Advanced Asymmetric Synthesis of (1R,3S)-3-Amino-1-Cyclopentanol for Commercial API Manufacturing
Advanced Asymmetric Synthesis of (1R,3S)-3-Amino-1-Cyclopentanol for Commercial API Manufacturing
The global pharmaceutical landscape is continuously evolving to meet the demands of potent antiretroviral therapies, with Bictegravir standing out as a cornerstone in modern HIV treatment regimens. As detailed in the groundbreaking patent CN110862325B, published on August 25, 2020, a novel preparation method for (1R,3S)-3-amino-1-cyclopentanol has been established, addressing critical bottlenecks in the supply chain of this vital intermediate. This compound serves as a key chiral building block for Bictegravir, a drug marketed under the brand name Biktarvy, which combines Bictegravir with Emtricitabine and Tenofovir alafenamide. 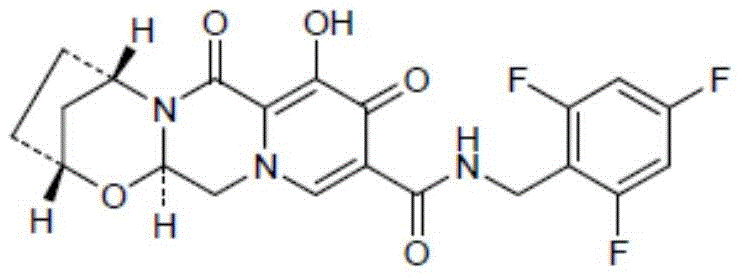 The structural complexity of Bictegravir necessitates precise stereochemical control, and the traditional reliance on chiral resolution has long plagued manufacturers with efficiency losses. This new technology leverages an N-acyl hydroxylamine compound as a chiral source to induce asymmetry during a cycloaddition reaction with cyclopentadiene, effectively constructing two chiral centers in a single strategic transformation. By shifting the paradigm from resolution to asymmetric synthesis, this innovation promises to redefine the economic and operational feasibility of producing high-purity pharmaceutical intermediates for the global market.
The structural complexity of Bictegravir necessitates precise stereochemical control, and the traditional reliance on chiral resolution has long plagued manufacturers with efficiency losses. This new technology leverages an N-acyl hydroxylamine compound as a chiral source to induce asymmetry during a cycloaddition reaction with cyclopentadiene, effectively constructing two chiral centers in a single strategic transformation. By shifting the paradigm from resolution to asymmetric synthesis, this innovation promises to redefine the economic and operational feasibility of producing high-purity pharmaceutical intermediates for the global market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of chiral cyclopentanol derivatives like (1R,3S)-3-amino-1-cyclopentanol has relied heavily on chiral resolution techniques, encompassing both enzymatic resolution and chemical resolution using chiral acids. While these methods are well-understood, they suffer from an intrinsic and insurmountable theoretical limitation: the maximum yield can never exceed 50% because the unwanted enantiomer must be discarded or recycled through energy-intensive processes. In practical industrial settings, the actual yield often drops even further, typically ranging between 30% and 45%, which represents a massive waste of valuable raw materials and processing capacity. Furthermore, alternative chiral source synthesis methods that do exist often involve starting materials that are exceptionally difficult to synthesize, prohibitively expensive, or economically unviable for large-scale operations. These conventional pathways create significant supply chain vulnerabilities, where the cost of goods sold is inflated by the sheer volume of wasted feedstock, and the environmental footprint is unnecessarily enlarged by the disposal of the opposite enantiomer. For procurement managers and supply chain heads, relying on these legacy technologies means accepting higher volatility in pricing and longer lead times due to the inefficiency of the underlying chemistry.
The Novel Approach
In stark contrast to the wastefulness of resolution, the methodology disclosed in patent CN110862325B introduces a highly efficient asymmetric cycloaddition strategy that fundamentally alters the economic equation of intermediate manufacturing. This novel approach utilizes an N-acyl hydroxylamine compound, which acts as a powerful chiral inducer, reacting directly with cyclopentadiene to construct the requisite two chiral centers of the target molecule with high stereoselectivity. The raw materials for this route, specifically the N-acyl hydroxylamine precursors, are derived from readily available chiral hydroxy acid esters and hydroxylamine via a simple one-step ammonolysis reaction, ensuring a wide source of supply and drastically reduced raw material costs. The process is characterized by its operational simplicity, mild reaction conditions, and superior atom economy, as it avoids the generation of stoichiometric amounts of unwanted isomers. By achieving high optical purity and stable quality directly from the synthesis rather than through purification of a racemate, this method offers a robust platform for large-scale industrial production that aligns perfectly with the goals of cost reduction in pharmaceutical intermediate manufacturing. The ability to tune the R group from C1-C4 alkyl to C6-C10 aryl provides additional flexibility for process optimization, making this a versatile solution for diverse production needs.
