Advanced Chiral Resolution Technology for High-Purity Pharmaceutical Intermediates and Commercial Scale-Up
Advanced Chiral Resolution Technology for High-Purity Pharmaceutical Intermediates and Commercial Scale-Up
The pharmaceutical industry continuously demands robust, scalable, and cost-effective methods for producing chiral intermediates, particularly for cardiovascular and antiviral therapies. Patent CN100546969C introduces a groundbreaking methodology for the optical resolution of 2-hydroxymethyl-3-phenylpropionic acid, a critical building block for renin inhibitors and dual ACE/NEP inhibitors like Sinorphan (Ecadotril). This technology addresses the longstanding challenges of high production costs and low efficiency associated with traditional chiral separation techniques. By utilizing N-Octylglucamine as a novel resolving agent, the process achieves exceptional optical purity while leveraging inexpensive, readily available raw materials. For R&D directors and procurement specialists seeking a reliable pharmaceutical intermediate supplier, this patent represents a significant leap forward in process chemistry, enabling the production of high-value chiral acids with minimized environmental impact and maximized economic return.
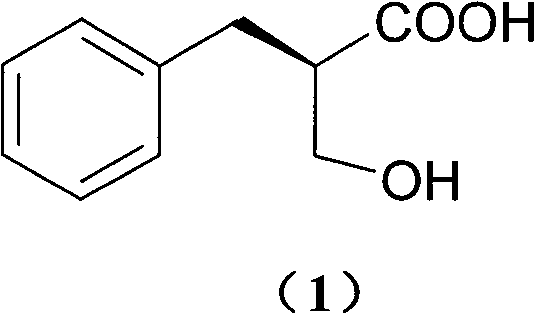
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the resolution of racemic 2-hydroxymethyl-3-phenylpropionic acid has been plagued by significant economic and operational bottlenecks. Prior art, such as that described in EP 906900, relies heavily on resolving agents like cis-1-amino-2-indanol or 3-methyl-2-phenyl-1-butylamine. While these agents can achieve reasonable separation, they suffer from severe drawbacks that hinder industrial adoption. Cis-1-amino-2-indanol is notoriously difficult to synthesize and commands a prohibitively high market price, drastically inflating the cost of goods sold (COGS) for the final API. Similarly, 3-methyl-2-phenyl-1-butylamine presents supply chain vulnerabilities due to its complex synthesis. Furthermore, alternative agents like alpha-phenylethylamine often exhibit poor resolution efficiency, requiring multiple recrystallization cycles that erode overall yield and increase solvent consumption. These legacy methods create a fragile supply chain where minor fluctuations in resolving agent availability can halt production lines, making them unsuitable for the rigorous demands of modern API manufacturing.
The Novel Approach
In stark contrast, the methodology disclosed in CN100546969C utilizes N-Octylglucamine, a sugar-derived amine that offers a superior balance of performance and economics. This novel approach capitalizes on the unique stereochemical interaction between the glucamine derivative and the racemic acid to form diastereomeric salts with distinct solubility profiles. The process is remarkably versatile, accommodating a wide range of green solvents including methanol, ethanol, isopropanol, and even aqueous mixtures, which simplifies downstream processing and solvent recovery. By shifting away from exotic, expensive amines to a commodity-grade resolving agent, manufacturers can achieve cost reduction in pharmaceutical intermediate manufacturing without sacrificing quality. The reaction scheme illustrates how the racemic mixture is efficiently converted into separable salts, paving the way for high-throughput production.
