Advanced Iron-Catalyzed One-Pot Synthesis of Symmetrical 2,4,6-Trisubstituted 1,3,5-Triazine Compounds for Commercial Pharmaceutical Manufacturing
Introduction to Novel Triazine Synthesis Technology
The landscape of nitrogen heterocycle synthesis is undergoing a significant transformation driven by the demand for greener, more cost-effective methodologies. As detailed in the recent intellectual property disclosure CN111454226B, a groundbreaking approach has been established for the construction of symmetrical 2,4,6-trisubstituted 1,3,5-triazine compounds. This technology leverages a simple yet highly effective one-pot reaction between readily available aromatic aldehydes and ammonium iodide, mediated by a trivalent iron salt catalyst. Unlike traditional methods that often rely on expensive nitriles or complex organometallic reagents, this innovation utilizes micromolecular aryl aldehydes as the primary carbon source. The process operates under relatively mild thermal conditions and achieves high selectivity, representing a substantial leap forward for the commercial scale-up of complex heterocycles used in pharmaceuticals and advanced materials.
The strategic importance of 1,3,5-triazine scaffolds cannot be overstated, given their ubiquitous presence in liquid crystals, reactive dyes, and organic light-emitting diodes (OLEDs). The ability to synthesize these cores directly from commodity chemicals like benzaldehyde derivatives drastically alters the supply chain dynamics. By eliminating the need for pre-functionalized nitrogen sources such as amidines or nitriles, this method streamlines the synthetic route, reducing both the number of operational steps and the generation of hazardous waste. For industry stakeholders, this translates to a more robust and reliable supply of high-purity triazine compounds, ensuring continuity for downstream applications in drug discovery and material science.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the 1,3,5-triazine ring has been fraught with synthetic challenges that hinder large-scale adoption. Conventional pathways typically involve the cyclotrimerization of nitriles or the condensation of amidine salts, processes that often necessitate harsh reaction conditions, including extreme temperatures and the use of strong bases or acids. Furthermore, many established protocols rely on transition metal catalysts such as palladium or copper, which introduce significant cost burdens due to the price of the metals and the stringent requirements for their removal to meet pharmaceutical purity standards. Additionally, methods utilizing organolithium reagents or boronic acids, while effective for specific substrates, suffer from limited functional group tolerance and sensitivity to moisture and air, making them impractical for industrial manufacturing environments where safety and simplicity are paramount.
The Novel Approach
In stark contrast to these legacy techniques, the methodology disclosed in patent CN111454226B offers a streamlined, atom-economical alternative that bypasses these historical bottlenecks. The core innovation lies in the direct oxidative coupling of aromatic aldehydes with ammonium iodide, catalyzed by inexpensive ferric chloride. This approach not only simplifies the raw material sourcing—shifting from specialized nitriles to commodity aldehydes—but also operates under an aerobic atmosphere, utilizing oxygen from the air as the terminal oxidant. 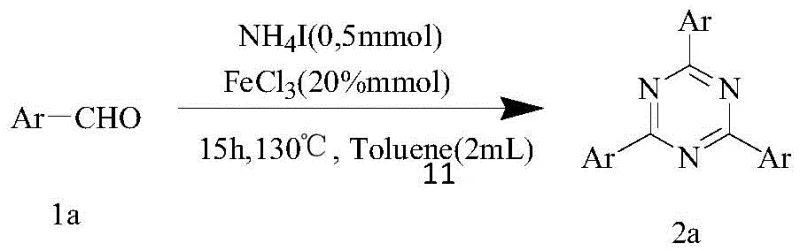 This shift enables a true one-pot synthesis where the C-N bond formation and subsequent cyclization occur seamlessly. The result is a process that is not only chemically elegant but also economically superior, providing a viable pathway for cost reduction in fine chemical manufacturing by minimizing reagent costs and waste disposal fees associated with heavy metal catalysts.
This shift enables a true one-pot synthesis where the C-N bond formation and subsequent cyclization occur seamlessly. The result is a process that is not only chemically elegant but also economically superior, providing a viable pathway for cost reduction in fine chemical manufacturing by minimizing reagent costs and waste disposal fees associated with heavy metal catalysts.