Mechanistic Insights into Asymmetric Cycloaddition and Chiral Induction
The core of this technological breakthrough lies in the sophisticated mechanism of the asymmetric cycloaddition reaction, where the chirality of the N-acyl hydroxylamine compound dictates the stereochemical outcome of the cyclopentane ring formation. As illustrated in the reaction schemes provided in the patent, the N-acyl hydroxylamine serves not merely as a reactant but as a chiral template that guides the approach of the cyclopentadiene diene. 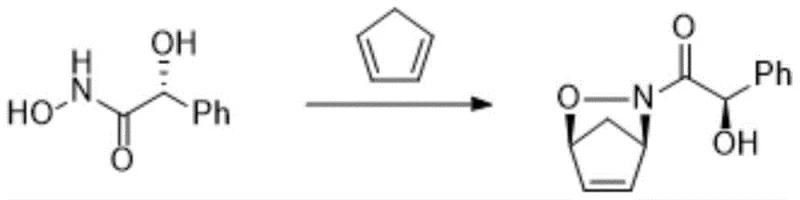 Under the influence of an oxidizing agent such as sodium periodate, oxygen, or N-bromosuccinimide, the reaction proceeds through a concerted transition state that favors the formation of specific diastereomers, evidenced by the high diastereomeric ratios (dr) reported, such as 3.5/1 in Example 1 and up to 4.5/1 in Example 4. This high level of stereocontrol is critical because it minimizes the burden on downstream purification steps, directly impacting the overall yield and purity of the final API intermediate. The subsequent hydrogenation steps, utilizing catalysts like palladium on carbon or Raney nickel under controlled pressures of 0.05-2 MPa, further refine the molecular architecture without compromising the established chirality. The careful selection of reaction temperatures, ranging from cryogenic conditions like -20°C for the cycloaddition to moderate temperatures of 20-50°C for the final reduction, ensures that the delicate chiral centers remain intact throughout the synthetic sequence. This mechanistic precision allows R&D directors to confidently scale the process, knowing that the impurity profile is managed at the molecular level through intelligent reaction design rather than brute-force separation.
Under the influence of an oxidizing agent such as sodium periodate, oxygen, or N-bromosuccinimide, the reaction proceeds through a concerted transition state that favors the formation of specific diastereomers, evidenced by the high diastereomeric ratios (dr) reported, such as 3.5/1 in Example 1 and up to 4.5/1 in Example 4. This high level of stereocontrol is critical because it minimizes the burden on downstream purification steps, directly impacting the overall yield and purity of the final API intermediate. The subsequent hydrogenation steps, utilizing catalysts like palladium on carbon or Raney nickel under controlled pressures of 0.05-2 MPa, further refine the molecular architecture without compromising the established chirality. The careful selection of reaction temperatures, ranging from cryogenic conditions like -20°C for the cycloaddition to moderate temperatures of 20-50°C for the final reduction, ensures that the delicate chiral centers remain intact throughout the synthetic sequence. This mechanistic precision allows R&D directors to confidently scale the process, knowing that the impurity profile is managed at the molecular level through intelligent reaction design rather than brute-force separation.
Furthermore, the patent elucidates a robust mechanism for impurity control through the strategic cleavage of the amide bond in Intermediate II. Whether performed under acidic catalysis using reagents like hydrochloric acid or p-toluenesulfonic acid, or under basic conditions with ammonia or hydrazine hydrate, this step is designed to cleanly release the chiral amine framework while removing the chiral auxiliary. The versatility in choosing between acid and base catalysis provides process chemists with the ability to optimize for specific impurity profiles or equipment constraints. For instance, acid-catalyzed alcoholysis allows for the direct isolation of the intermediate salt, streamlining the workflow by eliminating a neutralization and extraction step. The final hydrogenation of Intermediate III to the target (1R,3S)-3-amino-1-cyclopentanol is executed under rigorous conditions with high-activity palladium carbon to ensure complete reduction of any remaining unsaturation or functional groups that could act as genotoxic impurities. This multi-layered approach to mechanism design ensures that the final product meets the stringent purity specifications required for HIV medication, with optical purity consistently exceeding 99.5% as demonstrated in Examples 18 through 20. Such rigorous control over the chemical pathway is essential for maintaining regulatory compliance and ensuring patient safety in the final drug product.
How to Synthesize (1R,3S)-3-Amino-1-Cyclopentanol Efficiently
The synthesis of this critical pharmaceutical intermediate follows a logical four-step sequence that balances high yield with operational safety and scalability. The process begins with the preparation of the chiral N-acyl hydroxylamine precursor, followed by the pivotal asymmetric cycloaddition with cyclopentadiene to establish the ring system and stereochemistry. Subsequent hydrogenation reduces the double bond, and amide cleavage releases the chiral amine, which is finally reduced to the target amino-alcohol. Each step has been optimized in the patent examples to demonstrate feasibility across different scales and reagent choices, providing a comprehensive roadmap for process implementation. The detailed standardized synthesis steps below outline the specific conditions and parameters required to replicate this high-efficiency route in a GMP environment.