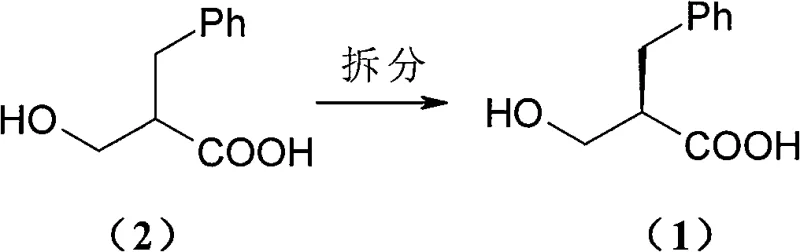
Mechanistic Insights into Diastereomeric Salt Formation
The core of this technology lies in the precise thermodynamic control of diastereomeric salt crystallization. When racemic 2-hydroxymethyl-3-phenylpropionic acid reacts with optically pure N-Octylglucamine, two distinct diastereomeric salts are formed in situ. Due to the specific spatial arrangement of the hydroxyl and carboxyl groups relative to the bulky octyl-glucamine backbone, one diastereomer exhibits significantly lower solubility in the chosen solvent system compared to its counterpart. This solubility differential is the driving force for the separation; upon cooling or concentration, the less soluble salt precipitates selectively, leaving the unwanted enantiomer in the mother liquor. The hydrogen bonding network facilitated by the multiple hydroxyl groups on the glucamine moiety enhances the lattice energy of the preferred salt, ensuring that the precipitate is enriched with the desired stereoisomer. This mechanism allows for the isolation of the target enantiomer with high initial optical purity, often exceeding 95% ee after the first crystallization, which is a critical metric for high-purity chiral intermediates.
Furthermore, the process incorporates a robust recycling loop for the resolving agent, which is essential for sustainable chemistry. After the diastereomeric salt is isolated and filtered, it is treated with a strong mineral acid, such as hydrochloric acid, to liberate the free optically active acid. The N-Octylglucamine remains in the aqueous phase as a salt, from which it can be regenerated by basification and extraction. This recovery capability is not merely an environmental benefit but a substantial economic driver, as it decouples the cost of the resolving agent from the per-batch production cost. The ability to tune the solvent system—using mixtures of alcohols and water or ketones—allows process chemists to optimize the solubility gap further, ensuring that impurities and the minor enantiomer remain in solution. This level of control over the crystallization kinetics is what enables the commercial scale-up of complex chiral resolutions with consistent batch-to-batch reproducibility.
How to Synthesize (R)-2-Hydroxymethyl-3-phenylpropionic Acid Efficiently
Implementing this resolution strategy requires careful attention to stoichiometry and solvent selection to maximize yield and optical purity. The patent outlines a straightforward protocol where the molar ratio of N-Octylglucamine to the racemic acid is typically maintained between 0.5 and 1.5 equivalents, with a 1:1 ratio often proving optimal for balancing cost and efficiency. The choice of solvent is critical; while methanol and ethanol are preferred for their ability to dissolve the reactants at elevated temperatures and precipitate the salt upon cooling, the system is forgiving enough to allow for process adjustments based on available infrastructure. Detailed standard operating procedures regarding temperature ramps, stirring rates, and filtration techniques are essential to prevent oiling out and ensure the formation of high-quality crystals. For a comprehensive breakdown of the specific experimental conditions and workup procedures, please refer to the standardized synthesis guide below.
- Mix racemic 2-hydroxymethyl-3-phenylpropionic acid with N-Octylglucamine in a suitable solvent such as methanol or ethanol to form diastereomeric salts.
- Separate the less soluble diastereomeric salt through filtration or centrifugation, followed by recrystallization to enhance optical purity.
- Liberate the optically active acid from the purified salt using acid or base treatment, followed by extraction and final crystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this N-Octylglucamine-based resolution process offers transformative advantages that directly impact the bottom line and operational resilience. Traditional methods relying on scarce chiral amines expose manufacturers to volatile pricing and supply disruptions, whereas this new method utilizes a resolving agent derived from abundant renewable resources. The elimination of expensive, hard-to-source reagents removes a major bottleneck in the supply chain, ensuring continuous production flow even during global raw material shortages. Moreover, the simplified solvent system reduces the complexity of waste management and solvent recovery units, leading to lower operational expenditures (OPEX). By adopting this technology, companies can secure a more stable supply of critical intermediates while simultaneously driving down the total cost of ownership for their synthetic routes.