Mechanistic Insights into FeCl3-Catalyzed Oxidative Cyclization
Understanding the mechanistic underpinnings of this transformation is crucial for R&D teams aiming to optimize the process for specific substrates. The reaction proceeds through a sophisticated redox cycle involving the interplay between ferric ions and iodide species. Initially, the aromatic aldehyde undergoes nucleophilic addition with ammonium iodide to form an imine intermediate. Crucially, the ferric chloride acts as a Lewis acid to activate the carbonyl group while simultaneously participating in a redox cycle where Fe(III) oxidizes iodide ions to molecular iodine. This generated iodine species facilitates the dehydrogenation of the intermediate, driving the equilibrium towards the formation of the reactive imine species necessary for cyclization. 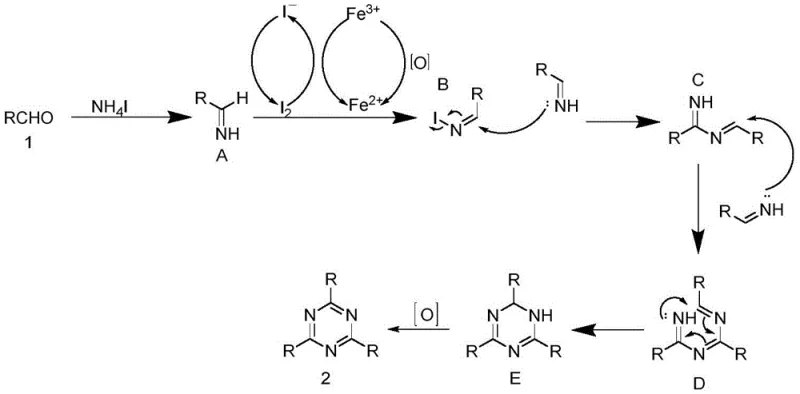 The subsequent steps involve the nucleophilic attack of the imine nitrogen on the electrophilic carbon of another imine molecule, leading to the formation of a linear trimer which then undergoes intramolecular cyclization and final oxidative aromatization to yield the stable triazine ring.
The subsequent steps involve the nucleophilic attack of the imine nitrogen on the electrophilic carbon of another imine molecule, leading to the formation of a linear trimer which then undergoes intramolecular cyclization and final oxidative aromatization to yield the stable triazine ring.
From an impurity control perspective, this mechanism offers distinct advantages. The reliance on an aerobic atmosphere ensures that any partially reduced intermediates are efficiently oxidized to the final aromatic product, minimizing the accumulation of tetrahydro-triazine byproducts. Furthermore, the use of ammonium iodide serves a dual purpose: it acts as the nitrogen source and provides the iodide ions necessary for the redox mediator cycle. This dual functionality reduces the complexity of the reaction mixture, thereby simplifying the downstream purification process. For quality control laboratories, this means a cleaner crude reaction profile, allowing for easier isolation of the target pharmaceutical intermediates with high purity specifications without the need for extensive chromatographic separation or recrystallization steps that often plague traditional triazine syntheses.
How to Synthesize 2,4,6-Triphenyl-1,3,5-triazine Efficiently
The practical implementation of this synthesis is remarkably straightforward, designed to be accessible even in standard laboratory settings without the need for specialized gloveboxes or high-pressure equipment. The protocol involves mixing the aromatic aldehyde substrate with a stoichiometric amount of ammonium iodide and a catalytic loading of ferric chloride in a benzene-based solvent, preferably toluene. The reaction is then heated to 130°C for approximately 15 hours under an open or air-filled system. This simplicity belies the chemical sophistication of the transformation, offering a robust route to symmetrical triazines. For detailed operational parameters and specific workup procedures, please refer to the standardized guide below.
- Combine aromatic aldehyde (0.5 mmol), ammonium iodide (0.5 mmol), and anhydrous ferric chloride (20 mol%) in toluene (2 mL) within a sealed reaction vessel.
- Heat the reaction mixture to 130°C under an air atmosphere with magnetic stirring for 15 hours to facilitate oxidative cyclization.
