Advanced Pd-Catalyzed Carbonylation for Scalable 3-Benzylidene-2,3-Dihydroquinolone Production
The pharmaceutical industry continuously seeks robust synthetic routes for nitrogen-containing heterocycles due to their prevalence in bioactive molecules. Patent CN113735826A introduces a significant advancement in the preparation of 3-benzylidene-2,3-dihydroquinolone compounds, a scaffold known for its potential in developing analgesic and antitumor agents. As illustrated in the structural diversity of related bioactive compounds, the ability to efficiently functionalize the dihydroquinolone core is critical for drug discovery pipelines. 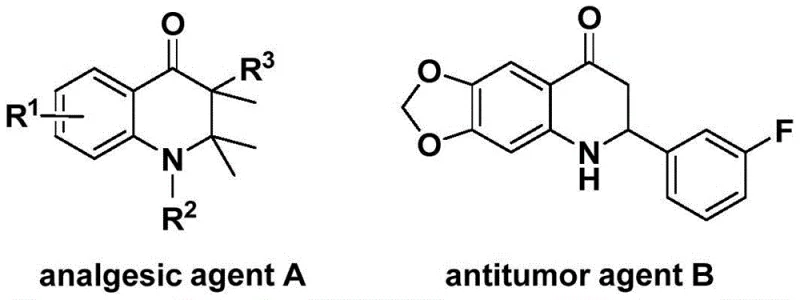 . This patent discloses a novel palladium-catalyzed carbonylation strategy that utilizes N-pyridylsulfonyl-o-iodoaniline and allenes as key starting materials. By employing a solid carbon monoxide surrogate instead of hazardous gaseous CO, the method addresses significant safety and handling concerns associated with traditional carbonylation reactions, positioning it as a highly attractive option for reliable pharmaceutical intermediate supplier networks seeking safer manufacturing protocols.
. This patent discloses a novel palladium-catalyzed carbonylation strategy that utilizes N-pyridylsulfonyl-o-iodoaniline and allenes as key starting materials. By employing a solid carbon monoxide surrogate instead of hazardous gaseous CO, the method addresses significant safety and handling concerns associated with traditional carbonylation reactions, positioning it as a highly attractive option for reliable pharmaceutical intermediate supplier networks seeking safer manufacturing protocols.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2,3-dihydroquinolone derivatives has often relied on multi-step sequences or reactions requiring harsh conditions that limit their utility in complex molecule synthesis. Traditional carbonylation approaches frequently necessitate the use of high-pressure carbon monoxide gas, which poses severe safety risks and requires specialized, expensive reactor infrastructure that many contract manufacturing organizations lack. Furthermore, conventional methods often suffer from poor atom economy and limited substrate tolerance, particularly when sensitive functional groups are present on the aromatic rings. The reliance on gaseous reagents also complicates the scalability of the process, as maintaining consistent gas-liquid mass transfer becomes increasingly difficult when moving from laboratory benchtop to commercial production vessels. These factors collectively contribute to higher production costs and extended lead times, creating bottlenecks in the supply chain for high-purity pharmaceutical intermediates.
The Novel Approach
The methodology described in CN113735826A overcomes these barriers by utilizing a solid carbon monoxide surrogate, specifically a phenol ester derivative, which releases CO in situ under mild thermal conditions. This innovation allows the reaction to proceed in standard glassware or stainless steel reactors without the need for high-pressure gas lines, drastically simplifying the engineering requirements for cost reduction in pharmaceutical intermediate manufacturing. The reaction couples N-pyridylsulfonyl-o-iodoaniline with various allenes in the presence of a palladium catalyst and a bidentate phosphine ligand. 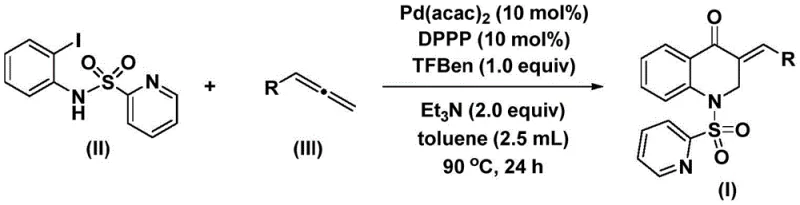 . This approach not only streamlines the synthetic route into a single pot but also demonstrates exceptional functional group compatibility, accommodating substituents such as methyl, methoxy, tert-butyl, and halogens at ortho, meta, and para positions without significant loss in yield. The result is a versatile platform technology capable of rapid library generation for medicinal chemistry campaigns.
. This approach not only streamlines the synthetic route into a single pot but also demonstrates exceptional functional group compatibility, accommodating substituents such as methyl, methoxy, tert-butyl, and halogens at ortho, meta, and para positions without significant loss in yield. The result is a versatile platform technology capable of rapid library generation for medicinal chemistry campaigns.
Mechanistic Insights into Palladium-Catalyzed Carbonylative Cyclization
The catalytic cycle begins with the oxidative addition of the palladium(0) species into the carbon-iodine bond of the N-pyridylsulfonyl-o-iodoaniline substrate, generating a reactive aryl-palladium intermediate. This step is facilitated by the electron-rich nature of the palladium center coordinated with the 1,3-bis(diphenylphosphino)propane (DPPP) ligand, which stabilizes the metal center throughout the cycle. Subsequently, carbon monoxide, released thermally from the solid surrogate, inserts into the aryl-palladium bond to form an acyl-palladium species. This acyl intermediate is pivotal, as it sets the stage for the subsequent interaction with the allene substrate. The coordination of the allene to the palladium center followed by migratory insertion creates a new carbon-carbon bond and an alkyl-palladium intermediate. Finally, an intramolecular nucleophilic attack or reductive elimination step closes the ring, releasing the 3-benzylidene-2,3-dihydroquinolone product and regenerating the active palladium catalyst. This mechanistic pathway ensures high regioselectivity and minimizes the formation of side products, which is essential for maintaining stringent purity specifications in API synthesis.