- Cost Reduction in Manufacturing: The primary economic driver of this innovation is the drastic reduction in raw material costs associated with the resolving agent. Unlike cis-1-amino-2-indanol, which requires multi-step synthesis and purification, N-Octylglucamine is commercially available at a fraction of the price. This substitution eliminates the need for costly custom synthesis of the chiral auxiliary, directly lowering the variable cost per kilogram of the final product. Additionally, the high recovery rate of the resolving agent means that the effective consumption of the amine is negligible over multiple batches, compounding the savings over the lifecycle of the product. This economic efficiency makes the process viable for large-scale generic drug production where margin pressure is intense.
- Enhanced Supply Chain Reliability: Supply chain continuity is paramount for pharmaceutical manufacturers, and this process significantly de-risks the sourcing of critical inputs. N-Octylglucamine is a commodity chemical with a robust global supply base, unlike specialized chiral amines that may have single-source suppliers. This diversity in sourcing options protects against geopolitical disruptions and logistics delays. Furthermore, the flexibility in solvent choice—allowing the use of common industrial solvents like ethanol or acetone—means that production is not held hostage by the availability of niche specialty solvents. This adaptability ensures that manufacturing schedules can be met consistently, reducing lead time for high-purity intermediates and improving customer satisfaction.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, the process is designed for green chemistry principles. The use of alcohol and water-based solvent systems minimizes the generation of hazardous organic waste, simplifying compliance with increasingly stringent environmental regulations. The crystallization-based separation is inherently scalable; moving from kilogram to ton-scale simply requires proportional increases in reactor volume without changing the fundamental thermodynamics of the separation. This linear scalability reduces the risk and cost associated with technology transfer from pilot plant to commercial manufacturing. The ability to recycle the resolving agent further reduces the E-factor (mass of waste per mass of product), aligning with corporate sustainability goals and reducing waste disposal costs.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this resolution technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent literature, offering clarity on how this method outperforms legacy techniques. Understanding these nuances is crucial for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios. The answers highlight the practical benefits regarding purity, cost, and operational simplicity that define this innovative approach.
Q: Why is N-Octylglucamine preferred over traditional resolving agents like cis-1-amino-2-indanol?
A: N-Octylglucamine is significantly more cost-effective and easier to source compared to expensive agents like cis-1-amino-2-indanol or 3-methyl-2-phenyl-1-butylamine, while maintaining comparable or superior resolution efficiency and optical purity.
Q: What solvents are compatible with this resolution process?
A: The process is highly flexible regarding solvents, supporting alcohols like methanol and ethanol, ketones like acetone and butanone, as well as aqueous mixtures, allowing for optimization based on solubility profiles and recovery needs.
Q: Can the resolving agent be recovered and reused?
A: Yes, the N-Octylglucamine resolving agent can be efficiently recovered from the mother liquor or aqueous phases after acid/base liberation, significantly reducing waste and overall production costs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Hydroxymethyl-3-phenylpropionic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of complex pharmaceutical intermediates requires more than just a patent; it demands deep process expertise and unwavering commitment to quality. Our team of expert chemists has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We understand the critical nature of chiral purity in cardiovascular drug synthesis and maintain stringent purity specifications through our rigorous QC labs, utilizing advanced chiral HPLC and polarimetry to guarantee every batch meets the highest standards. Our facility is equipped to handle the specific solvent systems and crystallization requirements outlined in CN100546969C, delivering a product that is ready for immediate downstream coupling reactions.
We invite global partners to collaborate with us to leverage this cost-effective resolution technology for their supply chains. Whether you are looking to optimize an existing route or develop a new supply source for 2-hydroxymethyl-3-phenylpropionic acid, our technical sales team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage potential clients to contact our technical procurement team to request specific COA data and route feasibility assessments, allowing you to make informed decisions based on real-world performance metrics. Let us be your strategic partner in delivering high-quality, affordable chiral building blocks for the next generation of life-saving medicines.