- Perform asymmetric cycloaddition of N-acyl hydroxylamine with cyclopentadiene using an oxidant to form Intermediate I.
- Conduct catalytic hydrogenation of Intermediate I using Pd/C or Raney Nickel to obtain Intermediate II.
- Cleave the amide bond of Intermediate II via acid or base catalysis to generate Intermediate III or its salt.
- Execute final hydrogenation reduction of Intermediate III to yield high-purity (1R,3S)-3-amino-1-cyclopentanol.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this asymmetric synthesis route offers transformative advantages that extend far beyond simple chemical yield improvements. The most significant impact is the elimination of the inherent 50% material loss associated with traditional chiral resolution, which fundamentally alters the cost structure of the intermediate. By utilizing every mole of the chiral starting material to produce the desired enantiomer, the process achieves a level of atom economy that translates directly into substantial cost savings on raw material procurement. This efficiency gain is compounded by the use of widely available and inexpensive reagents like cyclopentadiene and common oxidants, reducing dependency on exotic or proprietary chiral pools that often carry high price premiums and supply risks. Additionally, the mild reaction conditions, which avoid extreme temperatures or hazardous high-pressure environments in the initial steps, lower the barrier for manufacturing entry and reduce the capital expenditure required for specialized reactor infrastructure. These factors combine to create a supply chain that is not only more cost-effective but also more resilient against market fluctuations and raw material shortages.
- Cost Reduction in Manufacturing: The shift from resolution to asymmetric synthesis removes the economic penalty of discarding half the product, leading to a drastic reduction in the cost of goods sold. The ability to use simple, commodity-grade chemicals for the chiral induction step further drives down variable costs, while the high yields reported in the patent examples minimize waste disposal expenses. This holistic reduction in production costs allows for more competitive pricing strategies in the global API market without sacrificing margin.
- Enhanced Supply Chain Reliability: By relying on broad-spectrum raw materials such as chiral hydroxy acid esters and cyclopentadiene, manufacturers can diversify their supplier base and mitigate the risk of single-source bottlenecks. The robustness of the reaction conditions ensures consistent batch-to-batch quality, reducing the likelihood of production delays caused by failed batches or out-of-specification results. This reliability is crucial for maintaining the continuous supply of life-saving antiretroviral medications to patients worldwide.
- Scalability and Environmental Compliance: The process is explicitly designed for large-scale industrial production, with examples demonstrating successful execution from gram to multi-gram scales using standard equipment. The higher atom economy and reduced solvent usage associated with the streamlined workflow contribute to a smaller environmental footprint, aligning with increasingly strict global regulations on pharmaceutical manufacturing emissions and waste. This sustainability advantage enhances the corporate social responsibility profile of the supply chain while ensuring long-term regulatory compliance.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. They are derived directly from the experimental data and beneficial effects described in patent CN110862325B, providing clarity on the practical aspects of adopting this route for commercial production. Understanding these details is essential for technical teams evaluating the feasibility of integrating this process into their existing manufacturing portfolios.
Q: What is the primary advantage of this synthesis over chiral resolution?
A: Unlike chiral resolution which has a theoretical maximum yield of 50%, this asymmetric synthesis utilizes chiral induction to construct two chiral centers simultaneously, significantly improving atom economy and raw material utilization without the inherent waste of discarding the unwanted enantiomer.
Q: What oxidants are suitable for the initial cycloaddition step?
A: The patent specifies several effective oxidizing agents including sodium periodate, oxygen, hydrogen peroxide, and N-bromosuccinimide (NBS), allowing flexibility in process optimization based on safety and cost considerations.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the method features mild reaction conditions (e.g., -10°C to 50°C), simple operations, and widely available raw materials, making it highly scalable for commercial manufacturing of HIV drug intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (1R,3S)-3-Amino-1-Cyclopentanol Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the development and manufacture of next-generation HIV therapies like Bictegravir. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including the high optical purity (>99.5%) demanded by this specific chiral intermediate. We are committed to leveraging advanced synthetic methodologies, such as the asymmetric cycloaddition route described in CN110862325B, to deliver value-driven solutions that enhance your project's economic viability.
We invite you to engage with our technical procurement team to discuss how we can tailor this synthesis to your specific volume and quality requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the potential financial benefits of switching to this more efficient manufacturing route. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your supply chain for (1R,3S)-3-amino-1-cyclopentanol is built on a foundation of scientific excellence and commercial reliability.