- Upon completion, cool the mixture, wash with saturated NaCl, extract with ethyl acetate, dry over sodium sulfate, and purify via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this iron-catalyzed methodology presents a compelling value proposition centered on risk mitigation and cost efficiency. By shifting the raw material base from specialized nitriles or organometallics to widely available aromatic aldehydes, companies can leverage existing commodity supply chains, thereby reducing exposure to price volatility and supply disruptions. The elimination of precious metal catalysts further decouples production costs from the fluctuating markets of palladium or platinum, ensuring more predictable budgeting for long-term projects. Moreover, the one-pot nature of the reaction significantly reduces solvent consumption and processing time, contributing to a leaner manufacturing footprint.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the substitution of expensive reagents with low-cost commodities. Aromatic aldehydes and ammonium salts are produced on a massive global scale, ensuring competitive pricing and ready availability. Furthermore, the replacement of precious metal catalysts with ferric chloride—a ubiquitous and inexpensive industrial chemical—drastically lowers the bill of materials. The absence of ligands and the ability to run the reaction under air rather than inert gas also reduce utility costs associated with nitrogen or argon purging, leading to substantial overall cost savings in the production of high-value heterocyclic intermediates.
- Enhanced Supply Chain Reliability: Supply chain resilience is significantly bolstered by the simplicity of the reagent profile. Since the key starting materials are bulk chemicals rather than custom-synthesized fine chemicals, the risk of single-source dependency is minimized. The robustness of the reaction conditions, which tolerate a wide range of functional groups and do not require strictly anhydrous environments, allows for greater flexibility in manufacturing locations. This flexibility ensures that production can be scaled or shifted across different facilities without the need for specialized infrastructure, guaranteeing consistent delivery schedules for critical pharmaceutical intermediates and maintaining continuity for downstream clients.
- Scalability and Environmental Compliance: From an environmental and scalability standpoint, this method aligns perfectly with modern green chemistry principles. The use of iron, a non-toxic and abundant metal, eliminates the regulatory burden and disposal costs associated with heavy metal waste streams. The reaction generates minimal inorganic waste, primarily consisting of benign iron salts and ammonium byproducts, which are easier to treat than the complex organic waste generated by organolithium or palladium-catalyzed routes. This simplified waste profile facilitates easier permitting for larger-scale reactors, enabling seamless transition from kilogram-scale development to multi-ton commercial production while maintaining strict adherence to environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the scope and limitations of this synthesis method, derived directly from the experimental data provided in the patent documentation. These insights are intended to assist technical teams in evaluating the feasibility of this route for their specific target molecules. Understanding the nuances of substrate tolerance and reaction conditions is essential for successful process integration.
Q: What is the optimal catalyst loading for this triazine synthesis?
A: According to patent CN111454226B, the optimal catalyst loading is 20 mol% of ferric chloride (FeCl3). Increasing the loading beyond this point (e.g., to 30%) does not improve yield and may lead to decreased efficiency.
Q: Can this method tolerate electron-withdrawing groups on the aromatic ring?
A: Yes, the reaction demonstrates excellent substrate tolerance. Aromatic aldehydes substituted with electron-withdrawing groups such as halogens (fluorine, chlorine, bromine) and trifluoromethyl groups successfully undergo cyclization to form the corresponding triazine derivatives with good yields.
Q: Is an inert atmosphere required for this reaction?
A: No, an inert atmosphere is not required. In fact, the reaction relies on an oxygen-containing atmosphere (air or pure oxygen) to drive the final oxidative aromatization step. Reactions conducted under nitrogen atmosphere result in significantly lower yields (<5%).
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,4,6-Triphenyl-1,3,5-triazine Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this iron-catalyzed synthesis technology for the global supply of nitrogen heterocycles. As a premier CDMO partner, we possess the technical expertise to rapidly adapt and optimize this novel pathway for diverse client needs. Our facilities are equipped to handle the specific thermal and atmospheric requirements of this process, ensuring that the transition from lab-scale discovery to commercial manufacturing is seamless. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, coupled with stringent purity specifications enforced by our rigorous QC labs to guarantee that every batch meets the exacting standards required for pharmaceutical and electronic applications.
We invite potential partners to engage with our technical team to explore how this cost-effective synthesis route can be integrated into your supply chain. Whether you require custom synthesis of specific triazine derivatives or large-scale toll manufacturing, we are prepared to provide a Customized Cost-Saving Analysis tailored to your project. Please contact our technical procurement team today to request specific COA data and comprehensive route feasibility assessments, and let us demonstrate how we can become your trusted partner in delivering high-quality chemical solutions.
