlled addition of reagents at low temperatures initially to manage exotherms, followed by stirring at room temperature to allow the coupling to proceed to completion. Finally, the hydrolysis step requires efficient mixing and heating to ensure full deprotection. For detailed operational parameters, stoichiometry, and workup procedures, please refer to the standardized guide below which outlines the precise execution of this patented methodology.
- React 3-amino-1,2,4-triazole with N,N-dimethylformamide dimethyl acetal in 1,4-dioxane at 100°C for 5 hours to form the intermediate.
- Perform N-methylation using triphenylphosphine and diisopropyl azodicarboxylate in methanol at 25°C for 12 hours.
- Hydrolyze the resulting intermediate with sodium hydroxide in methanol/water at 70°C overnight to yield the final amine.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented synthesis route offers compelling advantages that directly address the pain points of procurement managers and supply chain heads. The reliance on commodity chemicals such as 3-amino-1,2,4-triazole, triphenylphosphine, and standard solvents means that the supply chain is robust and less susceptible to the volatility associated with exotic or specialized reagents. This accessibility translates into greater supply continuity and reduced risk of production delays caused by raw material shortages. Furthermore, the operational simplicity of the process, characterized by moderate temperatures and atmospheric pressure conditions, lowers the barrier for implementation in existing manufacturing facilities without requiring significant capital investment in specialized equipment. These factors collectively contribute to a more resilient supply chain capable of meeting the demanding timelines of the pharmaceutical industry.
- Cost Reduction in Manufacturing: The economic viability of this process is driven by the elimination of costly cryogenic operations and the use of high-efficiency reagents that minimize waste. By avoiding the need for expensive metal catalysts or complex protection-deprotection sequences found in alternative routes, the overall cost of goods sold is significantly optimized. The high yield of the final hydrolysis step further enhances material efficiency, ensuring that a greater proportion of input mass is converted into valuable product. This logical deduction of cost savings, derived from the streamlined nature of the chemistry, positions this method as a highly competitive option for cost reduction in API manufacturing, allowing companies to improve their margins without compromising on quality.
- Enhanced Supply Chain Reliability: The use of widely available starting materials ensures that the production of this intermediate is not bottlenecked by single-source suppliers. 3-Amino-1,2,4-triazole is a bulk chemical with a mature global supply network, providing a stable foundation for long-term production planning. Additionally, the robustness of the reaction conditions means that the process is less sensitive to minor variations in raw material quality or environmental factors, leading to consistent batch-to-batch performance. This reliability is crucial for reducing lead time for high-purity intermediates, enabling pharmaceutical companies to accelerate their development timelines and bring life-saving therapies to market faster with confidence in their material supply.
- Scalability and Environmental Compliance: The process is inherently designed for scalability, with reaction conditions that translate seamlessly from laboratory benchtop to multi-ton commercial reactors. The solvents used, primarily methanol and 1,4-dioxane, are well-understood in terms of recovery and recycling, facilitating effective waste management and adherence to environmental regulations. The absence of heavy metals or persistent organic pollutants in the reaction scheme simplifies the effluent treatment process, reducing the environmental footprint of the manufacturing operation. This alignment with green chemistry principles not only mitigates regulatory risk but also enhances the corporate sustainability profile of manufacturers adopting this technology for the commercial scale-up of complex heterocycles.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and application of 2-methyl-1,2,4-triazole-3-amine. These insights are derived directly from the technical specifications and beneficial effects outlined in the patent documentation, providing clarity for stakeholders evaluating this technology for integration into their supply chains. Understanding these details is essential for making informed decisions about process adoption and vendor selection.
Q: What are the key advantages of this synthesis route for 2-methyl-1,2,4-triazole-3-amine?
A: The patented route utilizes readily available starting materials like 3-amino-1,2,4-triazole and avoids harsh cryogenic conditions, making it highly suitable for industrial scale-up with an overall yield reaching up to 40.5%.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the process is designed for scalability, utilizing common solvents like 1,4-dioxane and methanol, and standard reagents such as triphenylphosphine, which ensures supply chain reliability and ease of operation.
Q: How is purity controlled in this synthetic method?
A: Purity is managed through controlled reaction temperatures and specific workup procedures, including silica gel chromatography, ensuring the final product meets stringent specifications for pharmaceutical applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Methyl-1,2,4-Triazole-3-Amine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of having a dependable partner for the supply of advanced pharmaceutical intermediates. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements with consistency and precision. We are committed to delivering products that adhere to stringent purity specifications, supported by our rigorous QC labs that employ state-of-the-art analytical techniques to verify every batch. Our capability to implement complex synthetic routes like the one described in CN111620829A demonstrates our technical prowess and dedication to quality, making us the ideal choice for your long-term sourcing needs.
We invite you to engage with our technical procurement team to discuss how we can support your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how our optimized manufacturing processes can benefit your bottom line. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your application, ensuring that you have all the necessary information to move forward with confidence in your supply chain strategy.