Impurity control in this system is inherently managed by the choice of the sulfonyl protecting group and the specific ligand environment. The pyridine sulfonyl group not only activates the aniline nitrogen for the cyclization event but also prevents unwanted N-alkylation side reactions that could occur with free amines. Furthermore, the use of triethylamine as an additive helps to neutralize any acidic byproducts generated during the decomposition of the CO surrogate, maintaining a neutral to slightly basic environment that favors the catalytic cycle. The robustness of this mechanism is evidenced by the consistent yields observed across a wide range of electronically diverse allenes, suggesting that the rate-determining step is likely insensitive to steric bulk on the allene terminus. This predictability allows process chemists to confidently project outcomes for new analogues, reducing the risk associated with scaling up complex organic syntheses.
How to Synthesize 3-Benzylidene-2,3-Dihydroquinolone Efficiently
The experimental procedure outlined in the patent provides a clear roadmap for executing this transformation with high reproducibility. The process involves charging a reaction vessel with the palladium catalyst, ligand, base, CO surrogate, and substrates in toluene, followed by heating to 90°C. The detailed standardized synthesis steps below outline the precise molar ratios and workup procedures required to achieve the reported yields of up to 93%.
- Combine Pd(acac)2 catalyst, DPPP ligand, triethylamine additive, solid CO surrogate, N-pyridine sulfonyl-o-iodoaniline, and the specific allene substrate in toluene solvent.
- Heat the reaction mixture to 90°C and maintain stirring for 24 to 48 hours to ensure complete conversion of the starting materials.
- Upon completion, filter the mixture, mix with silica gel, and purify the crude product via column chromatography to isolate the target 3-benzylidene-2,3-dihydroquinolone.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers tangible benefits regarding cost stability and operational flexibility. The shift from gaseous reagents to solid surrogates removes a major logistical hurdle, as solid chemicals are easier to transport, store, and handle than compressed gases, thereby reducing the regulatory burden and insurance costs associated with hazardous materials. Additionally, the starting materials, including the iodinated aniline derivatives and various allenes, are either commercially available or can be synthesized through straightforward pathways, ensuring a reliable supply chain for high-purity pharmaceutical intermediates. The simplicity of the workup, which involves filtration and standard chromatography, further reduces the processing time and solvent consumption compared to more cumbersome traditional methods.
- Cost Reduction in Manufacturing: The elimination of high-pressure equipment requirements represents a significant capital expenditure saving for manufacturing sites. By operating at atmospheric pressure with a solid CO source, facilities can utilize existing general-purpose reactors rather than investing in specialized autoclaves. Furthermore, the high catalytic efficiency and substrate compatibility mean that fewer purification cycles are needed to remove metal residues or side products, leading to substantial cost savings in downstream processing and waste management. The use of common solvents like toluene also aligns with standard solvent recovery systems, enhancing the overall economic viability of the process.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions ensures consistent batch-to-batch quality, which is critical for maintaining continuous supply to downstream API manufacturers. Since the reaction tolerates a wide variety of functional groups, suppliers can produce a diverse portfolio of intermediates using the same core platform technology, reducing the need for multiple dedicated production lines. This flexibility allows for rapid response to changing market demands and facilitates the quick scale-up of new drug candidates from clinical trials to commercial production without extensive process re-optimization.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, having been demonstrated effectively at the gram level with clear pathways to kilogram and tonne scales. The use of a solid CO surrogate minimizes the release of toxic gases into the environment, aligning with increasingly strict environmental regulations and corporate sustainability goals. The simplified post-treatment process reduces the volume of chemical waste generated, lowering disposal costs and minimizing the environmental footprint of the manufacturing operation, which is a key consideration for modern green chemistry initiatives.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this carbonylation technology. These insights are derived directly from the experimental data and mechanistic understanding provided in the patent documentation.
Q: What is the primary advantage of using a solid CO surrogate in this synthesis?
A: Using a solid carbon monoxide surrogate, such as the phenol ester described in the patent, eliminates the need for handling toxic and high-pressure gaseous CO, significantly enhancing operational safety and simplifying reactor requirements for scale-up.
Q: What is the substrate scope for the allene component in this reaction?
A: The process demonstrates excellent compatibility with various substituted aryl allenes, including those with electron-donating groups like methyl and methoxy, as well as electron-withdrawing halogens like bromine and chlorine, yielding products with high efficiency.
Q: Is this method suitable for large-scale industrial production?
A: Yes, the patent explicitly states the method is scalable to the gram level and potentially industrial scales due to the use of commercially available catalysts, mild reaction conditions (90°C), and straightforward post-processing via filtration and chromatography.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Benzylidene-2,3-Dihydroquinolone Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of efficient synthetic methodologies like the one described in CN113735826A for accelerating drug development timelines. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from laboratory discovery to market supply is seamless. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 3-benzylidene-2,3-dihydroquinolone intermediate meets the highest international standards for pharmaceutical applications.
We invite you to collaborate with us to leverage this advanced technology for your specific project needs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to support your R&D and supply chain objectives, ensuring a partnership built on technical excellence and reliability.
